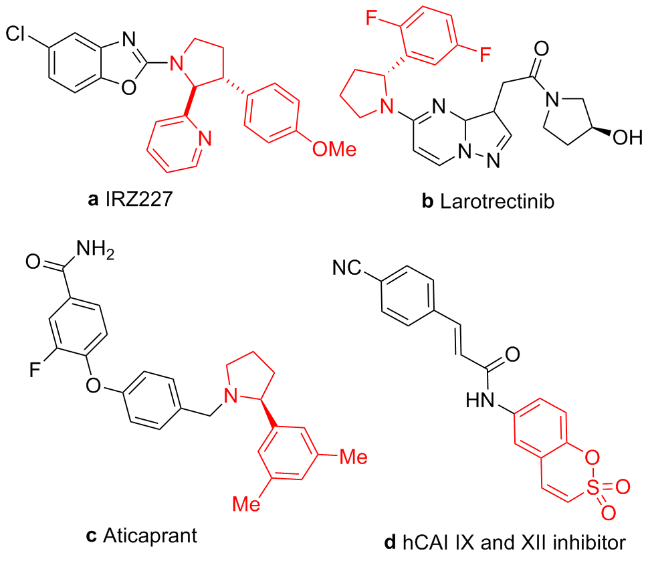
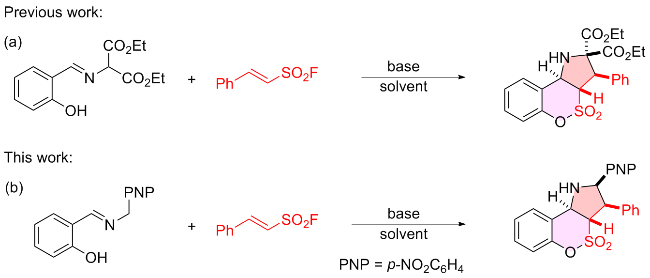
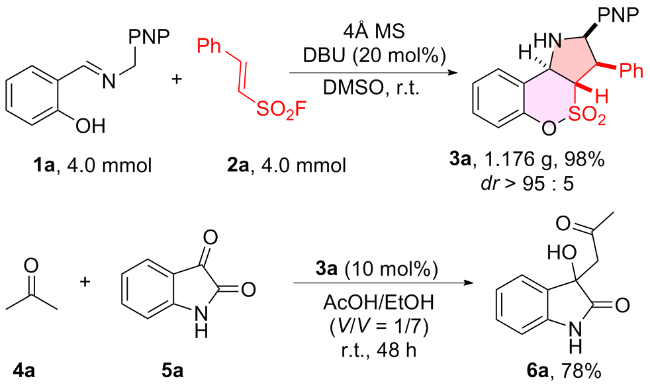
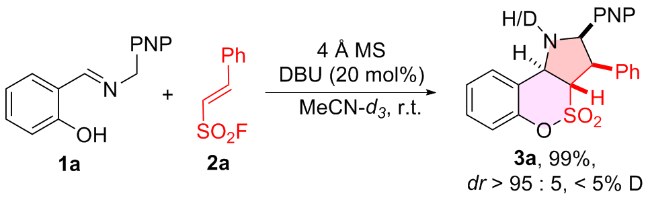
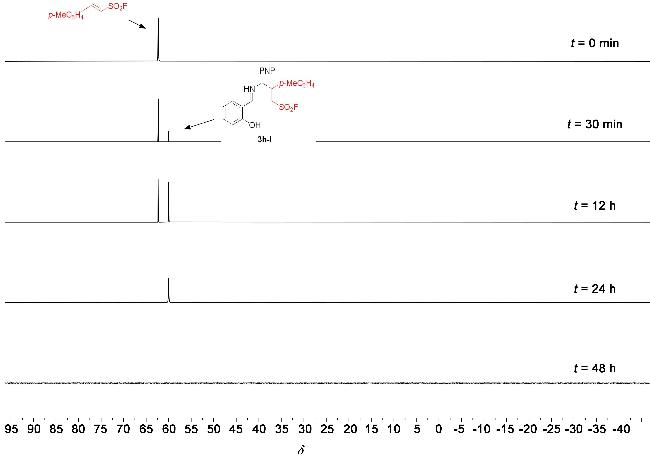
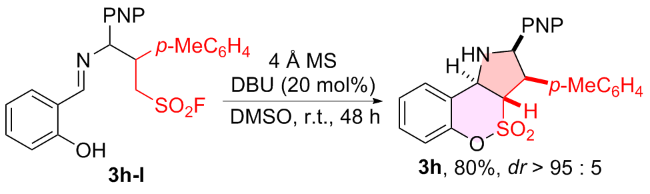
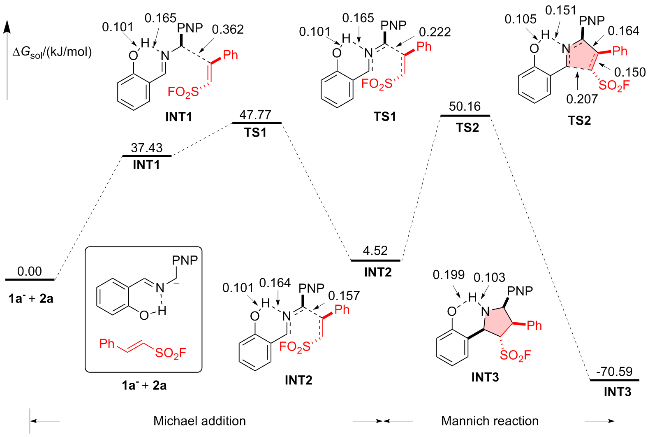
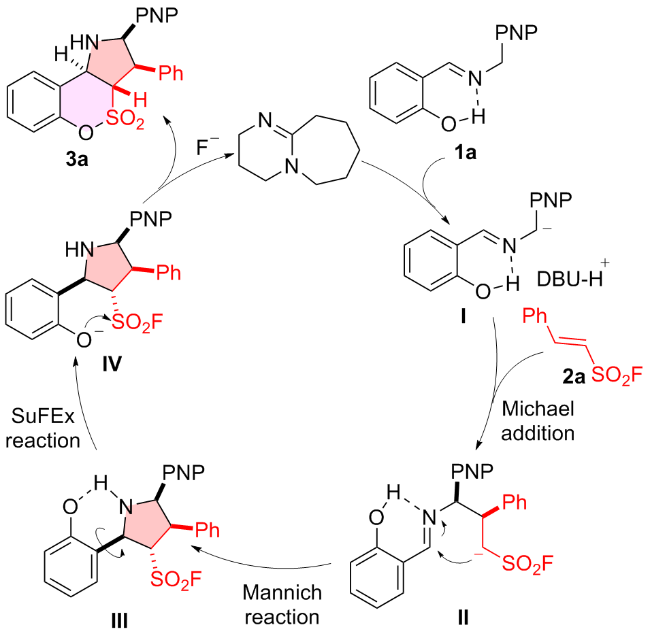
1 结果与讨论
表1 o-羟基席夫碱的筛选aTable 1 Screening of o-hydroxy Schiff bases  |
| Entry | Ar1 | Hirshfeld charge | Yieldb/% | drc |
|---|---|---|---|---|
| 1 | Ph | 0.0737 | N.D.d | — |
| 2 | 4-MeOC6H4 | 0.0733 | N.D.d | — |
| 3 | 4-CF3C6H4 | 0.0755 | <10% | — |
| 4 | 4-CNC6H4 | 0.0763 | <10% | — |
| 5 | 4-NO2C6H4 | 0.0767 | 56 | 83∶17 |
| 6 | 2,4,6-F3C6H2 | 0.0698 | N.D.d | — |
| 7 | 2,3,4,5,6-F5C6 | 0.0738 | N.D.d | — |
| 8 | 3,5-(CF3)2C6H3 | 0.0774 | Complex | — |
a Reaction conditions: 1 (0.20 mmol), 2h (0.20 mmol), Et3N (20 mol%), 4 Å MS (200 mg), MeCN (2.0 mL), r.t., 48 h, atmosphere. b Determined by 1H NMR using CH2Br2 as the internal standard. c Diastereomeric ratio determined by 1H NMR spectroscopy. d N.D.=Not detected. |
表2 反应条件优化aTable 2 Optimization of reaction conditions 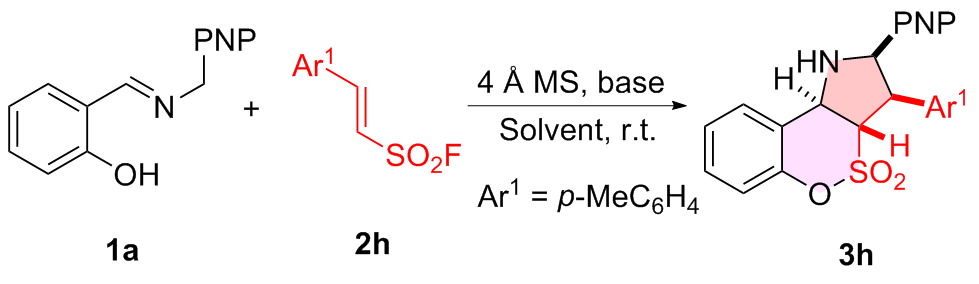 |
| Entry | Base | Solvent | Yieldb/% | drc |
|---|---|---|---|---|
| 1 | Et3N | MeCN | 56 | 83∶17 |
| 2 | DIPEA | MeCN | 42 | 92∶8 |
| 3 | DBU | MeCN | 92 | 94∶6 |
| 4 | DBN | MeCN | 89 | 91∶9 |
| 5 | Cs2CO3 | MeCN | 31 | 90∶10 |
| 6 | DBU | DMSO | 99 (99)d | >95∶5 |
| 7 | DBU | DMF | 94 | >95∶5 |
| 8 | DBU | DCM | 41 | 85∶15 |
| 9 | DBU | THF | 57 | 93∶7 |
| 10 | DBU | MeOH | 21 | 51∶49 |
| 11 | DBU | EtOH | 34 | 56∶44 |
| 12 | DBU | H2O | Trace | — |
| 13e | DBU | DMSO | 66 | >95∶5 |
| 14f | — | DMSO | N.D.j | — |
| 15g | DBU | DMSO | 40 | >95∶5 |
| 16h | DBU | DMSO | 92 | >95∶5 |
| 17i | — | DMSO | N.D.j | — |
a Reaction conditions: 1a (0.20 mmol), 2h (0.20 mmol), base (20 mol%), 4 Å MS (200 mg), solvent (2.0 mL), r.t., 48 h, atmosphere, PNP=p-nitrophenyl. b Determined by 1H NMR using CH2Br2 as the internal standard. c Diastereomeric ratio determined by 1H NMR spectroscopy. d Isolated yield. e DBU (10 mol%) was used. f Without DBU. g Without 4 Å MS. h 4 Å MS (100 mg) was used. i Without DBU and 4 Å MS. j N.D.=Not detected. |
表3 2-取代乙烯基磺酰氟普适性研究aTable 3 Scope of 2-substituted vinylsulfonyl fluorides  |
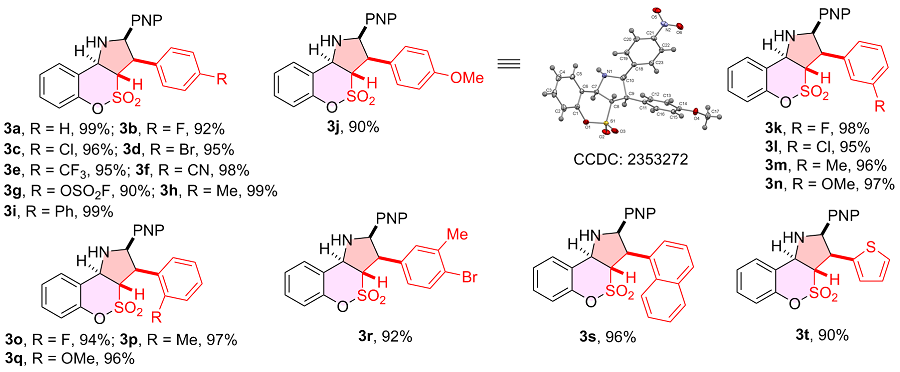 |
a Reaction conditions: 1a (0.20 mmol), 2 (0.20 mmol), DBU (20 mol%), 4 Å MS (200 mg), DMSO (2.0 mL), r.t., 48 h, atmosphere. Diastereomeric ratio determined by 1H NMR spectroscopy, all of the case dr>95∶5. Isolated yield. |
表4 o-羟基席夫碱普适性研究aTable 4 Scope of o-hydroxy Schiff bases 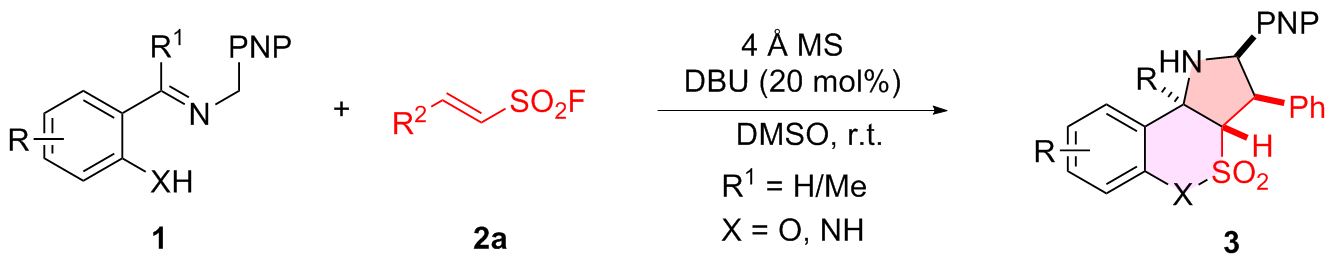 |
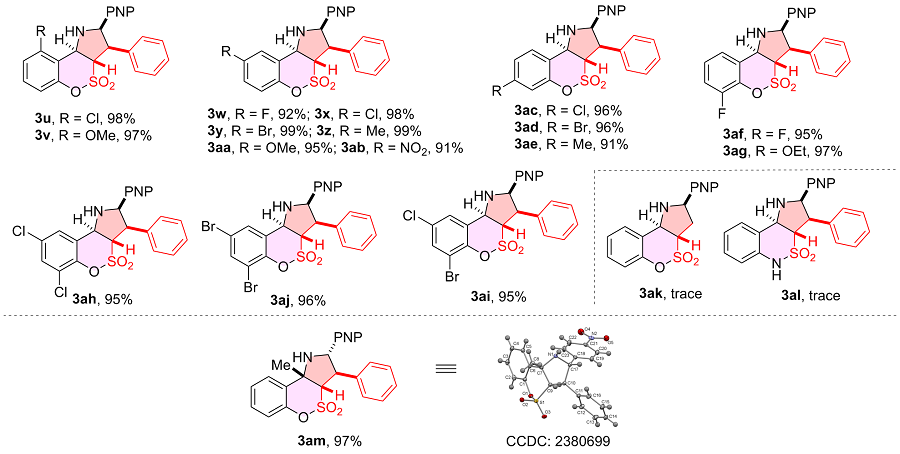 |
a Reaction conditions: 1 (0.20 mmol), 2a (0.20 mmol), DBU (20 mol%), 4 Å MS (200 mg), DMSO (2.0 mL), r.t., 48 h, atmosphere; diastereomeric ratio determined by 1H NMR spectroscopy, all of the case dr>95∶5. Isolated yield. |
表5 抑金黄壳囊孢菌活性测试aTable 5 Antifungal activity test of C. chrysosporium |
| Compd. | Inhibitory rate±SD/% | Compd. | Inhibitory rate±SD/% |
|---|---|---|---|
| 3a | 65.1±2.3 | 3l | 43.1±7.1 |
| 3b | 60.9±6.4 | 3m | 45.3±3.2 |
| 3c | 48.3±2.3 | 3n | 61.4±4.1 |
| 3d | 58.9±2.3 | 3o | 77.0±2.6 |
| 3e | 64.5±1.8 | 3p | 45.2±2.3 |
| 3f | 76.8±4.7 | 3q | 40.4±7.7 |
| 3g | 63.5±7.7 | 3r | 23.3±3.2 |
| 3h | 58.4±3.8 | 3s | 53.3±3.8 |
| 3i | 59.1±4.0 | 3t | 31.4±3.5 |
| 3j | 48.2±3.2 | 3u | 51.8±0.8 |
| 3k | 53.8±1.5 | 3v | 50.2±4.0 |
a 50 mg/L drug-containing culture medium was used; each test was performed in triplicate. |