

苯并磺内酰胺联吡唑化合物的简便合成
收稿日期: 2022-09-09
修回日期: 2022-12-14
网络出版日期: 2023-02-07
基金资助
国家重点研发计划(2017YFD0200301)
A Convenient Approach to Benzosultam-Linked Pyrazole Compounds
Received date: 2022-09-09
Revised date: 2022-12-14
Online published: 2023-02-07
Supported by
National Key Research and Development Program of China(2017YFD0200301)
雷容超 , 兰文捷 , 李梦竹 , 傅滨 . 苯并磺内酰胺联吡唑化合物的简便合成[J]. 有机化学, 2023 , 43(7) : 2553 -2560 . DOI: 10.6023/cjoc202209009
A base-mediated rapid synthesis of benzosultam-linked pyrazole compounds was developed. The reaction of N-sulfonyl ketimine with pyrazolin-5-one proceeded smoothly to provide the desired products in high yields in the presence of Et3N and Ac2O. The possible mechanism was proposed. This reaction could be conducted on a gram-scale with maintained high yield.
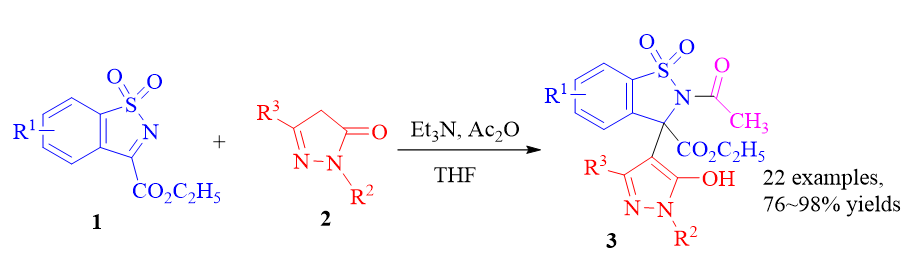
Key words: pyrazole; benzosultam; Mannich addition; pyrazolone
| [1] | Takeuchi, Y.; Liu, Z. Heterocycles 2002, 56, 693. |
| [2] | Mao, J.; Baker, D. C. US 6458962, 2003. |
| [3] | (a) Castro Pineiro, J. L.; Collins, I. J.; Harrison, T. US 20050014369, 2005. |
| [3] | (b) Supuran, C. T.; Casini, A.; Scozzafava, A. Med. Res. Rev. 2003, 23, 535 |
| [4] | (a) Zhang, H.; Sun, M.-C.; Yin, L.-M.; Wei, D. H.; Song, M.-P.; Yang, D. D.; Niu, J.-L. Org. Chem. Front. 2021, 8, 6888. |
| [4] | (b) Li, R.-L.; Fang, Q.-Y.; Li, M.-Y.; Wang, X.-S.; Zhao, L.-M. Chem. Commun. 2021, 57, 11322. |
| [4] | (c) Majumdar, K. C.; Shovan, M. Chem. Rev. 2011, 111, 7749. |
| [5] | (a) Sun, R.; Qiu, Z. X.; Cao, G. R.; Teng, D. W. Tetrahedron 2020, 76, 131201. |
| [5] | (b) Nishimura, T.; Noishiki, A.; Tsui, G. C.; Hayashi, T. J. Am. Chem. Soc. 2012, 134, 5056. |
| [5] | (c) Wang, H.; Jiang, T.; Xu, M.-H. J. Am. Chem. Soc. 2013, 135, 971. |
| [5] | (d) Qiao, B.; Huang, Y.-J.; Nie, J.; Ma, J.-A. Org. Lett. 2015, 17, 4608. |
| [5] | (e) Yu, J.-S.; Zhou, J. Org. Chem. Front. 2016, 3, 298. |
| [6] | (a) Watanabe, T.; Yuki, S.; Egawa, M.; Nishi, H. J. Pharmacol. Exp. Ther. 1994, 268, 1597. |
| [6] | (b) Kawai, H.; Nakai, H.; Suga, M.; Yuki, S.; Watanabe, T.; Saito, K. I. J. Pharmacol. Exp. Ther. 1997, 281, 921. |
| [7] | Wu, T. W.; Zeng, L. H.; Wu, J.; Fung, K. P. Life Sci. 2002, 71, 2249. |
| [8] | Shiohara, H.; Fujikura, H.; Fushimi, N.; Ito, F.; Isaji, M. WO 2002098893, 2002. |
| [9] | (a) Liu, S.; Bao, X.; Wang, B. Chem. Commun. 2018, 54, 11515. |
| [9] | (b) Hack, D.; Chauhan, P.; Decker, S. K.; Mizutani, Y.; Raabe, G.; Enders, D. Chem. Commun. 2015, 51, 2266. |
| [9] | (c) Yetra, S. R.; Mondal, S.; Suresh, E.; Biju, A. Org. Lett. 2015, 17, 1417. |
| [9] | (d) Zheng, Y.; Cui, L.; Wang, Y.; Zhou, Z. J. Org. Chem. 2016, 81, 4340. |
| [9] | (e) Chauhan, P.; Mahajan, S.; Kaya, U.; Peuronen, A.; Rissanen, K.; Enders, D. J. Org. Chem. 2017, 82, 705. |
| [9] | (f) Maity, R.; Gharui, C.; Sil, A. K.; Pan, S. C. Org. Lett. 2017, 19, 662. |
| [9] | (g) Cheng, C., Zhang, J.; Wang, X.; Miao, Z. J. Org. Chem. 2018, 83, 5450. |
| [9] | (h) Vila, C.; Amr, F. I.; Blay, G.; Muňoz, M. C.; Pedro, J. R. Chem.- Asian J. 2016, 11, 1532. |
| [9] | (i) Rao, K. S.; Ramesh, P.; Trivedi, R.; Kantam, M. L. Tetrahedron Lett. 2016, 57, 1227. |
| [10] | (a) Liao, Y. H.; Chen, W. B.; Wu, Z. J.; Du, X. L.; Cun, L. F.; Zhang, X. M.; Yuan, W. C. Adv. Synth. Catal. 2010, 352, 827. |
| [10] | (b) Meshram, H. M.; Kumar, N. S.; Nanubolu, J. B.; Rao, L. C.; Rao, N. N. Tetrahedron Lett. 2013, 54, 5941. |
| [10] | (c) Li, J. H.; Du, D. M. Org. Biomol. Chem. 2013, 11, 6215. |
| [10] | (d) Wang, H.; Wang, Y.; Song, H.; Zhou, Z.; Tang, C. Eur. J. Org. Chem. 2013, 2013, 4844. |
| [10] | (e) Zheng, Y.; Cui, L.; Wang, Y.; Zhou, Z. J. Org. Chem. 2016, 81, 4340. |
| [10] | (f) Hack, D.; Du?rr, A. B.; Deckers, K.; Chauhan, P.; Seling, N.; Ru?benach, L.; Mertens, L.; Raabe, G.; Schoenebeck, F.; Enders, D. Angew. Chem.,Int. Ed. 2016, 55, 1797. |
| [10] | (g) Rana, N. K.; Jha, R. K.; Joshi, H.; Singh, V. K. Tetrahedron Lett. 2017, 58, 2135. |
| [10] | (h) Xu, Y.; Sun, P.; Song, Q.; Lai, X.; Liu, W.; Xia, T.; Huang, Y. Y.; Shen, Y. Eur. J. Org. Chem. 2017, 2017, 2998. |
| [11] | (a) Gogoi, S.; Zhao, C. G.; Ding, D. Org. Lett. 2009, 11, 2249. |
| [11] | (b) Alba, A. N.; Zea, A.; Valero, G.; Calbet, T.; Font-Bardía, M.; Mazzanti, A.; Moyano, A.; Rios, R. Eur. J. Org. Chem. 2011, 2011, 1318. |
| [11] | (c) Enders, D.; Grossmann, A.; Gieraths, B.; Du?zdemir, M.; Merkens, C. Org. Lett. 2012, 14, 4254. |
| [11] | (d) Li, J. H.; Du, D. M. RSC Adv. 2014, 4, 14538. |
| [11] | (e) Yetra, S. R.; Mondal, S.; Suresh, E.; Biju, A. T. Org. Lett. 2015, 17, 1417. |
| [11] | (f) Kumarswamyreddy, N.; Kesavan, V. Org. Lett. 2016, 18, 1354. |
| [12] | Mazzanti, A.; Calbet, T.; Font-Bardia, M.; Moyano, A.; Rios, R. Org. Biomol. Chem. 2012, 10, 1645. |
| [13] | (a) Gogoi, S.; Zhao, C. G. Tetrahedron Lett. 2009, 50, 2252. |
| [13] | (b) Xie, J.; Xing, X. Y.; Sha, F.; Wu, Z. Y.; Wu, X. Y. Org. Biomol. Chem. 2016, 14, 8346. |
| [14] | (a) Keyume, A.; Esmayil, Z.; Wang, L. J.; Jun, F. Tetrahedron 2014, 70, 3976. |
| [14] | (b) Fan, W.-T.; Yang, X.-P.; Yang, H.-P.; Lv, H.-P.; Wang, X.-W.; Wang, Z. Org. Lett. 2020, 22, 3936. |
| [14] | (c) Bao, X. Z.; Wei, S.-Q.; Qu, J.-P.; Wang, B. M. Chem. Commun. 2018, 54, 2028. |
| [14] | (d) Li, L.; Liu, Z.-T.; Hu, X.-P. Chem. Commun. 2018, 54, 12033. |
| [14] | (e) Ni, C. J.; Tong, X. F. J. Am. Chem. Soc. 2016, 138, 7872. |
| [15] | (a) Feng, J.; Ablajan, K.; Sali, A. Tetrahedron 2014, 70, 484. |
| [15] | (b) Tan, C.-Y.; Lu, H.; Zhang, J.-L.; Liu, J.-Y.; Xu, P.-F. J. Org. Chem. 2020, 85, 594. |
| [15] | (c) Xu, Y.-W.; Li, L.; Hu, X.-P. Org. Lett. 2020, 22, 9534. |
| [15] | (d) Tang, C.-K.; Zhou, Z.-Y.; Xia, A.-B.; Bai, L.; Liu, J.; Xu, D.-Q.; Xu, Z.-Y. Org. Lett. 2018, 20, 5840. |
| [15] | (e) Kale, S. B.; Jori, P. K.; Thatikonda, T.; Gonnade, R. G.; Das, U. Org. Lett. 2019, 21, 7736. |
| [15] | (f) Hack, D.; Dürr, A. B.; Deckers, K.; Chauhan, P.; Seling, N.; Rüenach, L.; Mertens, L.; Raabe, G.; Schoenebeck, F.; Enders, D. Angew. Chem., nt. Ed. 2016, 55, 1797. |
| [16] | (a) Lan, W. J.; Lei, R. C.; Luo, J. Y.; Qin, Z. H.; Fu, B.; Xie, L. ChemistrySelect 2022, 7, 3670. |
| [16] | (b) Xie, L.; Ma, H.-L; Li, J.-Q.; Yu, X.; Qin, Z. H.; Fu, B. Org. Chem. Front. 2017, 4, 1858. |
| [17] | (a) Li, S.-W.; Wan, Q.; Kang, Q. Org. Lett. 2018, 20, 1312. |
| [17] | (b) Sharma, A.; Sharma, V.; Chimni, S. S. Org. Biomol. Chem. 2019, 17, 9514. |
| [18] | Yan, Z.; Wu, B.; Gao, X.; Chen, M. W.; Zhou, Y. G. Org. Lett. 2016, 18, 692. |
| [19] | (a) Bran?, M. F.; Gradillas, A.; Ovalles, A. G.; Lopez, B.; Acero, N.; Llinares, F.; Mingarro, D. M. Bioorg. Med. Chem. 2006, 14, 9. |
| [19] | (b) Wang, X.-j.; Tan, J.; Grozinger, K. Tetrahedron Lett. 2000, 41, 4713. |
/
| 〈 |
|
〉 |