

电化学介导羧酸与醇的酯化反应
收稿日期: 2023-02-05
修回日期: 2023-03-24
网络出版日期: 2023-04-23
基金资助
国家自然科学基金(22071082)
Electrochemical Mediated Esterification Reaction of Carboxylic Acids and Alcohols
Received date: 2023-02-05
Revised date: 2023-03-24
Online published: 2023-04-23
Supported by
The National Natural Science Foundation of China(22071082)
钟赟哲 , 陈颖 , 俞磊 , 周宏伟 . 电化学介导羧酸与醇的酯化反应[J]. 有机化学, 2023 , 43(8) : 2855 -2863 . DOI: 10.6023/cjoc202302004
A series of esters were prepared by electrochemical synthesis of carboxylic acids with alcohols. This protocol can be carried out at room temperature without inert atmosphere, strong acids or metal catalysts, etc. The esterification reaction processes in an undivided electrolytic cell with constant current mode and the esterification products are generated in good yields. Compared with esterification promoted by organic reagents, this protocol avoids the byproducts which have to be separated by column chromatography and successful for tertiary alcohol which is generally difficult to undergo esterification directly, providing a new access to the esterification of carboxylic acids and alcohols.
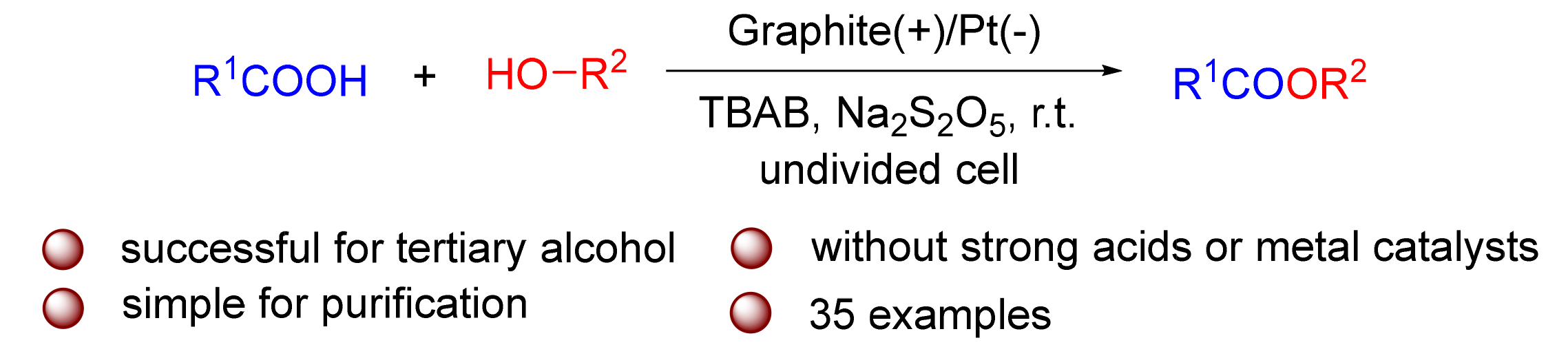
| [1] | (a) Larock, R. C. Comprehensive Organic Transformations, Wiley, New York, 2018, pp. 1-81. |
| [1] | (b) Khan, Z.; Javed, F.; Shamair, Z.; Hafeez, A.; Fazal, T.; Aslam, A.; Zimmerman, W. B.; Rehman, F. J. Ind. Eng. Chem. 2021, 103, 80. |
| [1] | (c) Zheng, Y.; Zhao, Y.; Tao, S.; Li, X.; Cheng, X.; Jiang, G.; Wan, X. Eur. J. Org. Chem. 2021, 18, 2713. |
| [1] | (d) Li, Y.; Wang, H.; Zhang, H.; Lei, A. Chin. J. Chem. 2021, 39, 3023. |
| [1] | (e) Maeda, B.; Sakakibara, Y.; Murakami, K.; Itami, K. Org. Lett. 2021, 23, 5113. |
| [2] | (a) Zare, M.; Golmakani, M.-T.; Sardarian, A. Green Chem. Lett. Rev. 2020, 13, 83. |
| [2] | (b) De Barros, D. P.; Azevedo, A. M.; Cabral, J. M.; Fonseca, L. P. Food Biochem. 2012, 36, 275. |
| [2] | (c) Sá, A. G.; De Meneses, A. C.; De Araújo, P. H.; Oliveira, D. Trends Food Sci. Technol. 2017, 69, 95. |
| [2] | (d) Khan, N. R.; Rathod, V. K. Process Biochem. 2015, 50, 1793. |
| [2] | (e) Zhu, Z.; Li, S.; Liu, R.; Yuan, J.; Wang, H.; Zhang, Y.; Liu, Y. Chin. J. Chem. 2010, 28, 2245. |
| [3] | (a) Yang, M.; Wang, X.; Zhao, J. ACS Catal. 2020, 10, 5230. |
| [3] | (b) Li, Q.-C.; Jiang, L.; Bai, R.; Han, Y.-K.; Li, Z.-N. Chin. J. Org. Chem. 2021, 41, 3390. (in Chinese) |
| [3] | ( 李泉城, 姜岚, 白瑞, 韩永康, 李争宁, 有机化学, 2021, 41, 3390.) |
| [3] | (c) Bińczak, J.; Dziuba, K.; Chrobok, A. Materials 2021, 14, 2881. |
| [3] | (d) Zhang, Q.; Li, Y.-H.; Xu, L.-C.; Ma, H.-Y.; Li, X.-D.; Wang, M.-A. Chin. J. Org. Chem. 2022, 42, 2438. (in Chinese) |
| [3] | ( 张倩, 李益豪, 许磊川, 马好运, 李向东, 王明安, 有机化学, 2022, 42, 2438.) |
| [4] | (a) Iwasaki, T. Kirk-Othmer Encyclopedia of Chemical Technology, Wiley, New York, 2000, pp. 1-33. |
| [4] | (b) Steele, J. H.; Bozor, M. X.; Boyce, G. R. J. Chem. Educ. 2020, 97, 4127. |
| [4] | (c) Jyoti, G.; Keshav, A.; Anandkumar, J. Int. J. Chem. React. Eng. 2016, 14, 571. |
| [4] | (d) Li, Y.-L.; Pang, J.-Y.; Lou, J.-C.; Sun, W.-W.; Liu, J.-K.; Wu, B. Asian J. Org. Chem. 2021, 10, 1. |
| [5] | (a) Zhu, L.; Zhu, Y.; Meng, X.; Hao, J.; Li, Q.; Wei, Y.; Lin, Y. Chem.-Eur. J. 2008, 14, 10923. |
| [5] | (b) Neises, B.; Steglich, W. Angew. Chem. 1978, 90, 556. |
| [5] | (c) Zhang, J.; Wang, Y.; Du, G.; Gu, C.-Z.; Dai, B. Chin. J. Chem. 2015, 33, 1211. |
| [6] | (a) Jyoti, G.; Keshav, A.; Anandkumar, J.; Bhoi, S. Int. J. Chem. Kinet. 2018, 50, 370. |
| [6] | (b) Sert, E.; Atalay, F. S. Ind. Eng. Chem. Res. 2012, 51, 6666. |
| [6] | (c) Buluklu, A. D.; Sert, E.; Karaku?, S.; Atalay, F. S. Int. J. Chem. Kinet. 2014, 46, 197. |
| [6] | (d) Chen, X.; Mao, J.; Liu, C.; Chen, C.; Cao, H.; Yu, L. Chin. Chem. Lett. 2020, 31, 3205. |
| [6] | (e) Zhang, L.; Zhang, H.; Shang, H. Can. J. Chem. Eng. 2021, 99, 1107. |
| [6] | (f) Xiao, X.; Shao, Z.; Yu, L. Chin. Chem. Lett. 2021, 32, 2933. |
| [6] | (g) Li, W.; Wang, F.; Shi, Y.; Yu, L. Chin. Chem. Lett. 2023, 34, 107505. |
| [6] | (h) Zeng, Z.; Chen, Y.; Zhu, X.; Yu, L. Chin. Chem. Lett. 2023, 34, 107728. |
| [7] | (a) Pongpamorn, P.; Kiattisewee, C.; Kittipanukul, N.; Jaroensuk, J.; Trisrivirat, D.; Maenpuen, S.; Chaiyen, P. Angew. Chem., Int. Ed. 2021, 60, 5749. |
| [7] | (b) Li, N.-G.; Shi, Z.-H.; Tang, Y.-P.; Li, B.-Q.; Duan, J.-A. Molecules 2009, 14, 2118. |
| [7] | (c) Maester, T. C.; Pereira, M. R.; Malaman, A. M.; Borges, J. P.; Pereira, P. A.; Lemos, E. G. Catalysts 2020, 10, 1100. |
| [7] | (d) Shi, H.; Zhu, W.; Li, H.; Liu, H.; Zhang, M. ; Yan, Y.; Wang, Z. Catal. Commun. 2010, 11, 588. |
| [7] | (e) Nguyen, H. C.; Wang, F.-M.; Dinh, K. K.; Pham, T. T.; Juan, H.-Y.; Nguyen, N. P.; Ong, H. C.; Su, C.-H. Energies 2020, 13, 2167. |
| [8] | (a) Stergiou, P.-Y.; Foukis, A.; Filippou, M.; Koukouritaki, M.; Parapouli, M.; Theodorou, L. G.; Hatziloukas, E.; Afendra, A.; Pandey, A.; Papamichael, E. M. Biotechnol. Adv. 2013, 31, 1846. |
| [8] | (b) Guldhe, A.; Singh, B.; Mutanda, T.; Permaul, K.; Bux, F. Renewable Sustainable Energy Rev. 2015, 41, 1447. |
| [8] | (c) Moseley, J. D.; Kappe, C. O. Green Chem. 2011, 13, 794. |
| [8] | (d) Li, X.-Y.; Tao, P.-F.; Cheng, Y.-Y.; Hu, Q.; Huang, W.-J.; Li, Y.; Luo, Z.-H.; Huang, G.-B. Chin. J. Org. Chem. 2022, 42, 4169. (in Chinese) |
| [8] | ( 李秀英, 陶萍芳, 程泳渝, 胡琼, 黄伟娟, 李芸, 罗志辉, 黄国保, 有机化学, 2022, 42, 4169.) |
| [9] | (a) Sperry, J. B.; Wright, D. L. Chem. Soc. Rev. 2006, 35, 605. |
| [9] | (b) Yoshida, J.-I.; Kataoka, K.; Horcajada, R.; Nagaki, A. Chem. Rev. 2008, 108, 2265. |
| [9] | (c) Frontana-Uribe, B. A.; Little, R. D.; Ibanez, J. G.; Palma, A.; Vasquez-Medrano, R. Green Chem. 2010, 12, 2099. |
| [9] | (d) Horn, E. J.; Rosen, B. R.; Baran, P. S. ACS Cent. Sci. 2016, 2, 302. |
| [9] | (e) Wiebe, A.; Gieshoff, T.; Mohle, S.; Rodrigo, E.; Zirbes, M.; Waldvogel, S. R. Angew. Chem., Int. Ed. 2018, 57, 5594. |
| [9] | (f) Wen, L. R.; Wang, N. N.; Du, W. B.; Zhu, M. Z.; Pan, C.; Zhang, L. B.; Li, M. Chin. J. Chem. 2021, 39, 1831. |
| [10] | (a) Little, R. D.; Moeller, K. D. Chem. Rev. 2018, 118, 4483. |
| [10] | (b) Minteer, S. D.; Baran, P. Acc. Chem. Res. 2020, 53, 545. |
| [10] | (c) Yuan, Y.; Lei, A. Nat. Commun. 2020, 11, 802. |
| [10] | (d) Mohle, S.; Zirbes, M; Rodrigo, E.; Gieshoff, T.; Wiebe, A.; Waldvogel, S. R. Angew. Chem., Int. Ed. 2018, 57, 6018. |
| [10] | (e) Wen, L. R.; Wang, N. N.; Du, W. B.; Zhu, M. Z.; Pan, C.; Zhang, L. B.; Li, M. Chin. J. Chem. 2021, 39, 1831. |
| [11] | (a) Waldvogel, S. R.; Janza, B. Angew. Chem., Int. Ed. 2014, 53, 7122. |
| [11] | (b) Yan, M.; Kawamata, Y.; Baran, P. S. Chem. Rev. 2017, 117, 13230. |
| [11] | (c) Schotten, C.; Nicholls, T. P.; Bourne, R. A.; Kapur, N.; Nguyen, B. A.; Willans, C. E. Green Chem. 2020, 22, 3358. |
| [11] | (d) Wang, P.; Tang, S.; Huang, P.; Lei, A. Angew. Chem., Int. Ed. 2017, 56, 3009. |
| [11] | (e) Zhang, L.; Cheng, X.; Zhou, Q. L. Chin. J. Chem. 2022, 40, 1687. |
| [12] | Kaboudin, B.; Behrouzi, L.; Kazemi, F.; Najafpour, M. M.; Aoyama, H. ACS Omega 2020, 5, 17947. |
| [13] | Cheng, S.-Y.; Ou, C.-H.; Lin, H.-M.; Jia, J. S.; Tang, H.-T.; Pan, Y.-M.; Huang, G.-B.; Meng, X.-J. Chin. J. Org. Chem. 2021, 41, 4718. (in Chinese) |
| [13] | ( 程诗砚, 欧楚鸿, 林洪敏, 贾均松, 唐海涛, 潘英明, 黄国保, 蒙秀金, 有机化学, 2021, 41, 4718.) |
| [14] | (a) Devadas, B.; Svoboda, J.; Krupicka, M. Electrochim. Acta 2020, 342, 136080. |
| [14] | (b) Gil, D. M. D.; Rebelo, M. J. F. Microchim. Acta 2009, 167, 253. |
| [14] | (c) Zhang, Z.; Wang, Y.; Tang, H.; Pan, Y.; Meng, X. Org. Biomol. Chem. 2023, 21, 3177. |
| [14] | (d) Villoria-del-álamo, B.; Rojas-Buzo, S.; García-García, B.; Corma, A. Chem.-Eur. J. 2021, 27, 4588. |
| [14] | (e) Lawal, M. M.; Govender, T.; Maguire, G. E.; Kruger, H. G.; Honarparvar, B. Int. J. Quantum Chem. 2018, 118, e25497. |
| [15] | (a) Jagadeesh, R. V.; Junge, H.; Pohl, M. M.; Radnik, J.; Bruckner, A.; Beller, M. J. Am. Chem. Soc. 2013, 135, 10776. |
| [15] | (b) Trost, B. M.; McClory, A. Angew. Chem., Int. Ed. 2007, 46, 2074. |
| [16] | Sun, X. Y.; Shan, G.; Sun, Y. H.; Rao, Y. Angew. Chem., Int. Ed. 2013, 52, 4440. |
| [17] | (a) Liang, A. P.; Han, S. J.; Wang, L.; Li, J. Y.; Zou, D. P.; Wu, Y. J.; Wu, Y. S. Adv. Synth. Catal. 2015, 357, 3104. |
| [17] | (b) Wang, X.-J.; Zhang, S.-L. New J. Chem. 2017, 41, 14826. |
| [18] | Garcia-Alvarez, P.; Graham, D. V.; Hevia, E.; Kennedy, A. R.; Klett, J.; Mulvey, R. E.; O'Hara, C. T.; Weatherstone, S. Angew. Chem., Int. Ed. 2008, 47, 8079. |
| [19] | Karmakar, A.; da Silva, M.; Pombeiro, A. J. L. Dalton Trans. 2014, 43, 7795. |
| [20] | (a) Salman, B. S.; Ahmed, M. R. Chem. Methodol. 2022, 6, 997. |
| [20] | (b) Day, C.; Jia, X.; Wei, L.; Xu, L.; Zu, W. Chem. Commun. 2020, 56, 8273. |
| [21] | Tian, H. T.; Xue, W. X.; Wu, J. T.; Yang, Z. G.; Lu, H. C.; Tang, C. H. Org. Chem. Front. 2022, 9, 4554. |
| [22] | (a) Tollini, F.; Brivio, L.; Innocenti, P.; Sponchioni, M.; Moscatelli, D. Chem. Eng. Sci. 2022, 260, 117875. |
| [22] | (b) Zhou, Y.; Yang, D.; Luo, G.; Zhao, Y.; Luo, Y.; Xue, N.; Qu, J. Tetrahedron 2014, 70, 4668. |
| [23] | Karthik, S.; Sreedharan, R.; Gandhi, T. ChemistrySelect 2019, 4, 175. |
| [24] | Gan, S. Y.; Yin, J. R.; Yu, Z. Y.; Song, L. J.; Shi, L. Green Chem. 2022, 24, 2232. |
| [25] | (a) Sarjeant, A. A.; Snurr, R. Q.; Stoddart, J. F.; Hupp, J. T.; Farha, O. K. J. Am. Chem. Soc. 2015, 137, 3585. |
| [25] | (b) Reed, M. A.; Chang, M. T.; Snieckus, V. Org. Lett. 2004, 6, 2297. |
| [26] | Luo, H. H.; Yang, Y. K.; Yang, B. B.; Xu, Z. J.; Wang, D. W. J. Chem. Res. 2021, 45, 708. |
| [27] | Tabasi, N. S.; Genc, S.; Gulcemal, D. Org. Biomol. Chem. 2022, 20, 6582. |
| [28] | Azadi, S.; Goudarzian, N.; Parish, M. H.; Hosseini, F. N. Monatsh. Chem. 2023, 154, 239. |
| [29] | Chakraborti, A. K.; Singh, B.; Chankeshwara, S. V.; Patel, A. R. J. Org. Chem. 2009, 74, 5967. |
| [30] | Miah, M. A. J.; Sibi, M. P.; Chattopadhyay, S.; Familoni, O. B.; Snieckus, V. Eur. J. Org. Chem. 2018, 2018, 440. |
| [31] | Bhatia, A.; Kannan, M.; Muthaiah, S. Synlett 2019, 30, 721. |
/
| 〈 |
|
〉 |