

伯醇的脱羟甲基反应的研究进展
收稿日期: 2023-03-04
修回日期: 2023-04-04
网络出版日期: 2023-04-14
基金资助
国家自然科学基金(22071185); 国家自然科学基金(22271224); 中央高校基本科研业务费专项资金(2042019kf0008); 武汉大学启动基金资助项目
Progress in the Study of Dehydroxymethylation of Primary Alcohol
Received date: 2023-03-04
Revised date: 2023-04-04
Online published: 2023-04-14
Supported by
The National Natural Science Foundation of China(22071185); The National Natural Science Foundation of China(22271224); The Fundamental Research Funds for the Central Universities(2042019kf0008); The Wuhan University Startup Funding
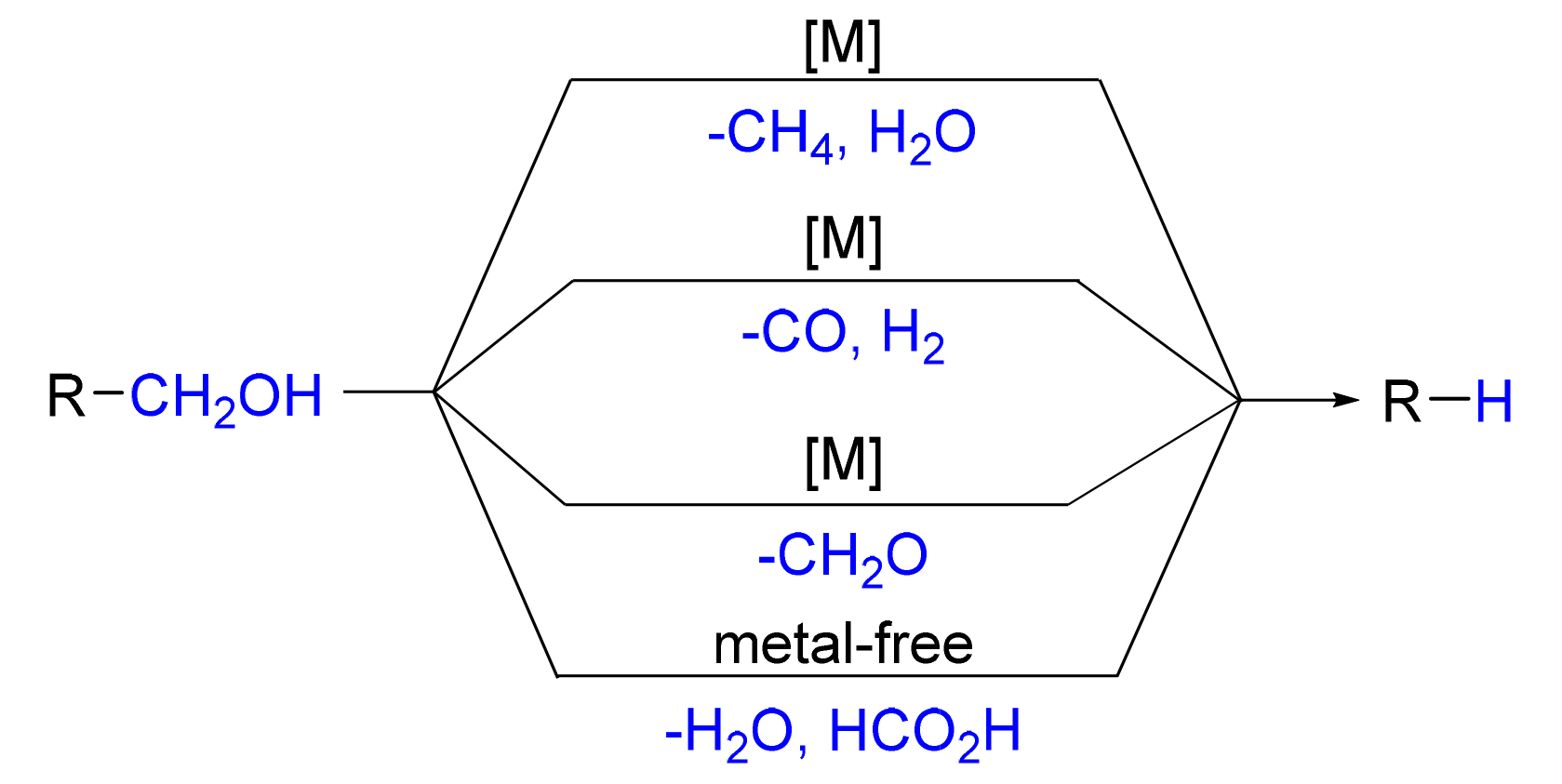
伯醇来源广泛且廉价易得, 是生产各种化学品, 如农用化学品、食品添加剂、香料和药物等的重要化工原料. 伯醇的脱羟甲基反应是一种利用伯醇制备少一个碳原子的碳氢化合物的方法, 在生物质降解和药物合成等方面有着广泛的应用, 引起了有机化学家的关注. 根据反应类型综述了伯醇的脱羟甲基反应的发展, 根据反应特点分为四个部分综述: 过渡金属催化脱羟甲基转化为甲烷和水的反应、过渡金属催化脱羟甲基转化为一氧化碳和氢气的反应、过渡金属催化脱羟甲基转化为甲醛的反应、无过渡金属脱羟甲基转化为甲酸和水的反应. 主要从反应体系、适用范围、反应机理等方面阐述了伯醇的脱羟甲基反应的研究进展.
陈新强 , 张敬 . 伯醇的脱羟甲基反应的研究进展[J]. 有机化学, 2023 , 43(8) : 2711 -2719 . DOI: 10.6023/cjoc202303009
Primary alcohols are abundant and readily available, which have been widely used as feedstock chemicals for the synthesis of agrochemicals, food additives, fragrances, pharmaceuticals and so on. The dehydroxymethylation reaction is an effective strategy to prepare hydrocarbons with one less carbon atom from primary alcohols, which has attracted tremendous attentions of many organic chemists and applied in biomass degradation and drug synthesis. The development of dehydroxy- methylation of primary alcohol on the base of the reaction characters is reviewed, including transition-metal catalyzed dehydroxymethylation to access methane and water, transition-metal catalyzed dehydroxymethylation to produce carbon monoxide and hydrogen, transition-metal catalyzed dehydroxymethylation to generate formaldehyde, as well as transition-metal free dehydroxymethylation to furnish formic acid and water. Each type of reaction is systematically summarized according to the catalytic system, the substrates scope and reaction mechanism.

| [1] | Lutz, M. D. R.; Morandi, B. Chem. Rev. 2021, 121, 300. |
| [2] | Dong, X.-J.; Jin, W.-W.; Liu, C.-J. Chin. J. Org. Chem. 2020, 40, 1860. (in Chinese) |
| [2] | ( 董晓娟, 金伟伟, 刘晨江, 有机化学, 2020, 40, 1860.) |
| [3] | Contente, M. L.; Paradisi, F. Nat. Catal. 2018, 1, 452. |
| [4] | Ahn, Y.; Ko, S.-B.; Kim, M.-J.; Park, J. Coord. Chem. Rev. 2008, 252, 647. |
| [5] | Modak, A.; Maiti, D. Org. Biomol. Chem. 2016, 14, 21. |
| [6] | Zeng, H.; Yu, J.; Li, C. J. Chem. Commun. 2020, 56, 1239. |
| [7] | Zhang, S.-X.; Li, X.-R.; Li, W.-X.; Rao, W.-D.; Ge, D.-H.; Shen, Z.-L.; Chu, X.-Q. Chin. J. Org. Chem. 2022, 42, 235. (in Chinese) |
| [7] | ( 张斯旋, 李祥瑞, 李文欣, 饶卫东, 葛丹华, 沈志良, 褚雪强, 有机化学, 2022, 42, 235.) |
| [8] | Xue, X.; Weng, Y.; Yang, S.; Meng, S.; Sun, Q.; Zhang, Y. RSC Adv. 2021, 11, 6163. |
| [9] | Surisetty, V. R.; Dalai, A. K.; Kozinski, J. Appl. Catal., A 2011, 404, 1. |
| [10] | Norjannah, B.; Ong, H. C.; Masjuki, H. H.; Juan, J. C.; Chong, W. T. RSC Adv. 2016, 6, 60034. |
| [11] | Chen, B.-S.; Ribeiro de Souza, F. Z. RSC Adv. 2019, 9, 2102. |
| [12] | Tian, Z.-W.; Da, W.-M.; Wang, L.; Yang, Y.-S.; Wei, M. Acta Chim. Sinica 2022, 80, 1322. (in Chinese) |
| [12] | ( 田钊炜, 达伟民, 王雷, 杨宇森, 卫敏, 化学学报, 2022, 80, 1322.) |
| [13] | Liu, H.; Dong, C.; Zhang, Z.; Wu, P.; Jiang, X. Angew. Chem., Int. Ed. 2012, 51, 12570. |
| [14] | Christensen, S. H.; Olsen, E. P.; Rosenbaum, J.; Madsen, R. Org. Biomol. Chem. 2015, 13, 938. |
| [15] | Kumar, A.; Shah, B. A. Org. Lett. 2015, 17, 5232. |
| [16] | Park, H.-S.; Kim, D.-S.; Jun, C.-H. ACS Catal. 2015, 5, 397. |
| [17] | Huang, G.; Lu, L.; Jiang, H.; Yin, B. Chem. Commun. 2017, 53, 12217. |
| [18] | Wu, X.; Cruz, F. A.; Lu, A.; Dong, V. M. J. Am. Chem. Soc. 2018, 140, 10126. |
| [19] | Hu, X.; Li, G.-X.; He, G.; Chen, G. Org. Chem. Front. 2019, 6, 3205. |
| [20] | Chen, Y.; Wang, X.; He, X.; An, Q.; Zuo, Z.-W J. Am. Chem. Soc. 2021, 143, 4896. |
| [21] | Gao, Y.; Liu, J.; Wei, C.; Li, Y.; Zhang, K.; Song, L.; Cai, L. Nat. Commun. 2022, 13, 7450. |
| [22] | Mahajan, B.; Aand, D.; Singh, A. K. ChemistrySelect 2018, 3, 12336. |
| [23] | Yu, X.; Wang, J.; Guo, W.; Tian, Y.; Wang, J. Organometallics 2016, 35, 1876. |
| [24] | Zhou, X.; Ding, H.; Chen, P.; Liu, L.; Sun, Q.; Wang, X.; Wang, P.; Lv, Z.; Li, M. Angew. Chem., Int. Ed. 2020, 59, 4138. |
| [25] | Liu, M.; Zhang, Z.; Liu, H.; Wu, T.; Han, B. Chem. Commun. 2020, 56, 7120. |
| [26] | Chheda, J. N.; Huber, G. W.; Dumesic, J. A. Angew. Chem., Int. Ed. 2007, 46, 7164. |
| [27] | Gallezot, P. ChemSusChem 2008, 1, 734. |
| [28] | Marshall, A. L.; Alaimo, P. J. Chem.-Eur. J. 2010, 16, 4970. |
| [29] | Zakzeski, J.; Bruijnincx, P. C.; Jongerius, A. A. L.; Weckhuysen, B. M. Chem. Rev. 2010, 110, 3552. |
| [30] | Serrano-Ruiz, J. C.; Dumesic, J. A. Energy Environ. Sci. 2011, 4, 83. |
| [31] | Li, H.; Wu, H.; Yu, Z.; Zhang, H.; Yang, S. ChemSusChem 2020, 13, 3565. |
| [32] | Agarwal, S.; Al-Abed, S. R.; Dionysiou, D. D. Environ. Sci. Technol. 2007, 41, 3722. |
| [33] | Monrad, R. N.; Madsen, R. J. Org. Chem. 2007, 72, 9782. |
| [34] | Agarwal, S.; Al-Abed, S. R.; Dionysiou, D. D. Environ. Sci. Technol. 2009, 43, 915. |
| [35] | Yamaguchi, S.; Kondo, H.; Uesugi, K.; Sakoda, K.; Jitsukawa, K.; Mitsudome, T.; Mizugaki, T. ChemCatChem 2021, 13, 1135. |
| [36] | Zhang, Z.; Zijlstra, D. S.; Lahive, C. W.; Deuss, P. J. Green Chem. 2020, 22, 3791. |
| [37] | Kreis, M.; Palmelund, A.; Bunch, L.; Madsen, R. Adv. Synth. Catal. 2006, 348, 2148. |
| [38] | Fessard, T. C.; Andrews, S. P.; Motoyoshi, H.; Carreira, E. M. Angew. Chem., Int. Ed. 2007, 46, 9331. |
| [39] | Iwai, T.; Fujihara, T.; Tsuji, Y. Chem. Commun. 2008, 6215. |
| [40] | Br?se, S.; Br?hmer, M.; Volz, N. Synlett 2009, 1383. |
| [41] | Akanksha; Maiti, D. Green Chem. 2012, 14, 2314. |
| [42] | Modak, A.; Deb, A.; Patra, T.; Rana, S.; Maity, S.; Maiti, D. Chem. Commun. 2012, 48, 46. |
| [43] | Roa, A. E.; Salazar, V.; López-Serrano, J.; O?ate, E.; Paneque, M.; Poveda, M. L. Organometallics 2012, 31, 716. |
| [44] | Huang, Y. B.; Yang, Z.; Chen, M. Y.; Dai, J. J.; Guo, Q. X.; Fu, Y. ChemSusChem 2013, 6, 1348. |
| [45] | Gutmann, B.; Elsner, P.; Glasnov, T.; Roberge, D. M.; Kappe, C. O. Angew. Chem., Int. Ed. 2014, 53, 11557. |
| [46] | Mitra, J.; Zhou, X.; Rauchfuss, T. Green Chem. 2015, 17, 307. |
| [47] | Shokri, A.; Que, L., Jr. J. Am. Chem. Soc. 2015, 137, 7686. |
| [48] | Hattori, T.; Takakura, R.; Ichikawa, T.; Sawama, Y.; Monguchi, Y.; Sajiki, H. J. Org. Chem. 2016, 81, 2737. |
| [49] | Ding, K.; Xu, S.; Alotaibi, R.; Paudel, K.; Reinheimer, E. W.; Weatherly, J. J. Org. Chem. 2017, 82, 4924. |
| [50] | Chatterjee, M.; Ishizaka, T.; Kawanami, H. Green Chem. 2018, 20, 2345. |
| [51] | Li, W. H.; Li, C. Y.; Li, Y.; Tang, H. T.; Wang, H. S.; Pan, Y. M.; Ding, Y. J. Chem. Commun. 2018, 54, 8446. |
| [52] | Matsuyama, T.; Yatabe, T.; Yabe, T.; Yamaguchi, K. ACS Catal. 2021, 11, 13745. |
| [53] | Lu, H.; Yu, T. Y.; Xu, P. F.; Wei, H. Chem. Rev. 2021, 121, 365. |
| [54] | B?eseken, J.; van Senden, G. H. Recl. Trav. Chim. Pays-Bas Belg. 1913, 32, 23. |
| [55] | Wojcik, B.; Adkins, H. J. Am. Chem. Soc. 1933, 55, 1293. |
| [56] | Adkins, H. Ind. Eng. Chem. Anal. Ed. 1932, 4, 342. |
| [57] | Covert, L. W.; Adkins, H. J. Am. Chem. Soc. 1932, 54, 4116. |
| [58] | Ipatieff, V. N.; Monroe, G. S.; Fischer, L. E.; Meisinger, E. E. Ind. Eng. Chem. 1949, 41, 1802. |
| [59] | Ipatieff, V. N.; Thompson, W. W.; Pines, H. J. Am. Chem. Soc. 1951, 73, 553. |
| [60] | Ipatieff, V. N.; Czajkowski, G. J.; Pines, H. J. Am. Chem. Soc. 1951, 73, 4098. |
| [61] | Pines, H.; Rodenberg, H. G.; Ipatieff, V. N. J. Am. Chem. Soc. 1953, 75, 6065. |
| [62] | Pines, H.; Rodenberg, H. G.; Ipatieff, V. N. J. Am. Chem. Soc. 1954, 76, 771. |
| [63] | Pines, H.; Shamaiengar, M.; Postl, W. S. J. Am. Chem. Soc. 1955, 77, 5099. |
| [64] | Reinecke, M. G.; Grins, G.; Kray, L. R.; Francis, R. F. Ann. N. Y. Acad. Sci. 1967, 145, 116. |
| [65] | Masayoshi, I.; Koji, S.; Masatoshi, K.; Kazuo, H. Bull. Chem. Soc. Jpn. 1970, 43, 2186. |
| [66] | Maier, W. F.; Grubmüller, P.; Thies, I.; Stein, P. M.; McKervey, M. A.; Schleyer, P. V. R. Angew. Chem., Int. Ed. Engl. 1979, 18, 939. |
| [67] | Di, L.; Yao, S.; Li, M.; Wu, G.; Dai, W.; Wang, G.; Li, L.; Guan, N. ACS Catal. 2015, 5, 7199. |
| [68] | Mizugaki, T.; Togo, K.; Maeno, Z.; Mitsudome, T.; Jitsukawa, K.; Kaneda, K. Sci. Rep. 2017, 7, 14007. |
| [69] | Kim, C.; Matsui, Y.; Orchin, M. J. Organomet. Chem. 1985, 279, 159. |
| [70] | Olsen, E. P.; Madsen, R. Chem.-Eur. J. 2012, 18, 16023. |
| [71] | Olsen, E. P.; Singh, T.; Harris, P.; Andersson, P. G.; Madsen, R. J. Am. Chem. Soc. 2015, 137, 834. |
| [72] | Ho, H. A.; Manna, K.; Sadow, A. D. Angew. Chem., Int. Ed. 2012, 51, 8607. |
| [73] | Modak, A.; Naveen, T.; Maiti, D. Chem. Commun. 2013, 49, 252. |
| [74] | Mazziotta, A.; Madsen, R. Eur. J. Org. Chem. 2017, 2017, 5417. |
| [75] | Pedersen, M. J.; Madsen, R.; Clausen, M. H. Chem. Commun. 2018, 54, 952. |
| [76] | Peng, L.; Ma, M.; Zhang, X.; Zhang, S.; Wang, J. Tetrahedron Lett. 2006, 47, 8175. |
| [77] | Zhang, K.; Chang, L.; An, Q.; Wang, X.; Zuo, Z.-W. J. Am. Chem. Soc. 2019, 141, 10556. |
| [78] | Xia, A.; Qi, X.; Mao, X.; Wu, X.; Yang, X.; Zhang, R.; Xiang, Z.; Lian, Z.; Chen, Y.; Yang, S. Org. Lett. 2019, 21, 3028. |
/
| 〈 |
|
〉 |