

钴催化烯胺远程硼氢化
收稿日期: 2023-02-25
修回日期: 2023-05-14
网络出版日期: 2023-05-15
基金资助
国家自然科学基金(22271086); 国家自然科学基金(21971059)
Cobalt-Catalyzed Remote Hydroboration of Enamines
Received date: 2023-02-25
Revised date: 2023-05-14
Online published: 2023-05-15
Supported by
National Natural Science Foundation of China(22271086); National Natural Science Foundation of China(21971059)
刘育园 , 雷雅钦 , 杨文 , 赵万祥 . 钴催化烯胺远程硼氢化[J]. 有机化学, 2023 , 43(5) : 1761 -1771 . DOI: 10.6023/cjoc202302027
A cobalt-catalyzed remote hydroboration of enamines via a sequence of chain walking and hydroboration in the presence of an earth abundant catalyst and a commercially available ligand is reported, providing a rapid access to borylamines. This protocol exhibites mild reaction conditions, broad substrate scope and good functional group compatibility. In addition, the practicability and the potential synthesis value of this reaction were demonstrated by a gram reaction and a variety of synthetic transformations of the borylamine products.
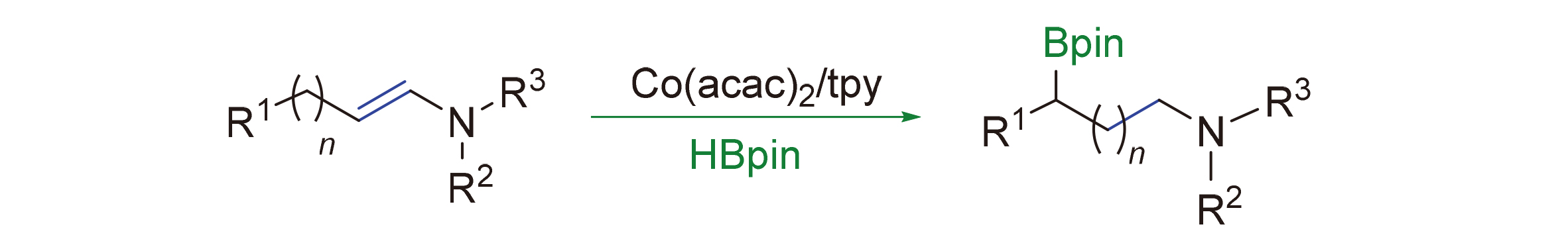
Key words: cobalt; enamines; remote hydroboration; bornic esters
| [1] | (a) Larionov, E.; Li, H.; Mazet, C. Chem. Commun. 2014, 50, 9816. |
| [1] | (b) Vasseur, A.; Bruffaerts, J.; Marek, I. Nat. Chem. 2016, 8, 209. |
| [1] | (c) Kochi, T.; Kanno, S.; Kakiuchi, F. Tetrahedron Lett. 2019, 60, 150938. |
| [1] | (d) Janssen-Mu?ller, D.; Sahoo, B.; Sun, S.-Z.; Martin, R. Isr. J. Chem. 2020, 60, 195. |
| [1] | (e) Wu, X.; Zhu, C. Acc. Chem. Res. 2020, 53, 1620. |
| [1] | (f) Wang, Y.; He, Y.; Zhu, S. Acc. Chem. Res. 2022, 55, 3519. |
| [1] | (g) Li, J.; Yu, B.; Lu, Z. Chin. J. Chem. 2021, 39, 488. |
| [2] | (a) Sommer, H.; Juliá-Hernández, F.; Martin, R.; Marek, I. ACS Cent. Sci. 2018, 4, 153. |
| [2] | (b) Dhunganà, R. K.; Sapkota, R. R.; Niroula, D.; Giri, R. Chem. Sci. 2020, 11, 9757. |
| [2] | (c) Yin, Y.; Liu, H.; Ouyang, D. Zhang, Q.; Zhu, R. Green Synth. Catal. 2023, 4, 64. |
| [3] | (a) Han, C.; Fu, Z.; Guo, S.; Fang, X.; Lin, A.; Yao, H. ACS Catal. 2019, 9, 4196. |
| [3] | (b) Zuo, Z.; Wang, J.; Liu, J.; Wang, Y.; Luan, X. Angew. Chem. Int. Ed. 2020, 59, 653. |
| [3] | (c) Baumgartner, Y.; Baudoin, O. ACS Catal. 2020, 10, 10508. |
| [4] | (a) He, Y.; Cai, Y.; Zhu, S. J. Am. Chem. Soc. 2017, 139, 1061. |
| [4] | (b) Zhou, L.; Zhu, C.; Bi, P.; Feng, C. Chem. Sci. 2019, 10, 1144. |
| [4] | (c) Lee, C.; Seo, H.; Jeon, J.; Hong, S. Nat. Commun. 2021, 12, 5657. |
| [5] | (a) Suresh, R.; Massad, I.; Marek, I. Chem. Sci. 2021, 12, 9328. |
| [5] | (b) Tang, K. H. N.; Uchida, K.; Nishihara, K.; Ito, M.; Shibata, T. Org. Lett. 2022, 24, 1313. |
| [5] | (c) Zou, X.; Xu, S. Chin. J. Org. Chem. 2021, 41, 2610. |
| [6] | (a) Burkhardt, E. R.; Matos, K. Chem. Rev. 2006, 106, 2617. |
| [6] | (b) Xu, S.; Lee, C. T.; Rao, H.; Negishia, E. I. Adv. Synth. Catal. 2011, 353, 2981. |
| [6] | (c) Lennox, A. J. J.; Lloyd-Jones, G. C. Chem. Soc. Rev. 2014, 43, 412. |
| [6] | (d) Neeve, E. C.; Geier, S. J.; Mkhalid, I. A. I.; Westcott, S. A.; Marder, T. B. Chem. Rev. 2016, 116, 9091. |
| [6] | (e) Ilies, L.; Itabashi, Y.; Shang, R.; Nakamura, E. ACS Catal. 2017, 7, 89. |
| [6] | (f) Sandford, C.; Aggarwal, V. K. Chem. Commun. 2017, 53, 5481. |
| [6] | (g) Cheng, Z.; Xing, S.; Guo, J.; Cheng, B.; Hu, L.; Zhang, X.; Lu, Z. Chin. J. Chem. 2019, 37, 457. |
| [6] | (h) Lu, H.; Li, B. Chin. J. Org. Chem. 2022, 42, 457. (in Chinese) |
| [6] | (陆候祥, 李必杰, 有机化学, 2022, 42, 457.) |
| [6] | (i) Lu, H.; Li, B. Chin. J. Org. Chem. 2022, 42, 3167. (in Chinese) |
| [6] | (陆候祥, 李必杰, 有机化学, 2022, 42, 3167.) |
| [7] | (a) Obligacion, J. V.; Chirik, P. J. J. Am. Chem. Soc. 2013, 135, 19107. |
| [7] | (b) Ruddy, A. J.; Sydora, O. L.; Small, B. L.; Stradiotto, M.; Turculet, L. Chem.-Eur. J. 2014, 20, 13918. |
| [7] | (c) Scheuermann, M. L.; Johnson, E. J.; Chirik, P. J. Org. Lett. 2015, 17, 2716. |
| [7] | (d) Palmer, W. N.; Obligacion, J. V.; Pappas, I.; Chirik, P. J. J. Am. Chem. Soc. 2016, 138, 766. |
| [7] | (e) Chen, X.; Cheng, Z.; Guo, J.; Lu, Z. Nat. Commun. 2018, 9, 3939. |
| [7] | (f) Hu, M.; Ge, S. Nat. Commun. 2020, 11, 765. |
| [8] | Lei, Y.; Huang, J.; Zhao, W. Org. Lett. 2021, 23, 7797. |
| [9] | (a) Michael, J. P. Nat. Prod. Rep. 2002, 19, 719. |
| [9] | (b) Saibabu Kotti, S. R.; Timmons, C.; Li, G. Chem. Biol. Drug. Des. 2006, 67, 101. |
| [9] | (c) Plechkova, N. V.; Seddon, K. R. Chem. Soc. Rev. 2008, 37, 123. |
| [9] | (d) McGrath, N. A.; Brichacek, M.; Njardarson, J. T. J. Chem. Educ. 2010, 87, 1348. |
| [9] | (e) Ruiz-Castillo, P.; Buchwald, S. L. Chem. Rev. 2016, 116, 12564. |
| [10] | Bélanger, G.; Doré, M.; Ménard, F.; Darsigny, V. J. Org. Chem. 2006, 71, 7481. |
| [11] | He, Y.; Song, H.; Chen, J.; Zhu, S. Nat. Commun. 2021, 12, 638. |
| [12] | Hosokawa, S.; Teramoto, K.; Motoyama, Y. ChemistrySelect 2016, 1, 2594. |
| [13] | Richa; Kumar, R.; Zhang, X.; Su, W. Org. Chem. Front. 2020, 7, 2965. |
| [14] | Fisher, G. B.; Lee, L.; Klettke, F. W. Synth. Commun. 1994, 24, 1541. |
| [15] | Fukumoto, Y.; Asai, H.; Shimizu, M.; Chatani, N. J. Am. Chem. Soc. 2007, 129, 13792. |
| [16] | Alcaide, B.; Almendros, P.; Alonso, J. M.; Aly, M. F. Org. Lett. 2001, 3, 3781. |
| [17] | Crotti, P.; Favero, L.; Macchia, F.; Pineschi, M. Tetrahedron Lett. 1994, 35, 7089. |
| [18] | Fu, R.; Liu, Y.; Wu, T.; Zhang, X.; Zhu, Y.; Luo, J.; Zhang, Z.; Jiang, Y. Chem. Commun. 2022, 58, 3525. |
| [19] | Ueno, S.; Usui, K.; Kuwano, R. Synlett 2011, 1303. |
| [20] | Pan, Y.; You, Y.; He, D.; Chen, F.; Chang, X.; Jin, M. Y.; Xing, X. Org. Lett. 2020, 22, 7278. |
| [21] | Bhadra, S.; Yamamoto, H. Angew. Chem. Int. Ed. 2016, 55, 13043. |
| [22] | Nakamura, Y.; Ohta, T.; Oe, Y. Chem. Commun. 2015, 51, 7459. |
| [23] | Das, K.; Sarkar, K.; Maji, B. ACS Catal. 2021, 11, 7060. |
| [24] | Wei, H. X.; Lu, D.; Sun, V.; Zhang, J.; Gu, Y.; Osenkowski, P.; Ye, W.; Selkoe, D. J.; Wolfe, M. S.; Augelli-Szafran, C. E. Bioorg. Med. Chem. Lett. 2016, 26, 2133. |
| [25] | Stütz, A.; Georgopoulos, A.; Granitzer, W.; Petranyi, G.; Berney, D. J. Med. Chem. 1986, 29, 112. |
| [26] | Singh, B.; Chetia, D. Kumawat, M. K. Pharm. Chem. J. 2021, 55, 724. |
| [27] | Luo, L.; Song, Q.; Li, Y.; Cao, Z.; Qiang, X.; Tan, Z.; Deng, Y. Bioorg. Med. Chem. 2020, 28, 115400. |
| [28] | Bai, X. Y.; Zhao, W.; Sun, X.; Li, B. J. J. Am. Chem. Soc. 2019, 141, 19870. |
| [29] | Sun, S. Z.; Martin, R. Angew. Chem., Int. Ed. 2018, 57, 3622. |
| [30] | Jarava-Barrera, C.; Parra, A.; López, A.; Cruz-Acosta, F.; Collado-Sanz, D.; Cárdenas, D. J.; Tortosa, M. ACS Catal. 2016, 6, 442. |
| [31] | Mlynarski, S. N.; Karns, A. S.; Morken, J. P. J. Am. Chem. Soc. 2012, 134, 16449. |
| [32] | Yang, C.; Gao, Y.; Bai, S.; Jiang, C.; Qi, X. J. Am. Chem. Soc. 2020, 142, 11506. |
/
| 〈 |
|
〉 |