

烯胺酮平台构建转化生物质产品Cyrene为增值化合物
收稿日期: 2022-12-03
修回日期: 2023-01-06
网络出版日期: 2023-01-18
基金资助
国家自然科学基金(22161022)
Engineering Biomass Feedstock Cyrene to Value-Added Compounds by Enaminone Platform Construction
Received date: 2022-12-03
Revised date: 2023-01-06
Online published: 2023-01-18
Supported by
National Natural Science Foundation of China(22161022)
黄丽珠 , 刘云云 , 万结平 . 烯胺酮平台构建转化生物质产品Cyrene为增值化合物[J]. 有机化学, 2023 , 43(6) : 2096 -2103 . DOI: 10.6023/cjoc202212002
The enamination of Cyrene in the ketone α-site leading to the enamino Cyrene (EC) has been developed. The practical preparation of EC as a platform synthon opens broad space of chemical transformation for Cyrene biomass feedstock in the synthesis of diverse value-added chemicals. In the present work, the transformation of the EC for the synthesis of many different chiral organic derivatives, including NH-/N,N-disubstituted ECs, the cyanoalkenyl- and vinyl phosphine oxide-elabo- rated Cyrene, Cyrene fused furan, and 2,2-difluorodihydrofuran fused Cyrene, has been realized.
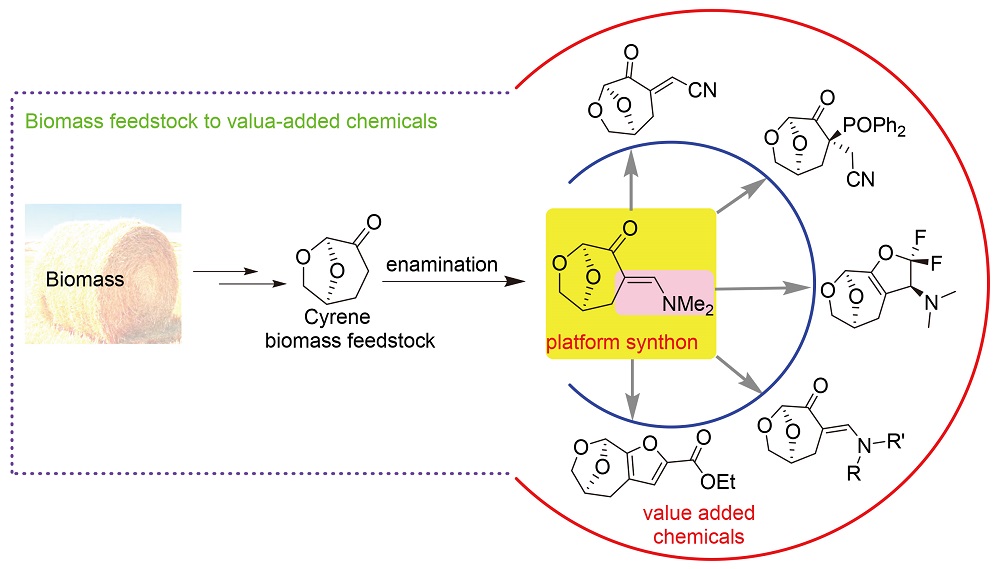
Key words: Cyrene; biomass feedstock; enamination; platform; value-added
| [1] | (a) Sherwood, J.; De bruyn, M.; Constantinou, A.; Moity, L.; McElroy, C. R.; Farmer, T. J.; Duncan, T.; Raverty, W.; Hunt, A. J.; Clark, J. H. Chem. Commun. 2014, 50, 9650. |
| [1] | (b) Craythorne, S. J.; Anderson, K.; Lorenzini, F.; McCausland, C.; Smith, E. F.; Licence, P.; Marr, A. C.; Marr, P. C. Chem.-Eur. J. 2009, 15, 7094. |
| [1] | (c) De bruyn, M.; Fan, J.; Budarin, V. L.; Macquarrie, D. J.; Gomez, L. D.; Simister, R.; Farmer, T. J.; Raverty, W. D.; McQueen-Mason, S. J.; Clark, J. H. Energy Environ. Sci. 2016, 9, 2571. |
| [1] | (d) Miura, M.; Kaga, H.; Yoshida, T.; Ando, K. J. Wood Sci. 2001, 47, 502. |
| [1] | (e) Sui, X.-W.; Wang, Z.; Liao, B.; Zhang, Y.; Guo, Q.-X. Bioresour. Technol. 2012, 103, 466. |
| [2] | See also information in Merck product introduction: https://www.merckgroup.com/en/research/science-space/envisioning-tomorrow/scarcity-of-resources/cyrene.html. |
| [3] | Camp, J. E. ChemSusChem 2018, 11, 3048. |
| [4] | (a) Duval, A.; Avérous, L. Green Chem. 2022, 24, 338. |
| [4] | (b) Meng, X.; Pu, Y.; Li, M.; Ragauskas, A. J. Green Chem. 2020, 22, 2862. |
| [4] | (c) Marathianos, A.; Liarou, E.; Hancox, E.; Grace, J. L.; Lester, D. W.; Haddleton, D. M. Green Chem. 2020, 22, 5833. |
| [4] | (d) Gao, F.; Bai, R.; Ferlin, F.; Vaccaro, L.; Li, M.; Gu, Y. Green Chem. 2020, 22, 6240. |
| [4] | (e) Brouwer, T.; Schuur, B. ACS Sustainable Chem. Eng. 2020, 8, 14807. |
| [5] | (a) Bousfield, T. W.; Pearce, K. P. R.; Nyamini, S. B.; Angelis-Dimakis, A.; Camp, J. E. Green Chem. 2019, 21, 3675. |
| [5] | (b) Mistry, L.; Mapesa, K.; Bousfield, T. W.; Jason, E. C. Green Chem. 2017, 19, 2123 |
| [6] | Yu, H.; Yu, D.; Xue, Z.; Zhang, B.; Mu, T. Chem. Eng. J. 2022, 431, 133397. |
| [7] | (a) Wilson, K. L.; Kennedy, A. R.; Murray, J.; Greatrex, B.; Jamieson, C.; Watson, A. J. B. Beilstein J. Org. Chem. 2016, 12, 2005. |
| [7] | (b) Wilson, K. L.; Murray, J.; Jamieson, C.; Watson, A. J. B. Synlett 2018, 29, 650. |
| [8] | Mention, M. M.; Flourat, A. L.; Peyrot, C.; Allais, F. Green Chem. 2020, 22, 2077. |
| [9] | Gonzalo, G. Biocatal. Biotransfor. 2022, 40, 252. |
| [10] | Tamargo, R. J. I.; Rubio, P. Y. M.; Mohandoss, S.; Shim, J.-J.; Lee, Y. R. ChemSusChem 2021, 14, 2133. |
| [11] | Ray, P.; Hughes, T.; Smith, C.; Hibbert, M.; Saito, K.; Simon, G. P. Polym. Chem. 2019, 10, 3334. |
| [11] | (b) Diot-Néant, F.; Mouterde, L.; Fadlallah, S.; Miller, S. A.; Allais, F. ChemSusChem 2020, 13, 2613. |
| [12] | De bruyn, M.; Budarin, V. L.; Misefari, A.; Shimizu, S.; Fish, H.; Cockett, M.; Hunt, A. J.; Hofsteffter, H.; Weckhuysen, B. M.; Clark, J. H.; Macquarrie, D. J. ACS Sustainable Chem. Eng. 2019, 7, 7878. |
| [13] | Bonneau, G.; Peru, A. A. M.; Flourat, A. L.; Allais, F. Green Chem. 2018, 20, 2455. |
| [14] | Krishna, S. H.; McClelland, D. J.; Rashke, Q. A.; Dumesic, J. A.; Huber, G. W. Green Chem. 2017, 19, 1278. |
| [15] | Hughes, L.; McElroy, C. R.; Whitwood, A. C.; Hunt, A. J. Green Chem. 2018, 20, 4423. |
| [16] | Alhifthi, A.; Harris, B. L.; Goerigk, L.; White, J. M.; Williams, S. J. Org. Biomol. Chem. 2017, 15, 10105. |
| [17] | (a) Wang, Z.; Zhao, B.; Liu, Y.; Wan, J.-P. Adv. Synth. Catal. 2022, 364, 1508. |
| [17] | (b) Zheng, X.; Liu, Y.; Wan, J.-P. Chin. J. Org. Chem. 2021, 41, 2700. |
| [17] | (c) Guo, H.; Liu, Y.; Wen, C.; Wan, J.-P. Green Chem. 2022, 24, 5058. |
| [17] | (c) Fu, L.; Xu, W.; Pu, M.; Wu, Y-D.; Liu, Y.; Wan, J.-P. Org. Lett. 2022, 24, 3003. |
| [17] | (d) Guo, Y.; Liu, Y.; Wan, J.-P. Chin. Chem. Lett. 2022, 33, 855. |
| [17] | (d) Yu, Q.; Liu, Y.; Wan, J.-P. Chin. Chem. Lett. 2021, 32, 3514. |
| [17] | (e) Duan, X.; Li, H.; Li, W.; Wang, J.; Liu, N. ChemistrySelect 2021, 6, 6478. |
| [17] | (f) Zhang, B.; Fu, Z.; Yang, H.; Liu, D.; Sun, Y.; Xu, Y.; Yu, F.; Yan, S. Adv. Synth. Catal. 2022, 364, 1602. |
| [17] | (g) Fu, R.; Liu, Y.; Wu, T.; Zhang, X.; Zhu, Y.; Luo, J.; Zhang, Z.; Jiang, Y. Chem. Commun. 2022, 58, 3525. |
| [17] | (h) Wu, Y.; Du, J.; Hao, W.; Jiang, B. Chin. J. Org. Chem. 2021, 41, 1563. (in Chinese) |
| [17] | (吴亚男, 杜建宇, 郝文娟, 姜波, 有机化学, 2021, 41, 1563.) |
| [17] | (i) Xie, Y.-Y.; Wang, Y.-C.; Qu, H.-E.; Tan, X.-C.; Wang, H.-S.; Pan, Y.-M. Adv. Synth. Catal. 2014, 356, 3347. |
| [18] | CCDC 2201314 and 2201818 contain the crystallographic data associated with this work. |
| [19] | The relative configuration of the chiral centres in products 8 and 9 were assigned by NOE NMR analysis. |
/
| 〈 |
|
〉 |