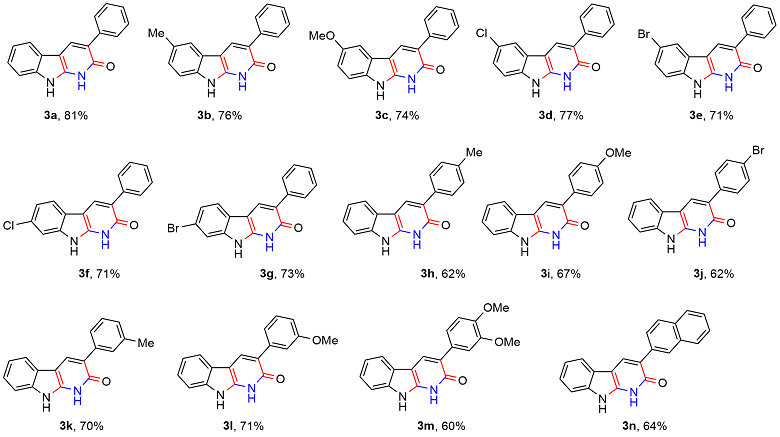
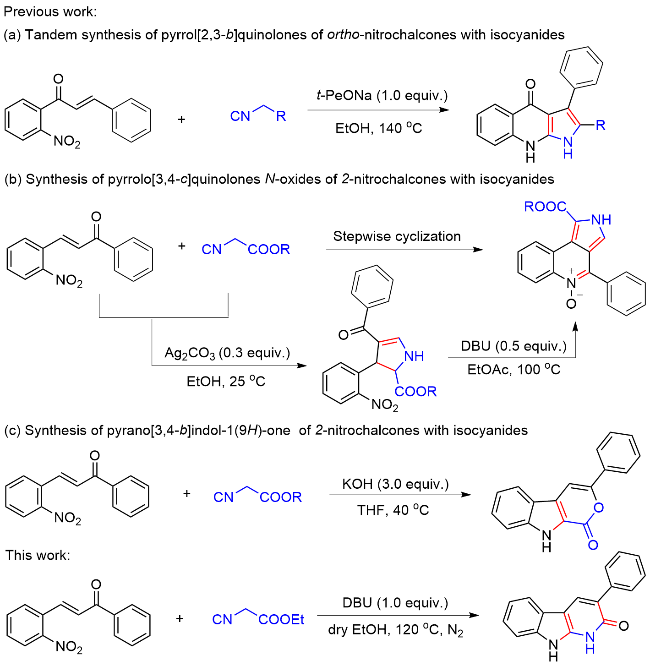
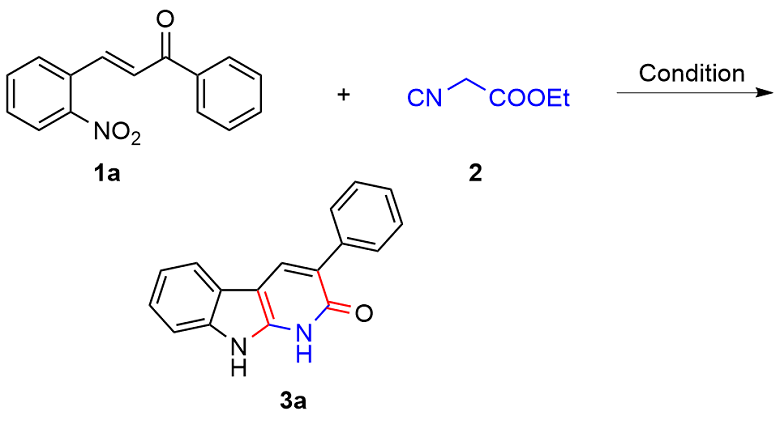
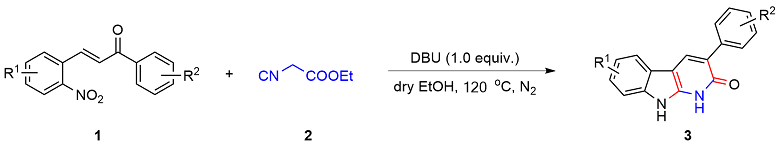
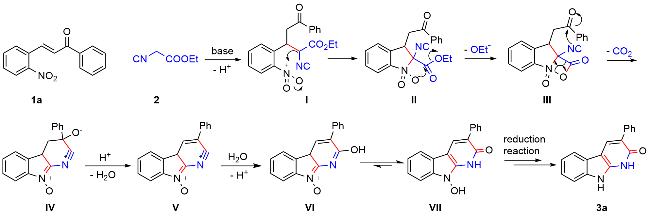
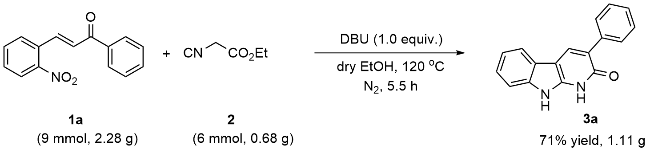以底物3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3a)的合成为例: 真空环境下, 在25 mL的圆底烧瓶中将(E)-3-(2-硝基苯基)-1-苯基丙-2-烯-1-酮(1a) (0.076 g, 0.3 mmol)与异氰基乙酸乙酯(2) (0.023 g, 0.2 mmol)溶解于10 mL超干乙醇溶剂中, 向体系加入DBU (0.070 g, 0.3 mmol), 120 ℃ N2保护条件下搅拌反应4 h. 待反应完成后(TLC监测), 将体系缓慢倒入水中, 加饱和的NH4Cl水溶液以淬灭反应. 然后将混合溶液用乙酸乙酯(20 mL×3)萃取, 合并有机相, Na2SO4干燥, 真空浓缩. 残余物经硅胶柱层析纯化(层析液: 石油醚/乙酸乙酯, V/ V=20/1), 得到3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3a), 产率为81%. 以相同方法合成化合物3a~3n.
3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3a): 收率81%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.02 (s, 1H), 11.61 (s, 1H), 8.12 (d, J=6.0 Hz, 1H), 7.81 (d, J=12.0 Hz, 2H), 7.54 (d, J=6.0 Hz, 1H), 7.49 (t, J=9.0 Hz, 2H), 7.44~7.39 (m, 3H), 7.21 (t, J=9.0 Hz, 1H); 13C NMR (151 MHz, DMSO-d6) δ: 156. 6, 139.9, 136. 8, 135.1, 129.2 (2C), 128.9, 127.6, 127.0, 127.0 (2C), 125.3, 122.7, 122.0, 120.2, 113.0, 99.2. HRMS (ESI) calcd for C17H13N2O [M+H]+ 261.1023, found 261.1030.
6-甲基-3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3b): 收率76%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.00 (s, 1H), 11.57 (s, 1H), 8.11 (d, J=12.0 Hz, 1H), 7.71 (d, J=12.0 Hz, 2H), 7.54 (d, J=12.0 Hz, 1H), 7.43 (t, J=9.0 Hz, 1H), 7.36 (d, J=6.0 Hz, 1H), 7.30 (d, J=6.0 Hz, 2H), 7.20 (t, J=6.0 Hz, 1H), 2.36 (s, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 156.5, 138.2, 137.1, 135.0, 129.2 (2C), 128.9, 128.7, 127.0 (2C), 126. 9, 124.6, 124.5, 123.8, 121.4, 114.6, 99.1, 60.2. HRMS (ESI) calcd for C18H15N2O [M+H]+ 275.1179, found 275.1168.
6-甲氧基-3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3c): 收率74%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 11.90 (s, 1H), 11.57 (s, 1H), 7.86 (d, J=12.0 Hz, 2H), 7.72 (d, J=6.0 Hz, 1H), 7.54 (d, J=6.0 Hz, 2H), 7.49~7.45 (m, 3H), 7.14~7.12 (m, 1H), 3.90 (s, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 156.6, 154.1, 136.0, 135.2, 135.0, 129.2 (2C), 128.6, 128.2, 126.8 (2C), 124.9, 123.0, 117.6, 113.8, 103.0, 99.3, 55.9. HRMS (ESI) calcd for C18H15N2O2 [M+H]+ 291.1129, found 291.1113.
6-氯-3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3d): 收率77%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.23 (s, 1H), 11.69 (s, 1H), 8.25 (d, J=6.0 Hz, 1H), 7.80 (d, J=12.0 Hz, 2H), 7.55 (d, J=6.0 Hz, 1H), 7.50 (t, J=6.0 Hz, 2H), 7.44~7.41 (m, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 156.5, 138.2, 137.1, 135.0, 129.2 (2C), 128.9, 128.7, 127.0 (2C), 126. 9, 124.6, 124.5, 123.9, 121.4, 114.6, 99.1. HRMS (ESI) calcd for C17H12ClN2O [M+H]+ 295.0633, found 295.0630.
6-溴-3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3e): 收率71%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.24 (s, 1H), 11.68 (s, 1H), 8.39 (s, 1H), 7.80 (d, J=12.0 Hz, 2H), 7.55~7.54 (m, 1H), 7.51~7.48 (m, 3H), 7.46 (s, 1H), 7.42 (t, J=6.0 Hz, 1H); 13C NMR (151 MHz, DMSO-d6) δ: 156.5, 138.5, 137.1, 135.0, 129.4, 129.2 (2C), 128.9, 128.5, 127.0 (2C), 124.5, 124.5, 124.4, 115.0, 112.3, 99.1. HRMS (ESI) calcd for C17H12BrN2O [M+H]+ 339.0128, found 339.0131.
7-氯-3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3f): 收率71%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.19 (s, 1H), 11.69 (s, 1H), 8.15 (d, J=12.0 Hz, 1H), 7.80 (d, J=12.0 Hz, 2H), 7.53 (s, 1H), 7.50 (t, J=6.0 Hz, 2H), 7.43 (t, J=6.0 Hz, 1H), 7.40 (s, 1H), 7.24~7.23 (m, 1H); 13C NMR (151 MHz, DMSO-d6) δ: 156.4, 140.2, 137.4, 135.0, 131.4, 129.2 (2C), 128.9, 128.3, 127.1 (2C),125.0, 123.6, 121.6, 120.5, 112.5, 98.9. HRMS (ESI) calcd for C17H12ClN2O [M+H]+ 295.0633, found 295.0623.
7-溴-3-苯基-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3g): 收率73%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.19 (s, 1H), 11.70 (s, 1H), 8.10~8.08 (d, J=12.0 Hz, 1H), 7.81~7.79 (d, J=12.0 Hz, 2H), 7.69~7.68 (d, J=6.0 Hz, 1H), 7.51~7.48 (t, J=9.0 Hz, 2H), 7.44~7.43 (d, J=6.0 Hz, 1H), 7.39 (s, 1H), 7.36~7.34 (m, 1H); 13C NMR (151 MHz, DMSO-d6) δ: 156.4, 140.6, 137.4, 135.0, 129.2 (2C), 128.9, 128.2, 127.1 (2C), 125.0, 123.9, 123.1, 121.9, 119.7, 115.5, 98.9. HRMS (ESI) calcd for C17H12BrN2O [M+H]+ 339.0128, found 339.0116.
3-(4-甲基苯基)-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3h): 收率62%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.00 (s, 1H), 11.57 (s, 1H), 8.11 (d, J=12.0 Hz, 1H), 7.71 (d, J=12.0 Hz, 2H), 7.54 (d, J=12.0 Hz, 1H), 7.43 (t, J=6.0 Hz, 1H), 7.35 (s, 1H), 7.30 (d, J=6.0 Hz, 2H), 7.20 (t, J=6.0 Hz, 1H), 2.36 (s, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 156.5, 139.9, 138.2, 136.8, 132.4, 129.7 (2C), 127.5, 126.8, 126.8 (2C), 125.2, 122.7, 121.9, 120.0, 113.0, 98.5, 21.2. HRMS (ESI) calcd for C18H15N2O [M+H]+ 275.1179, found 275.1171.
3-(4-甲氧基苯基)-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3i): 收率67%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 11.96 (s, 1H), 11.52 (s, 1H), 8.10 (d, J=12.0 Hz, 1H), 7.77~7.74 (m, 2H), 7.54 (d, J=6.0 Hz, 1H), 7.44~7.41 (m, 1H), 7.29 (s, 1H), 7.19 (t, J=6.0 Hz, 1H),7.05~7.04 (m, 2H), 3.82 (s, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 159.9, 156.6, 139.9, 136.7, 128.3 (2C), 127.6, 127.3, 126.8, 125.4, 122.7, 121.9, 119.9, 114.6 (2C), 113.0, 98.0, 55.7. HRMS (ESI) calcd for C18H15N2O2 [M+H]+ 291.1129, found 291.1120.
3-(4-溴苯基)-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3j): 收率62%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.06 (s, 1H), 11.66 (s, 1H), 8.11 (d, J=12.0 Hz, 1H), 7.77~7.76 (m, 2H), 7.69~7.67 (m, 2H), 7.55 (d, J=6.0 Hz, 1H), 7.45~7.42 (m, 2H), 7.21 (t, J=6.0 Hz, 1H); 13C NMR (151 MHz, DMSO-d6) δ: 156.5, 139.9, 135.5, 134.3, 132.0 (2C), 129.0 (2C), 127.9, 126.9, 125.0, 122.7, 122.0, 121.9, 120.2, 113.0, 99.6. HRMS (ESI) calcd for C17H12BrN2O [M+H]+ 339.0128, found 339.0130.
3-(3-甲基苯基)-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3k): 收率70%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.01 (s, 1H), 11.54 (s, 1H), 8.12 (d, J=6.0 Hz, 1H), 7.66 (s, 1H), 7.61 (d, J=6.0 Hz, 1H), 7.54 (d, J=12.0 Hz, 1H), 7.44~7.42 (m, 1H), 7.39~7.35 (m, 2H), 7.24~7.19 (m, 2H), 2.40 (s, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 156.5, 139.9, 138.4, 136.9, 135.1, 129.7, 129.4, 129.1, 127.6, 126.8, 125.2, 124.1, 122.8, 122.0, 120.1, 113.0, 98.9, 21.5. HRMS (ESI) calcd for C18H15N2O [M+H]+ 275.1179, found 275.1163.
3-(3-甲氧基苯基)-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3l): 收率71%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.01 (s, 1H), 11.54 (s, 1H), 8.12 (d, J=6.0 Hz, 1H), 7.66 (s, 1H), 7.61 (d, J=6.0 Hz, 1H), 7.54 (d, J=12.0 Hz, 1H), 7.44~7.42 (m, 1H), 7.39~7.35 (m, 2H), 7.24~7.19 (m, 2H), 2.40 (s, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 157.0, 156.5, 139.9, 136.5, 136.5, 130.2, 127.8, 126.9, 125.1, 122.8, 122.0, 120.1, 119.2, 114.8, 113.0, 112.0, 99.2, 55.7. HRMS (ESI) calcd for C18H15N2-O2 [M+H]+ 291.1129, found 291.1133.
3-(3,4-二甲氧基苯基)-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3m): 收率60%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 11.97 (s, 1H), 11.55 (s, 1H), 8.12 (d, J=6.0 Hz, 1H), 7.54 (d, J=6.0 Hz, 1H), 7.44~7.41 (m, 2H), 7.39~7.37 (m, 1H), 7.35 (s, 1H), 7.20 (t, J=6.0 Hz, 1H), 7.06 (d, J=6.0 Hz, 1H), 3.91 (s, 3H), 3.82 (s, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 156.6, 149.3, 149.2, 139.9, 136.8, 127. 8, 127.3, 126.8, 125.4, 122.7, 122.0, 119.9, 119.3, 113.0, 112.3, 110.6, 98.1, 56.1, 56.1. HRMS (ESI) calcd for C19H17N2O3 [M+H]+ 321.1234, found 321.1224.
3-(萘-2-基)-1,9-二氢-2H-吡啶并[2,3-b]吲哚-2-酮(3n): 收率64%, 淡黄色油状物. 1H NMR (600 MHz, DMSO-d6) δ: 12.07 (s, 1H), 11.72 (s, 1H), 8.41 (s, 1H), 8.16 (d, J=12.0 Hz, 1H), 8.03 (d, J=12.0 Hz, 1H), 8.00~7.96 (m, 4H), 7.58~7.56 (m, 3H), 7.45 (t, J=6.0 Hz, 1H), 7.23 (t, J=6.0 Hz, 1H); 13C NMR (151 MHz, DMSO-d6) δ: 156.6, 139.9, 136.5, 133.4, 133.0, 132.4, 128.8, 128. 7, 128.0, 127.8, 127.1, 127.0, 126.9, 125.7, 125.2, 125.0, 122.8, 122.0, 120.2, 113.0, 99.7. HRMS (ESI) calcd for C21H15N2O [M+H]+ 311.1179, found 311.1186.