

碘化亚铜/4-羟基吡啶酰肼催化的(杂)芳基氯代物和苯基亚磺酸钠的偶联反应
收稿日期: 2024-11-14
修回日期: 2024-11-26
网络出版日期: 2024-11-28
基金资助
国家自然科学基金(21991110)
CuI/4-Hydroxy Picolinohydrazide Catalyzed Coupling Reaction of (Hetero)aryl Chlorides with Sodium Aryl Sulfinates
Received date: 2024-11-14
Revised date: 2024-11-26
Online published: 2024-11-28
Supported by
National Natural Science Foundation of China(21991110)
李赛洛 , 马大为 . 碘化亚铜/4-羟基吡啶酰肼催化的(杂)芳基氯代物和苯基亚磺酸钠的偶联反应[J]. 有机化学, 2025 , 45(4) : 1276 -1282 . DOI: 10.6023/cjoc202411014
By using a newly developed 4-hydroxy picolinohydrazide as the ligand, Cu-catalyzed coupling of (hetero)aryl chlorides with sodium aryl sulfonates proceeded smoothly at 130 ℃ to give a series of biarylsulfones in 53%~96% yields. This represents the first metal-catalyzed coupling reaction of (hetero)aryl chlorides with sodium aryl sulfonates. Aryl and heteroaryl chlorides bearing either electron-donating or electron-withdrawing groups were applicable for this coupling reaction.
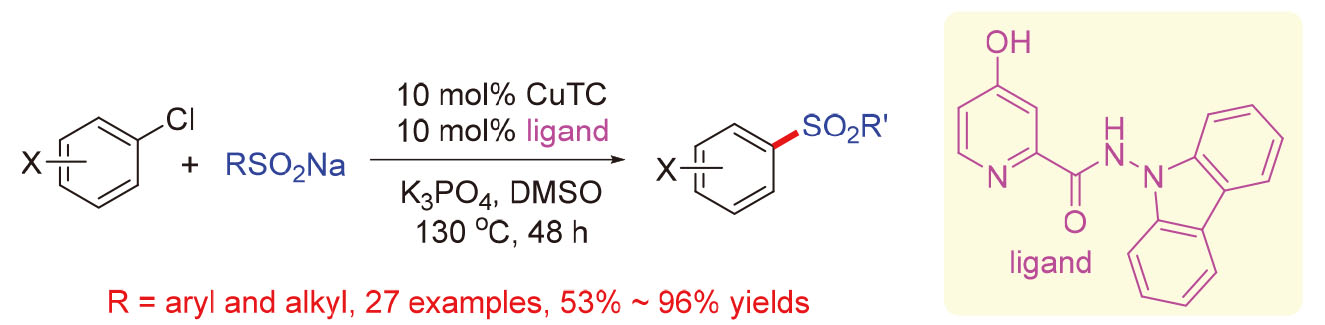
Key words: coupling reaction; copper catalysis; ligand; (hetero)aryl chloride; biarylsulfone
| [1] | Repichet, S.; Roux, C. L.; Dubac, J. J. Org. Chem. 1999, 64, 6429. |
| [2] | Fowler, J.; Logan, J.; Azzaro, A. Neuropsychopharmacol 2010, 35, 623. |
| [3] | Codony, X.; Vela, J. M.; Ramirez, M. J. Curr. Opin. Pharmacol. 2011, 11, 94. |
| [4] | Koch, O.; Bering, L. US 2020/0123130, 2020. |
| [5] | for a review, see: Liu, N.-W.; Liang, S.; Manolikakes, G. Synthesis 2016, 48, 1939. |
| [6] | for Cu-catalyzed coupling reactions with aryl halides, see: (a) Suzuki, H.; Abe, H. Tetrahedron Lett. 1995, 36, 6239. |
| [6] | (b) Baskin, J. M.; Wang, Z. Org. Lett. 2002, 4, 4423. |
| [6] | (c) Zhu, W.; Ma, D. J. Org. Chem. 2005, 70, 2696. |
| [6] | (d) Bian, M.; Xu, F.; Ma, C. Synthesis 2007, 19, 2951. |
| [6] | (e) Yuan, Y. Q.; Guo, S. R. Synlett 2011, 18, 2750. |
| [6] | (f) Ma, D.; Niu, S.; Zhao, J.; Jiang, X.; Jiang, Y.; Zhang, X.; Sun, T. Chin. J. Chem. 2017, 35, 1661. |
| [6] | (g) Zhao, J.; Niu, S.; Jiang, X.; Jiang, Y.; Zhang, X.; Sun, T.; Ma, D. J. Org. Chem. 2018, 83, 6589. |
| [6] | (h)for a review, see: Bhunia, S.; Pawar, G. G.; Kumar, S. V.; Jiang, Y.; Ma, D. Angew. Chem., Int. Ed. 2017, 56, 16136. |
| [7] | for Pd-catalyzed coupling reactions with aryl halides and triflates, see: (a) Cacchi, S.; Fabrizi, G.; Goggiamani, A.; Parisi, L. M. Org. Lett. 2002, 4, 4719. |
| [7] | (b) Smyth, L.; Phillips, E.; Chen, V.; Napolitana, J.; Henry, R.; Shekhar, S. J. Org. Chem. 2016, 81, 1285. |
| [8] | for Cu-catalyzed coupling reactions with other electrophiles, see: (a), Beaulieu, C.; Guay, D.; Wang, Z.; Evans, D. A. Tetrahedron Lett. 2004, 45, 3233. |
| [8] | (b) Kir, A.; Sayyed, I. A.; Lo, W. F.; Kaiser, H. M.; Beller, M.; Tse, M. K. Org. Lett. 2007, 9, 3405. |
| [8] | (c) Huang, F.; Batey, R. A. Tetrahedron 2007, 63, 7667. |
| [8] | (d) Yang, H.; Li, Y.; Jiang, M.; Wang, J.; Fu, H. Chem.-Eur. J. 2011, 17, 5652. |
| [8] | (e) Gund, S. H.; Shelkar, R. S.; Nagarkar, J. M. RSC Adv. 2015, 5, 62926. |
| [8] | (f) Zhang, K.; Xu, X. H.; Qing, F. L. J. Org. Chem. 2015, 80, 7658. |
| [9] | Li, Q.; Xu. L.; Ma, D. Angew. Chem., Int. Ed. 2022, 62, e202210483. |
| [10] | Xu, L.; Zhou, J.; Shen, X.; Chai, J.; Shi, L.; Wu, B.; Li, W.; Ma, D. Angew. Chem., Int. Ed. 2024, 64, e202412552. |
/
| 〈 |
|
〉 |