

新型嘧啶并[5,4-e][1,2,4]三唑并[1,5-c]嘧啶衍生物的合成与除草活性
收稿日期: 2023-01-08
修回日期: 2023-03-01
网络出版日期: 2023-03-31
基金资助
国家自然科学基金(21562026); 国家自然科学基金(21762025); 江西师范大学青年英才资助项目.
Synthesis and Herbicidal Activity of Novel Pyrimido[5,4-e]-[1,2,4]triazolo[1,5-c]pyrimidine Derivatives
Received date: 2023-01-08
Revised date: 2023-03-01
Online published: 2023-03-31
Supported by
National Natural Science Foundation of China(21562026); National Natural Science Foundation of China(21762025); Young Talents Program of Jiangxi Normal University.
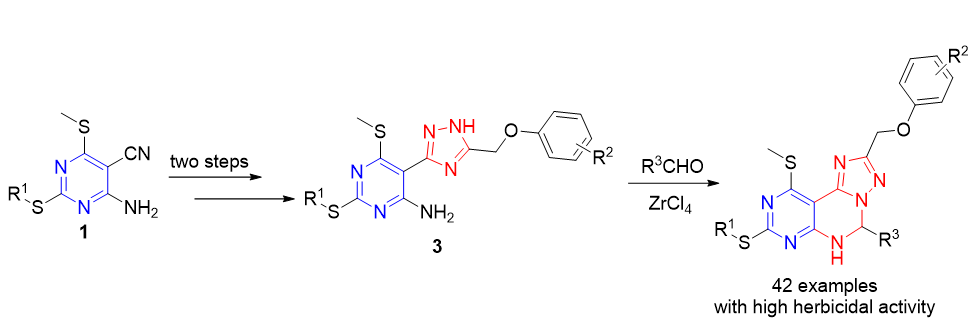
为了寻找结构新颖且具有优异除草活性的含氮稠杂环化合物, 以2-烷硫基-5-氰基-6-硫甲基嘧啶-4-胺为原料合成了42个新型的嘧啶并[5,4-e][1,2,4]三唑并[1,5-c]嘧啶衍生物消旋体(Ι-1~Ι-42), 其结构经熔点、1H NMR、13C NMR、红外吸收光谱法(IR)和高分辨质谱(HRMS)确证. 初步的除草活性测试表明, 在浓度为100 mg/L时, 大部分化合物对单子叶植物(小麦、稗草和高粱)和双子叶植物(萝卜、油菜和黄瓜)表现出显著的除草活性, 特别是化合物Ι-13, Ι-14, Ι-23, Ι-25和Ι-30~Ι-42对6种供试植物的根和茎长抑制率都达到了100%.
关键词: 嘧啶并[5,4-e][1,2,4]三唑并[1,5-c]嘧啶; 合成; 除草活性
林海 , 聂会祥 , 赵安林 , 王涛 , 罗劲 . 新型嘧啶并[5,4-e][1,2,4]三唑并[1,5-c]嘧啶衍生物的合成与除草活性[J]. 有机化学, 2023 , 43(7) : 2462 -2475 . DOI: 10.6023/cjoc202301008
To discover N-containing fused heterocyclic compounds with novel structure and excellent herbicidal activity, forty-two pyrimido[5,4-e][1,2,4]triazolo[1,5-c]pyrimidine derivatives (Ι-1~Ι-42) were synthesied using 2-(alkylthio)-5- carbonitrile-6-(methylthio)pyrimidine-4-aminos as starting materials. Their structures were clearly confirmed by melting point, 1H NMR, 13C NMR, infrared absorption spectroscopy (IR), and high-resolution mass spectra (HRMS). The preliminary bioassay indicated that many target compounds exhibited remarkable herbicidal activities against the monocotyledons (Triticum aestivum L., Echinochloa crusgalli, and Sorghum bicolor) and dicotyledons (Raphanus sativus, Brassica campestris, and Cucumis sativus) at the concentration of 100 mg/L. Especially, compounds Ι-13, Ι-14, Ι-23, Ι-25 and Ι-30~Ι-42 showed excellent herbicidal activities against the roots and stalks of the six test plant at 100 mg/L with 100% inhibition rates.

| [1] | Navjeet, K.; Neha, A.; Yamini, V.; Pooja, G.; Pranshu, B.; Nirmala, J. K. Curr. Org. Chem. 2021, 25, 1270. |
| [2] | Eftekhari-Sis, B.; Zirak, M.; Akbari, A. Chem. Rev. 2013, 113, 2958. |
| [3] | Yadagiri, D.; Rivas, M.; Gevorgyan, V. J. Org. Chem. 2020, 85, 11030. |
| [4] | Kerru, N.; Gummidi, L.; Maddila, S.; Gangu, K. K.; Jonnalagadda, S. B. Molecules 2020, 25, 1909. |
| [5] | Gao, B.; Yang, B.; Feng, X. D.; Li, C. Nat. Prod. Rep. 2022, 39, 139. |
| [6] | Pedrood, K.; Montazer, M. N.; Larijani, B.; Mahdavi, M. Synthesis 2021, 53, 2342. |
| [7] | Sbei, N. Listratova, A. V.; Titov, A. A.; Voskressensky, L. G. Synthesis 2019, 51, 2455. |
| [8] | Lv, Z. C.; Wang, H. M.; Quan, Z. C.; Gao, Y.; Lei, A. W. Chem. Commun. 2019, 55, 12332. |
| [9] | Liu, Z. K.; Zhao, Q. Q.; Gao, Y.; Hou, Y. X.; Hu, X. Q. Adv. Synth. Catal. 2021, 363, 411. |
| [10] | Chen, X.; Yang, H. Y.; Hu?lsey, M. J.; Yan, N. ACS Sustainable Chem. Eng. 2017, 5, 11096. |
| [11] | Dastan, A.; Kulkarni, A.; T?r?k, B. Green Chem. 2012, 14, 17. |
| [12] | Romdhane, A.; Said, A. B.; Cherif, M.; Jannet, H. B. Med. Chem. Res. 2016, 25, 1358. |
| [13] | Hassan, A. Y.; Abdel-Aziem, A.; Hussain, A. O. J. Heterocycl. Chem. 2020, 57, 542. |
| [14] | Hidalgo, F. J.; Zamora, R. J. Agric. Food Chem. 2022, 70, 79. |
| [15] | Saranya, P. V.; Neetha, M.; Radhika, S.; Anilkumar, G. J. Heterocycl. Chem. 2021, 58, 673. |
| [16] | Wu, P.; Zhang, Y.; Cheng, Y. J. Org. Chem. 2022, 87, 2779. |
| [17] | Li, M.; Wang, Y.; Xu, Y. H. Chin. J. Org. Chem. 2021, 41, 3073 (in Chinese). |
| [17] | (李曼, 汪颖, 徐允河, 有机化学, 2021, 41, 3073.) |
| [18] | Giudice, M. R. D.; Borioni, A.; Mustazza, C.; Gatta, F. J. Heterocycl. Chem. 1994, 31, 1503. |
| [19] | Mohadeszadeh, M.; Rahimizadeh, M.; Eshghi, H. Shiri, A.; Gholizadeh, M.; Shams, A. Helv. Chim. Acta 2015, 98, 474. |
| [20] | Abdelhamid, A. O.; El-Sayed, I. E.; Hussein, M. Z.; Mangoud, M. M. Molecules 2016, 21, 1072. |
| [21] | Sandmann, G. Pestic. Biochem. Phys. 2001, 70, 86. |
| [22] | Zhu, J. J.; He, L. H.; Luo, J.; Xiong, J.; Wang, T. Phosphorus, Sulfur Silicon Relat. Elem. 2021, 196, 948. |
| [23] | Yang, L. Y.; Sun, Y.; Lu, Z. F.; Liang, J. R.; Wang, T.; Luo, J. J. Heterocycl. Chem. 2022, 59, 704. |
| [24] | Yang, L. Y.; Sun, Y.; He, L. H.; Fan., Y. J.; Wang, T.; Luo, J. J. Mol. Struct. 2022, 1259, 132722. |
| [25] | Sun, Q.; Wu, R. Z.; Cai, S. T.; Lin, Y.; Sellers, L.; Sakamoto, K.; He, B.; Peterson, B. R. J. Med. Chem. 2011, 54, 1126. |
| [26] | Zhao, A. L.; Liu, S.; Zhu, Y. M.; Wang, T.; Luo, J. Chin. J. Org. Chem. 2017, 37, 1877 (in Chinese). |
| [26] | (赵安林, 刘姝, 朱咏梅, 王涛, 罗劲, 有机化学, 2017, 37, 1877.) |
/
| 〈 |
|
〉 |