

介孔ETS-10沸石担载Pd高效催化内炔氧化制备1,2-二酮
收稿日期: 2023-01-17
修回日期: 2023-03-03
网络出版日期: 2023-03-31
基金资助
国家自然科学基金(21776022); 国家自然科学基金(22178029)
Pd Supported on Mesoporous ETS-10 Zeolite Catalyst with Superior Catalytic Performances in Synthesizing 1,2-Diones from the Oxidation of Internal Alkynes
Received date: 2023-01-17
Revised date: 2023-03-03
Online published: 2023-03-31
Supported by
The National Natural Science Foundation of China(21776022); The National Natural Science Foundation of China(22178029)
使用担载Pd的介孔ETS-10 (Pd/METS-10)催化剂在清洁的反应条件下, 实现了内炔氧化合成1,2-二酮类衍生物. 研究发现, 催化剂上高分散的Pd(II)和炔烃配位形成电子受体供体复合物(EDX)是反应顺利进行的关键. 随后二甲基亚砜(DMSO)亲核进攻生成的EDX, 相继发生加成和消除反应, 从而生成目的产物. Pd/METS-10催化的内炔氧化反应具有优异的产物选择性和好的底物范围, 能够同时兼容对称/不对称内炔, 且对底物的电子性质和立体效应不敏感. 此外, Pd/METS-10催化剂在反应中表现出优异的重复性能, 在循环使用5次后没有明显的活性损失. 最后, 根据表征结果和控制实验结果, 提出了Pd/METS-10催化的内炔氧化制备1,2-二酮的反应机理.
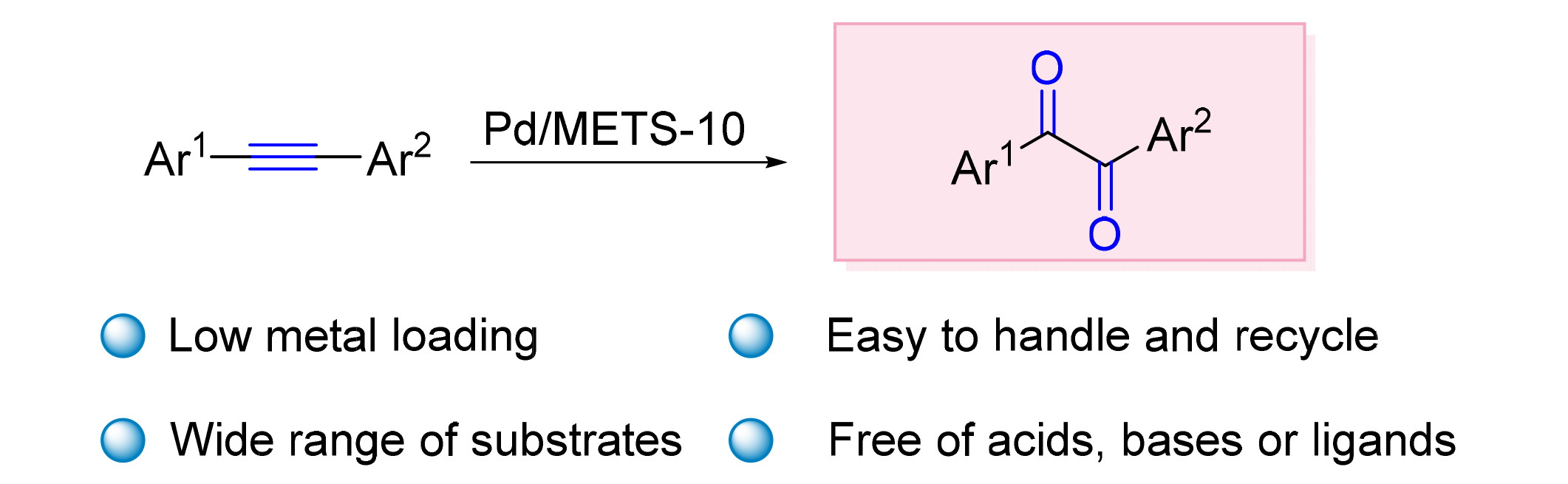
关键词: Pd/METS-10; 内炔; 1,2-二酮类衍生物; 电子供体受体复合物; 氧化反应
刘长俊 , 胡慧玲 , 刘宬宏 , 朱超杰 , 唐天地 . 介孔ETS-10沸石担载Pd高效催化内炔氧化制备1,2-二酮[J]. 有机化学, 2023 , 43(8) : 2953 -2960 . DOI: 10.6023/cjoc202301016
The 1,2-dione compounds was synthesized under clean reaction conditions over Pd/METS-10 catalyst. It has been found that the key step of the protocol is forming an electron donor-acceptor complex (EDX), as the coordination of Pd(II) and alkynyl. Then the nucleophilic addition and elimination reactions of dimethyl sulfoxide (DMSO) to the EDX were occurred, affording the target product. Pd/METS-10 catalyzed endo-acetylene oxidation possesses excellent product selectivity and good substrate scope. It is compatible with both symmetric and asymmetric endo-acetylene and is insensitive to the electronic properties and stereoscopic effects of the substrate. In addition, Pd/METS-10 catalyst showed excellent repetition performance in the reaction, with no significant loss of activity after five cycles. Finally, according to the characterization and control experiment results, the reaction mechanism of Pd/METS-10 catalyzed acetylene oxidation to produce 1,2-dione was proposed.

| [1] | (a) Li, W.; Asada, Y.; Yoshikawa, T. Planta Med. 1998, 64, 746. |
| [1] | (b) Worayuthakarn, R.; Boonya-udtayan, S.; Ruchirawat, S.; Thasana, N. Eur. J. Org. Chem. 2014, 2014, 2496. |
| [2] | Hyatt, J. L.; Stacy, V.; Wadkins, R. M.; Yoon, K. J. P.; Wierdl, M.; Edwards, C. C.; Zeller, M.; Hunter, A. D.; Danks, M. K.; Crundwell, G.; Potter, P. M. J. Med. Chem. 2005, 48, 5543. |
| [3] | (a) Shaikh, M.; Sahu, M.; Gavel, P. K.; Ranganath, K. V. S. Catal. Commun. 2015, 64, 18 |
| [3] | (b) Qiu, S.-G.; Lei, Y.; Wu, Y.; Chen, Y.-X.; Wu, L.-Y. ChemistrySelect 2020, 5, 495 |
| [4] | (a) Yuan, Y.; Zhu, H.-T. Eur. J. Org. Chem. 2012, 37, 329. |
| [4] | (b) Li, Z.; Yin, J.-J.; Wen, G.; Li, T.-P.; Shen, X.-L. RSC Adv. 2014, 4, 32298. |
| [4] | (c) Wang, H.; Ren, S.-B.; Zhang, J.; Zhang, W.; Liu, Y.-K. J. Org. Chem. 2015, 80, 6856. |
| [4] | (d) Chen, L.-S.; Zhang, L.-B.; Tian, Y.; Li, J.-H.; Liu, Y.-Q. Eur. J. Org. Chem. 2020, 32, 5523. |
| [4] | (e) Zhu, J.-L.; Sai, Y.-T. J. Org. Chem. 2021, 86, 813. |
| [5] | (a) Chen, S.-L.; Liu, Z.-J.; Shi, E.-B.; Chen, L.; Wei, W.; Li, H.; Cheng, Y.-N.; Wan, X.-B. Org. Lett. 2011, 13, 2274. |
| [5] | (b) Thiruvengetam, P.; Chand, D. K. J. Org. Chem. 2022, 87, 4061. |
| [6] | Raja, P.; Ghatak, T. J. Mol. Struct. 2023, 1271, 134004. |
| [7] | Sawama, Y.; Takubo, M.; Mori, S.; Monguchi, Y.; Sajiki, H. Eur. J. Org. Chem. 2011, 24, 3361. |
| [8] | Byun, S.; Chung, J.; Lim, T.; Kwon, J.; Kim, B. M. RSC Adv. 2014, 4, 34084. |
| [9] | Song, J.-X.; Zhang, K.; Huang, Z.-Y.; Zhao, J.-Y.; Yang, Z.-Y.; Zong, L.-B.; Chen, J.-B.; Xie, C.-X.; Jia, X.-F. Catal. Sci. Technol. 2022, 12, 722. |
| [10] | (a) Li, Y.; Li, L.; Yu, J.-H. Chem 2017, 3, 928. |
| [10] | (b) Sun, Q.-M.; Wang, N.; Yu, J.-H. Adv. Mater. 2021, 33, 2104442. |
| [11] | (a) Das, T. K.; Chandwadkar, A. J.; Sivasanker, S. Chem. Commun. 1996, 10, 1105. |
| [11] | (b) Krisnandi, Y. K.; Lachowski, E. E.; Howe, R. F. Chem. Mater. 2006, 18, 928. |
| [12] | (a) Pavel, C. C.; Palkovits, R.; Schüth, F.; Schmidt, W. J. Catal. 2008, 254, 84. |
| [12] | (b) Ni, X.-J.; Xiang, M.; Fu, W.-Q.; Ma, Y.-L.; Zhu, P.-Y.; Wang, W.-C.; He, M.-Y.; Yang, K.-Q.; Xiong, J.; Tang, T.-D. J. Porous Mater. 2016, 23, 423. |
| [13] | Xiang, M.; Ni, X.-J.; Yi, X.-F.; Zheng, A.-M.; Wang, W.-C.; He, M.-Y.; Xiong, J.; Liu, T-T.; Ma, Y.-L.; Zhu, P.-Y.; Zheng, X.; Tang, T.-D. ChemCatChem 2015, 7, 521. |
| [14] | (a) Kaplum, V.; Cogo, J.; Sangi, D.P.; Ueda-Nakamura, T.; Correa, A. G.; Nakamura, C. V. Antimicrob. Agents Chemother. 2016, 60, 3433. |
| [14] | (b) Wang, Y.-B.; Shi, L.-L.; Zhang, X.-J.; Fu, L.-R.; Hu, W.-N.; Zhang, W.-J.; Zhu, X.-J.; Hao, X.-Q.; Song, M.-P. J. Org. Chem. 2021, 86, 947. |
| [15] | (a) Sawama, Y.; Takubo, M.; Mori, S.; Monguchi, Y.; Sajiki, H. Eur. J. Org. Chem. 2011, 2011, 3361. |
| [15] | (b) Gao, A.; Yang, F.; Li, J.; Wu, Y.-J. Tetrahedron 2012, 68, 4950. |
| [15] | (c) Shaik, J. B.; Ramkumar, V.; Sankararaman, S. J. Organomet. Chem. 2018, 860, 1. |
/
| 〈 |
|
〉 |