

异喹啉-3-羧酸根螯合的B,B-二芳基四配位硼络合物的合成
收稿日期: 2023-01-02
修回日期: 2023-03-01
网络出版日期: 2023-04-14
基金资助
国家自然科学基金(21963010); 国家自然科学基金(22061036)
Synthesis of Isoquinoline-3-carboxylate Chelated B,B-Diaryl Tetracoordinated Organoboron Complexes
Received date: 2023-01-02
Revised date: 2023-03-01
Online published: 2023-04-14
Supported by
National Natural Science Foundation of China(21963010); National Natural Science Foundation of China(22061036)
田维娜 , 徐亮 , 韦玉 , 李鹏飞 . 异喹啉-3-羧酸根螯合的B,B-二芳基四配位硼络合物的合成[J]. 有机化学, 2023 , 43(5) : 1792 -1798 . DOI: 10.6023/cjoc202301001
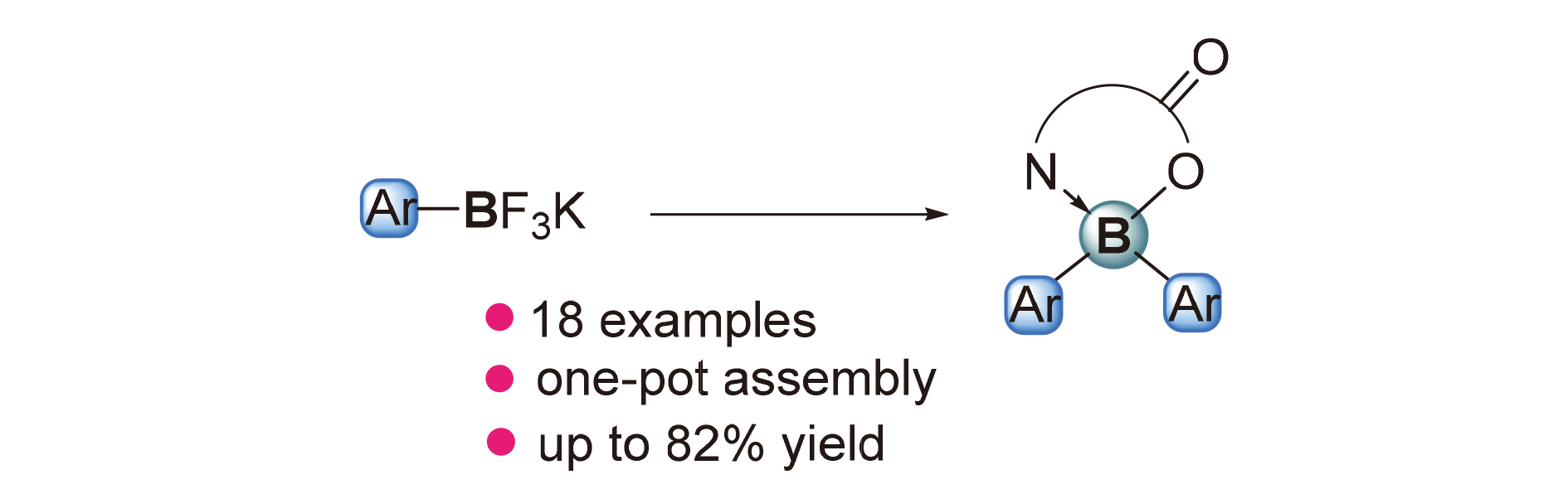
A method for the preparation of N,O-chelated B,B-diaryl tetracoordinated organoboron complexes is disclosed. Using stable and readily available potassium aryltrifluoroborates (ArBF3K) as the surrogate of the diarylboron (Ar2B) moieties and isoquinoline-3-carboxylic acid as the precursor of the N,O-chelating ligand, the isoquinoline-3-carboxylate chelated diarylboron complexes can be obtained in the presence of Mn, p-toluenesulfonyl chloride, and base. This reaction is featured by broad substrate scope and functional group compatibility, thus providing a convenient and efficient pathway towards diarylboron complexes.
| [1] | Vidyasagar, C. C.; Mu?oz Flores, B. M.; Jiménez-Pérez, V. M.; Gurubasavaraj, P. M. Mater. Today Chem. 2019, 11, 133. |
| [2] | Li, D.; Zhang, H.; Wang, Y. Chem. Soc. Rev. 2013, 42, 8416. |
| [3] | Huang, Z.; Wang, S.; Dewhurst, R. D.; Ignat'ev, N. V.; Finze, M.; Braunschweig, H. Angew. Chem. Int. Ed. 2020, 59, 8800. |
| [4] | Mellerup, S. K.; Wang, S. Chem. Soc. Rev. 2019, 48, 3537. |
| [5] | Loudet, A.; Burgess, K. Chem. Rev. 2007, 107, 4891. |
| [6] | Yang, T.; Tang, N.; Wan, Q.; Yin, S.-F.; Qiu, R. Molecules 2021, 26, 1401. |
| [7] | Guan, C.; Huang, L.; Ren, C.; Zou, G. Org. Process Res. Dev. 2018, 22, 824. |
| [8] | Marciasini, L.; Cacciuttolo, B.; Vaultier, M.; Pucheault, M. Org. Lett. 2015, 17, 3532. |
| [9] | Pais, V. F.; Ramírez-López, P.; Romero-Arenas, A.; Collado, D.; Nájera, F.; Pérez-Inestrosa, E.; Fernández, R.; Lassaletta, J. M.; Ros, A.; Pischel, U. J. Org. Chem. 2016, 81, 9605. |
| [10] | Más-Montoya, M.; Usea, L.; Espinosa Ferao, A.; Montenegro, M. F.; Ramírez de Arellano, C.; Tárraga, A.; Rodríguez-López, J. N.; Curiel, D. J. Org. Chem. 2016, 81, 3296. |
| [11] | Fu, Y.; Qiu, F.; Zhang, F.; Mai, Y.; Wang, Y.; Fu, S.; Tang, R.; Zhuang, X.; Feng, X. Chem. Commun. 2015, 51, 5298. |
| [12] | Yang, K.; Zhang, G.; Song, Q. Chem. Sci. 2018, 9, 7666. |
| [13] | Wang, Z.; Cheng, C.; Kang, Z.; Miao, W.; Liu, Q.; Wang, H.; Hao, E. J. Org. Chem. 2019, 84, 2732. |
| [14] | Sawazaki, T.; Shimizu, Y.; Oisaki, K.; Sohma, Y.; Kanai, M. Org. Lett. 2018, 20, 7767. |
| [15] | Zu, W.; Day, C.; Wei, L.; Jia, X.; Xu, L. Chem. Commun. 2020, 56, 8273. |
| [16] | Ding, S.; Zu, W.; Miao, Z.; Xu, L. Chin. J. Org. Chem. 2022, 42, 812. (in Chinese) |
| [16] | (丁思懿, 祖维赛, 苗宗成, 徐亮, 有机化学, 2022, 42, 812.) |
| [17] | You, G.; Xu, L.; Wei, Y. Front. Chem. 2022, 10, DOI: 10.3389/fchem.2022.856832. |
| [18] | Wei, L.; Zhang, J.; Xu, L. ACS Sustain. Chem. Eng. 2020, 8, 13894. |
| [19] | Wei, L.; Wei, Y.; Zhang, J.; Xu, L. Green Chem. 2021, 23, 4446. |
/
| 〈 |
|
〉 |