

Co@NaKETS-10双功能催化剂高选择性催化伯胺脱氢制备腈
收稿日期: 2024-09-22
修回日期: 2024-10-28
网络出版日期: 2024-11-28
基金资助
安徽省教育厅重点研发基金(2024AH051331); 巢湖学院科研启动基金(KYQD-2023053)
Highly Selective Dehydrogenation of Primary Amine to Nitrile over Bifunctional Co@NaKETS-10 Catalyst
Received date: 2024-09-22
Revised date: 2024-10-28
Online published: 2024-11-28
Supported by
Key Research and Development Plan of Anhui Province(2024AH051331); Research Start-Up Funds of Chaohu University(KYQD-2023053)
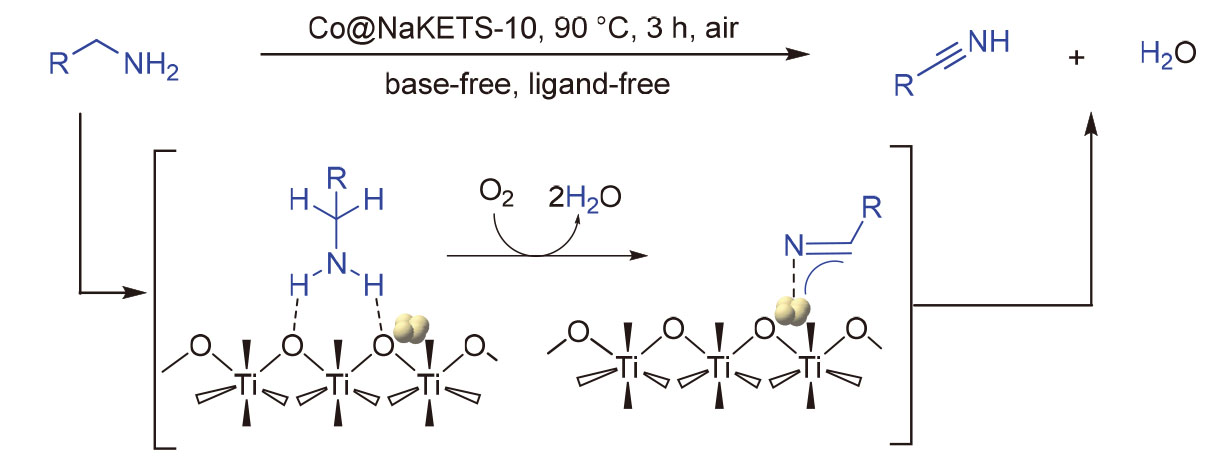
使用碱性NaKETS-10沸石担载金属Co作为双功能催化剂, 在温和条件下催化多种伯胺发生脱氢反应, 实现了高选择性地制备腈. NaKETS-10晶体中的-Ti-O-Ti-链具有电子传输性能, 导致其中的氧原子具有高的电子密度, 因而具有优异的给电子能力. 研究发现, NaKETS-10中富电子氧是伯胺转化过程中的吸附位点, 氨基上的两个H原子同时被临近的富电子氧吸附; Co金属位点与活化后的胺分子配位, 和富电子氧协同催化吸附态胺分子的脱氢反应. 此外, 作为非均相催化剂, Co@NaKETS-10展示出了优异的稳定性, 在循环使用5次后没有明显的活性损失. 最后, 结合平行实验和对照实验结果, 初步研究了伯胺在Co@NaKETS-10催化剂上脱氢历程的基元反应步骤, 并提出了可能的反应机理.
关键词: Co@NaKETS-10; 伯胺; 脱氢反应; 富电子氧
朱超杰 , 王乐乐 , 孟荣 , 王永吉 , 王成 , 王小东 . Co@NaKETS-10双功能催化剂高选择性催化伯胺脱氢制备腈[J]. 有机化学, 2025 , 45(4) : 1268 -1275 . DOI: 10.6023/cjoc202409030
Basic zeolite NaKETS-10 was used as a support for Co metal catalyst in the double dehydrogenation reactions of various benzylamines under mild conditions, leading to the high selective formation of nitriles. The -Ti-O-Ti- chain within NaKETS-10 crystal possesses an electron transport function, resulting in the oxygen atom possessing a high electron density and thus acting as an effective electron donor. Research indicates that the electron-rich oxygen in the NaKETS-10 framework is the adsorption and activation site for benzylamine. This study also reveals that the Co metal site coordinates with the activated benzylamine molecule, working in conjunction with the electron-rich oxygen to facilitate the dehydrogenation process of benzylamine. Furthermore, as a heterogeneous catalyst, the Co@NaKETS-10 demonstrates a remarkable stability and maintains its activity even after being recycled at least five times. Combining the results of parallel experiments and controlled experiments, the elementary reaction steps of benzylamine double dehydrogenation on the Co@NaKETS-10 catalyst were investigated, and a possible mechanism was proposed.

| [1] | (a) Sheldon, R. A.; Arends, I. W. C. E.; Dijisman, D. Catal. Today 2000, 57, 157. |
| [1] | (b) Miller, J. S.; Manson, J. L. Acc. Chem. Res., 2001, 34, 563. |
| [1] | (c) Fleming, F. F.; Wang, Q. Chem. Rev. 2003, 103, 2035. |
| [1] | (d) Kim, J.; Kim, H. J.; Chang, S. Angew. Chem., Int. Ed. 2012, 51, 11948. |
| [2] | Sandmeyer, T.; Dtsch, B. Chem. Ges. 1884, 17, 1633. |
| [3] | Rosenmund, K. W.; Struck, E.; Dtsch, B. Chem. Ges. 1919, 52, 1749. |
| [4] | Zuin, V. G.; Eilks, I.; Elschami, M.; Kümmerer, K. Green Chem. 2021, 23, 1594. |
| [5] | (a) Dobereiner, G. E.; Crabtree, R. H. Chem. Rev. 2010, 110, 681. |
| [5] | (b) Gunanathan, C.; Milstein, D. Science 2013, 341, 1229712. |
| [5] | (c) Chen, C.; Verpoort, F.; Wu, Q. RSC Adv. 2016, 6, 55599. |
| [6] | (a) Tseng, K. N. T.; Rizzi, A. M.; Szymczak, N. K. J. Am. Chem. Soc. 2013, 135, 16352. |
| [6] | (b) Tseng, K. N. T.; Szymczak, N. K. Synlett 2014, 25, 2385. |
| [6] | (c) Espinosa, D. V.; Beltrln, A. M.; Mata, J. A. Chem.-Eur. J. 2016, 22, 17758. |
| [7] | Paramathevar, N.; Kumar, V. Org. Chem. Front. 2019, 6, 2570. |
| [8] | (a) Franc, G.; Jutand, A. Dalton Trans. 2010, 39, 7873. |
| [8] | (b) Perego, L. A.; Blieck, R.; Groue, A.; Monnier, F.; Taillefer, M.; Ciofini, I.; Grimaud, L. ACS Catal. 2017, 7, 4253. |
| [9] | Yan, X.-L.; Dong, Q.; Li, Y.; Meng, L.-Z.; Hao, Z.-Q.; Han, Z.-G.; Lu, G.-L.; Lin, J. Dalton Trans. 2020, 49, 3480. |
| [10] | Ray, R.; Chandra, S.; Yadav, V.; Mondal, P.; Maiti, D.; Lahiri, G. K. Chem. Commun. 2017, 53, 4006. |
| [11] | Ovoshchnikov, D. S.; Donoeva, B. G.; Golovko, V. B. ACS Catal. 2015, 5, 34. |
| [12] | Zhang, J.-T.; Wang, Z.-T.; Wang, Y.; Wan, C.-F.; Zheng, X.-Q.; Wang, Z.-Y. Green Chem. 2009, 11, 1973. |
| [13] | Kim, J. W.; Yamaguchi, K.; Mizuno, N. Angew. Chem., Int. Ed., 2008, 47, 9249. |
| [14] | (a) Liu, C.-J.; Hu, H.-L.; Liu, C.-H.; Zhu, C.-J.; Tang, T.-D. Chin. J. Org. Chem. 2023, 43, 2953 (in Chinese). |
| [14] | (刘长俊, 胡慧玲, 刘宬宏, 朱超杰, 唐天地, 有机化学, 2023, 43, 2953.) |
| [14] | (b) Liu, H.-L.; Zhu, C.-J.; Tang, T.-D. Chin. J. Org. Chem. 2022, 42, 1792 (in Chinese). |
| [14] | (刘会丽, 朱超杰, 唐天地, 有机化学, 2022, 42, 1792.) |
| [15] | Zhu, C.-J.; Wu, D.-F.; Liu, H.-L.; Meng, C.-W.; Tang, T.-D. Green Chem. 2022, 24, 9033. |
| [16] | Zhu, C.-J.; Fu, W.-Q.; Zhang, L.; Zhang, Y.; Wu, D.-F.; Tang, T.-D. Molecular Catal. 2022, 533, 112751. |
| [17] | Nie, X.-F.; Zheng, Y.-L.; Ji, L.; Fu, H.-Y.; Chen, H.; Li, R.-X. J. Catal. 2020, 391, 378. |
/
| 〈 |
|
〉 |