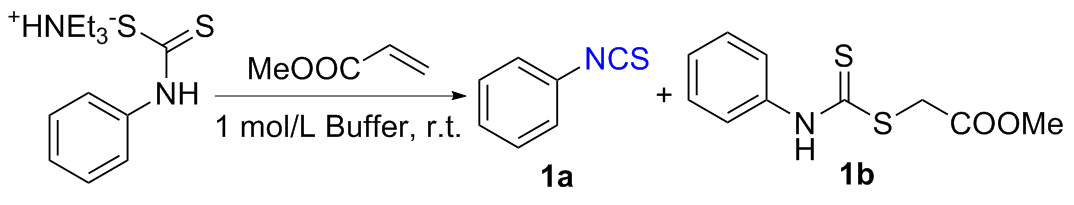
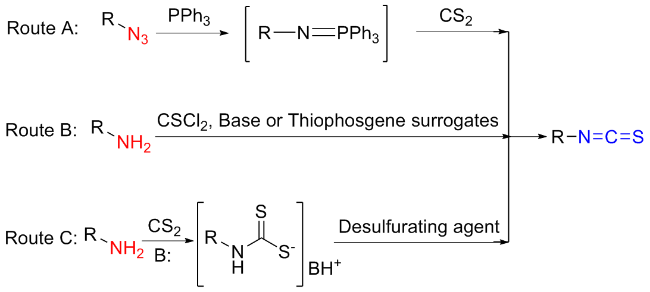
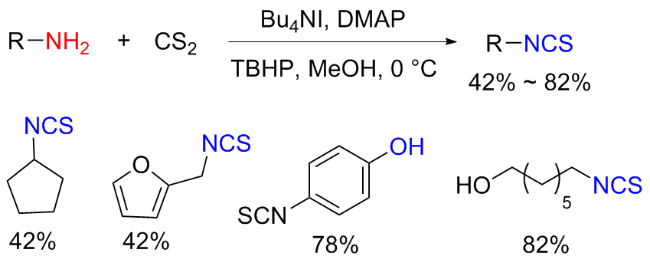
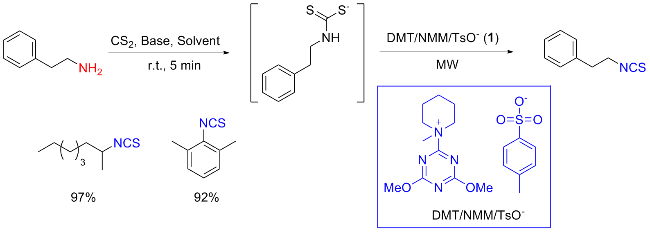
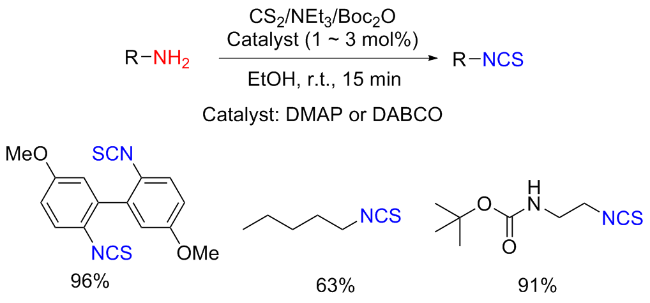
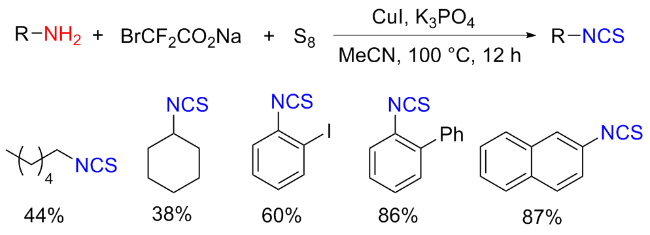
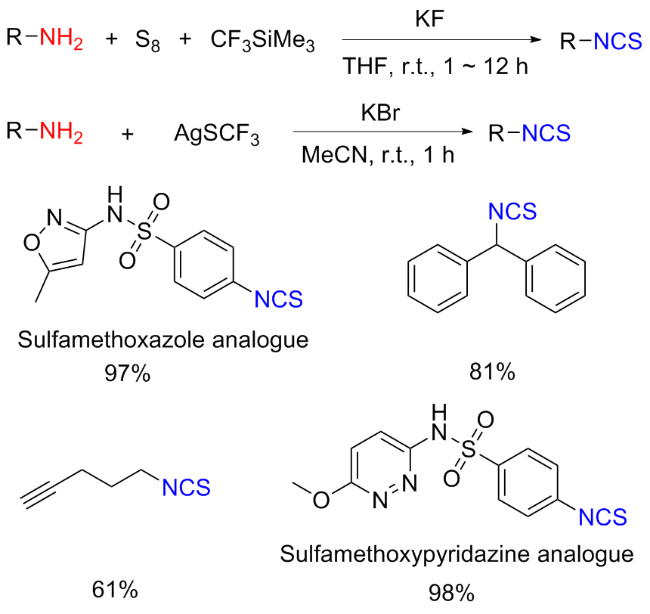
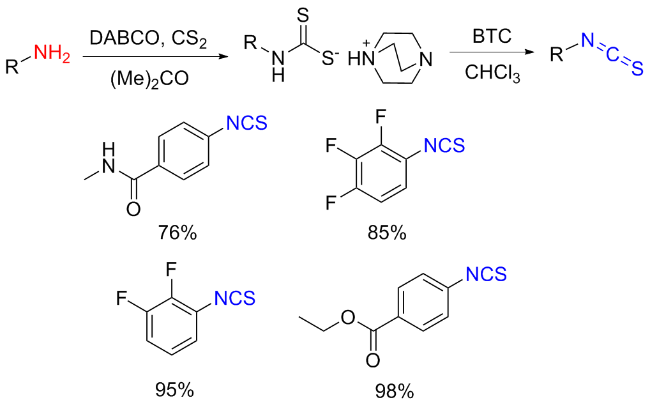
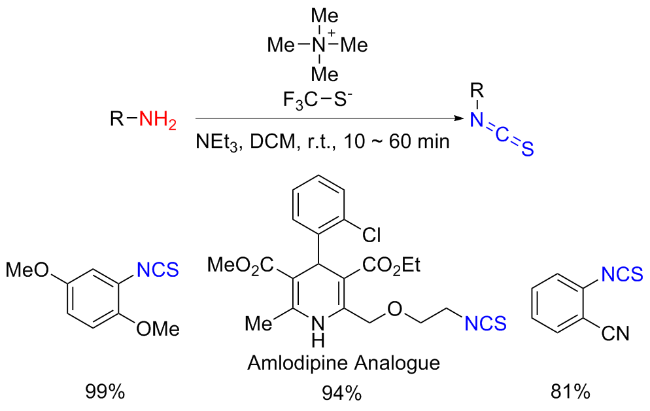
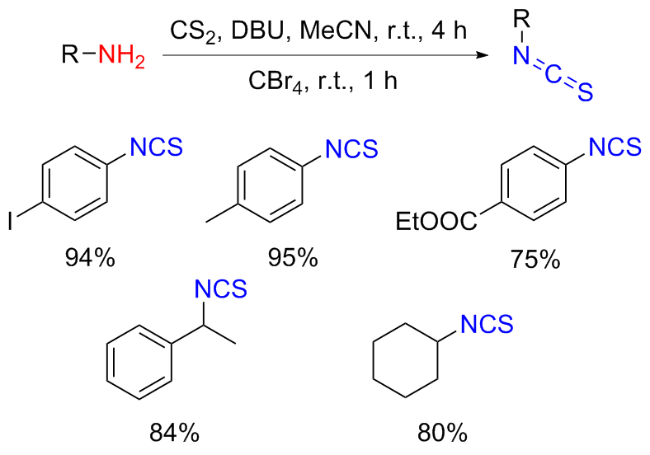
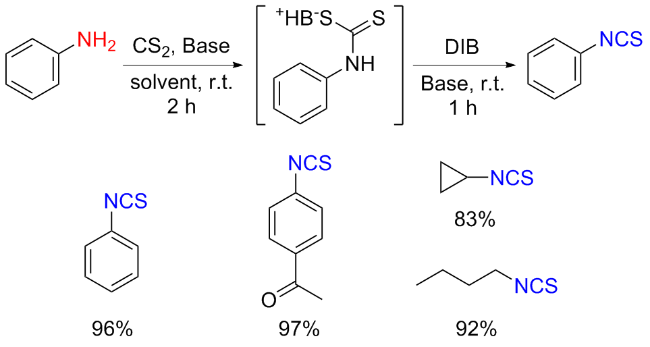
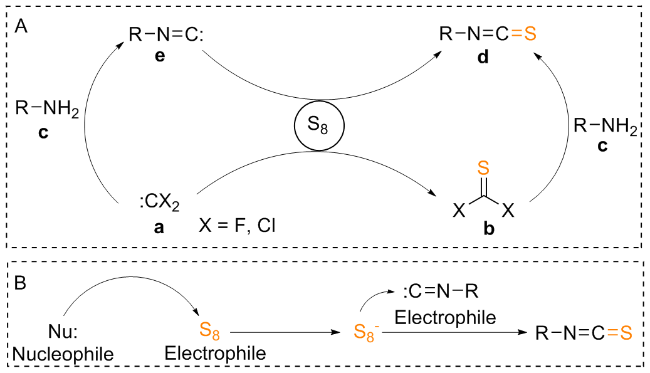
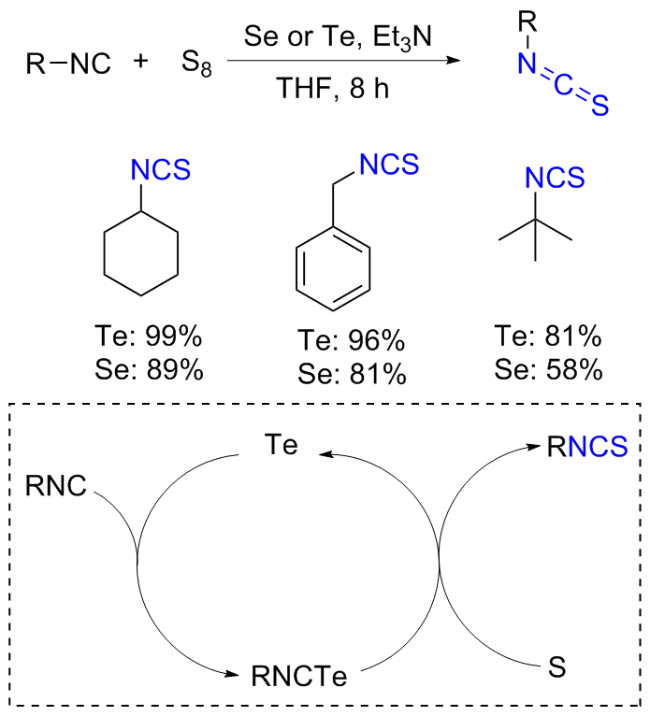
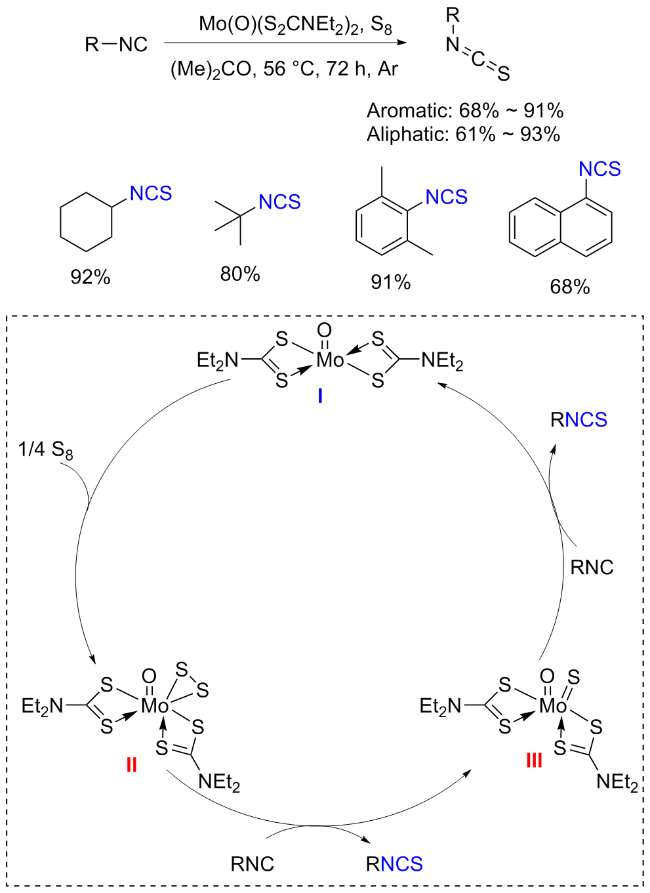
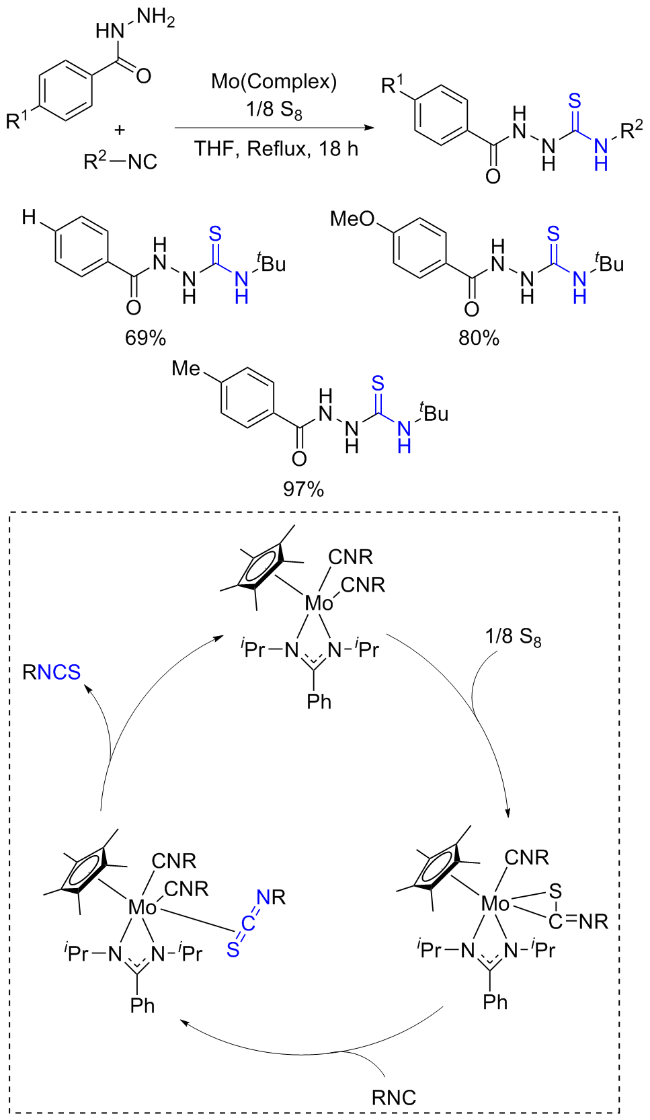
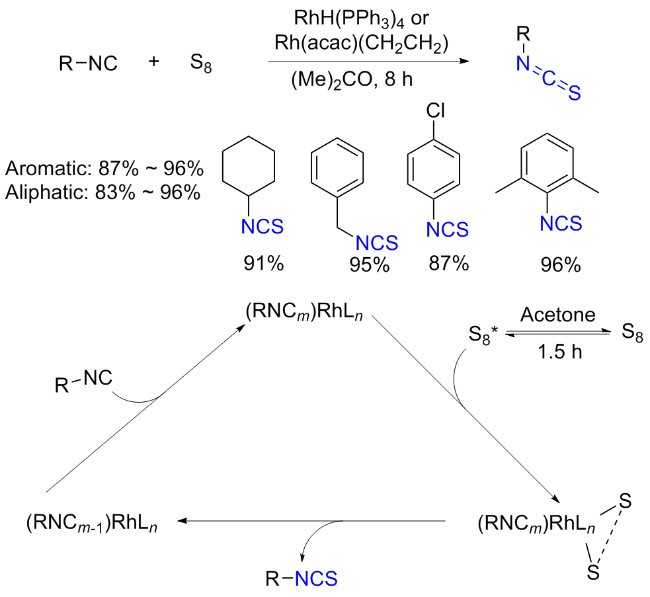
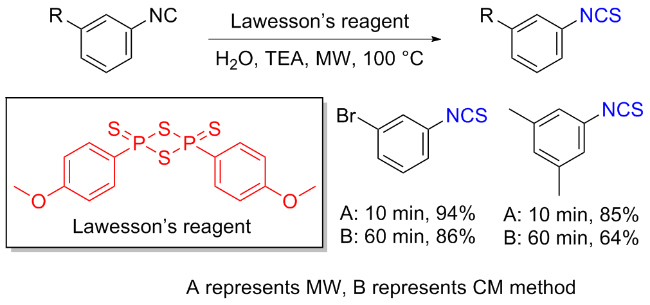
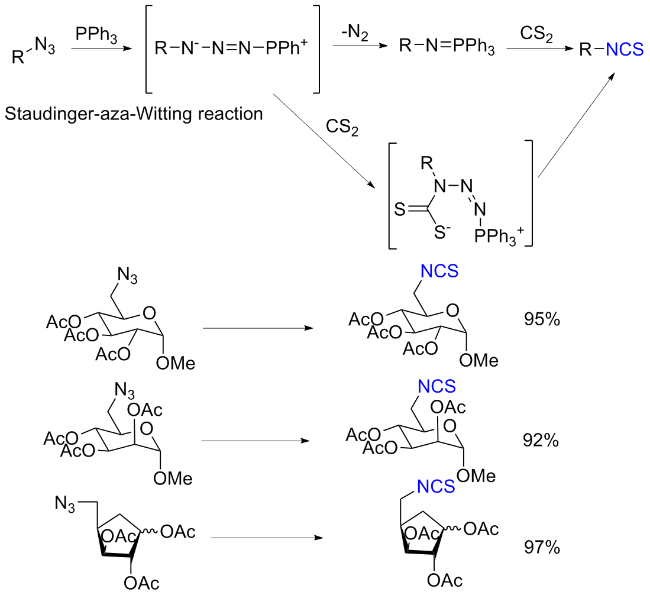
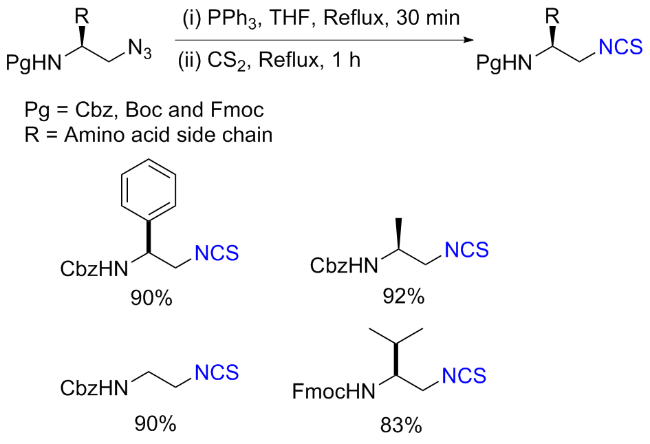
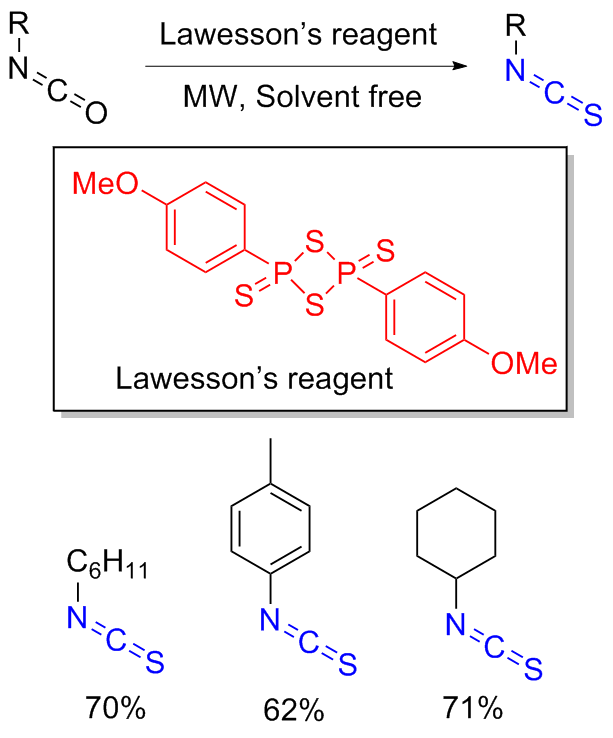
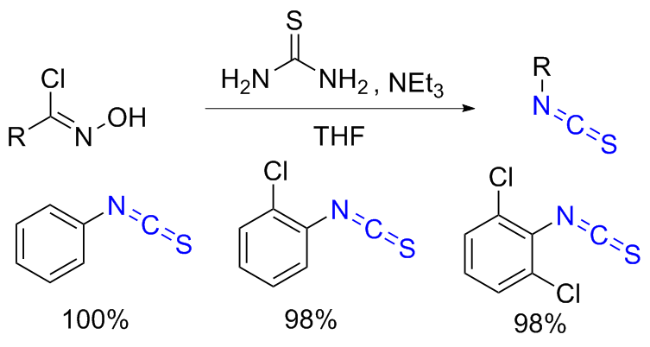
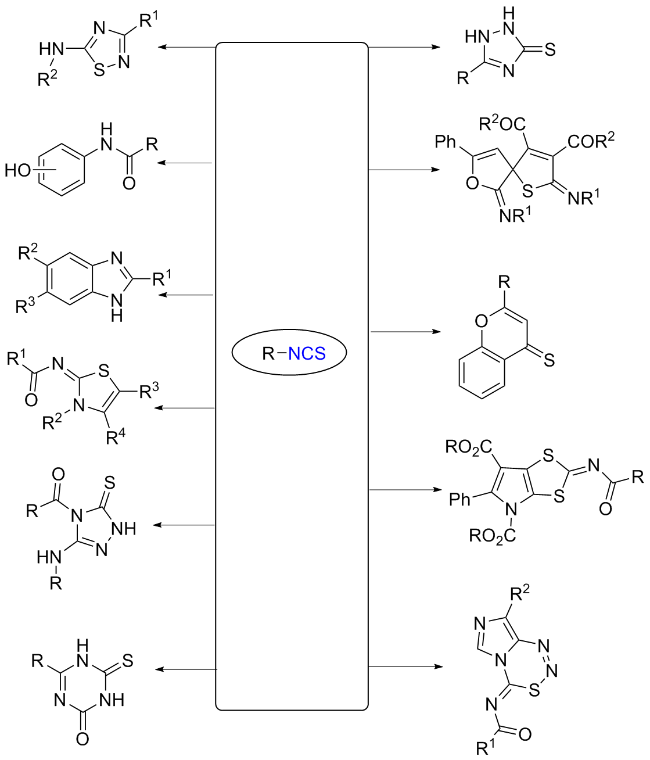
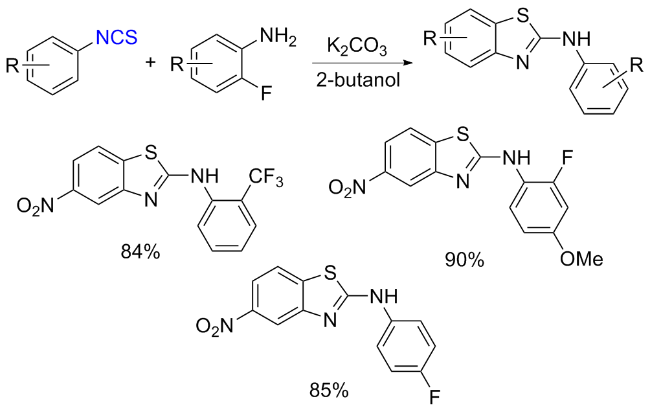
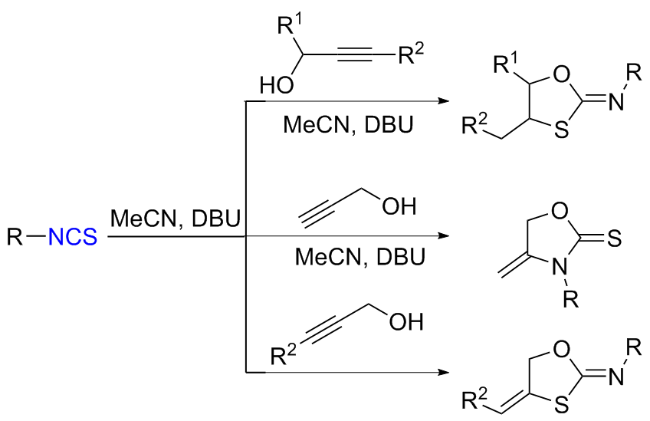
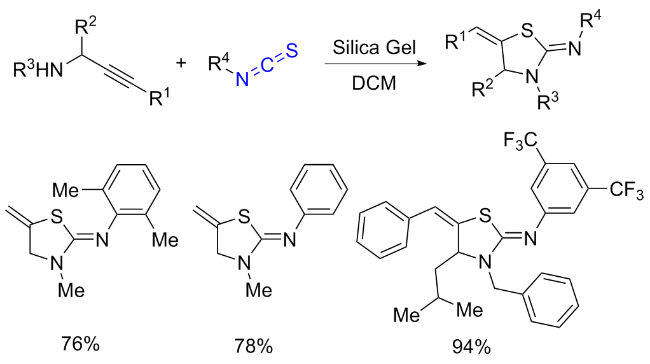
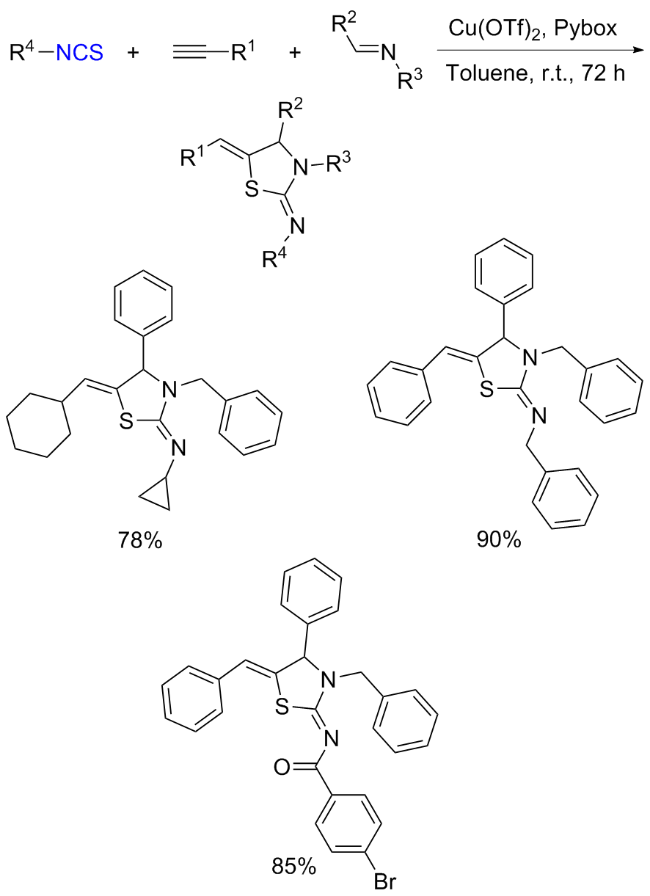
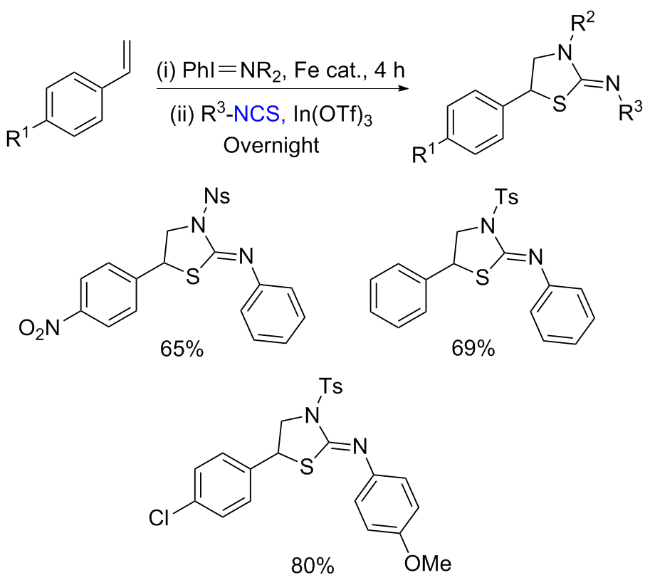
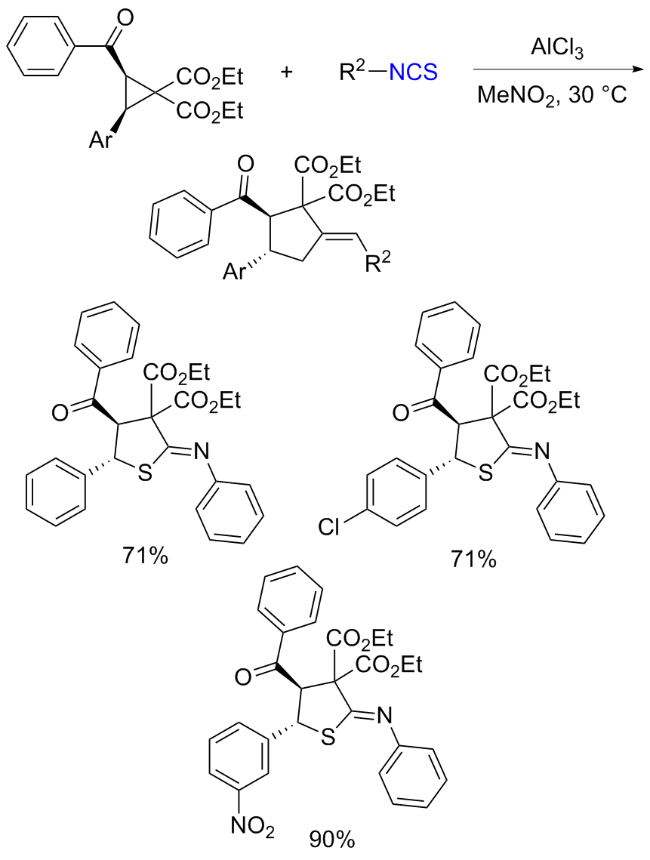
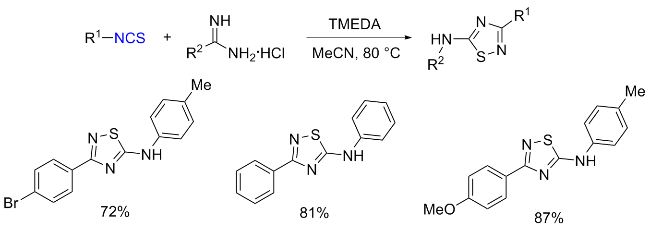
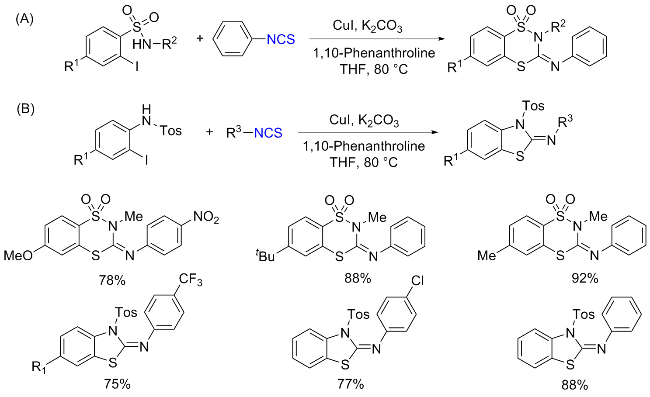
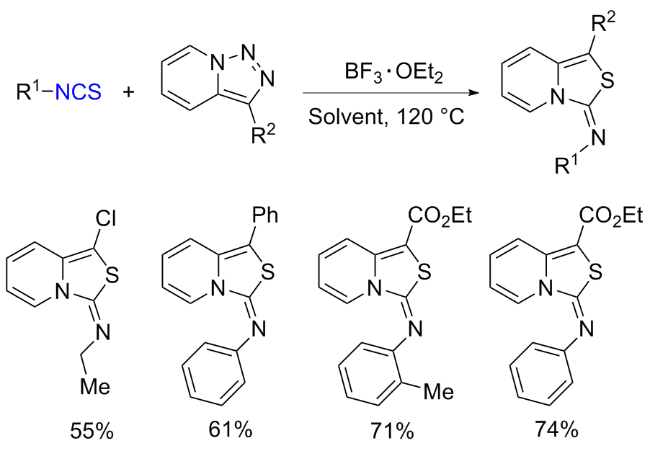
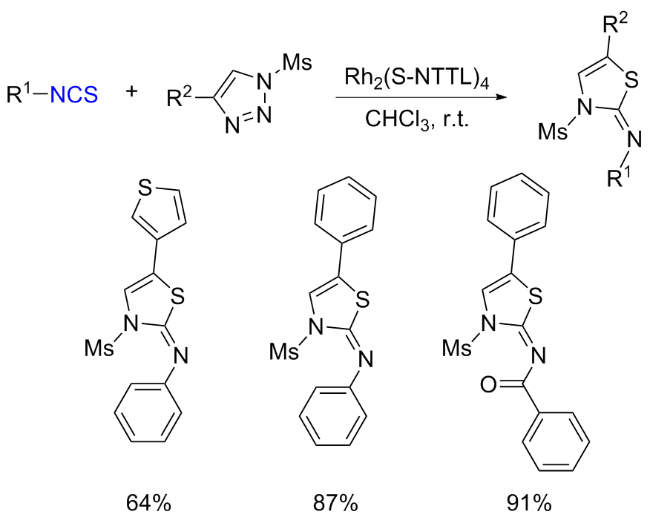
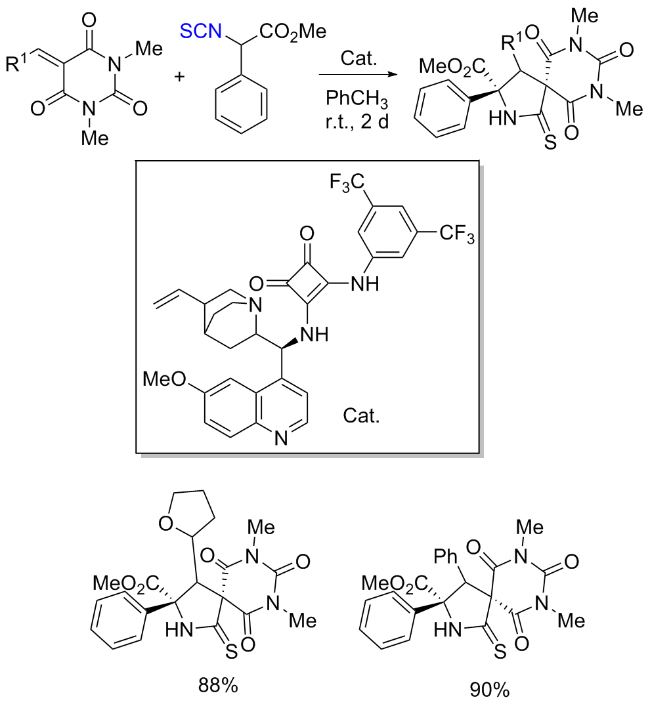
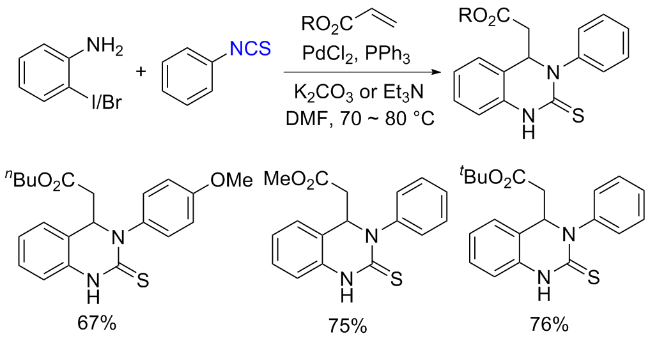
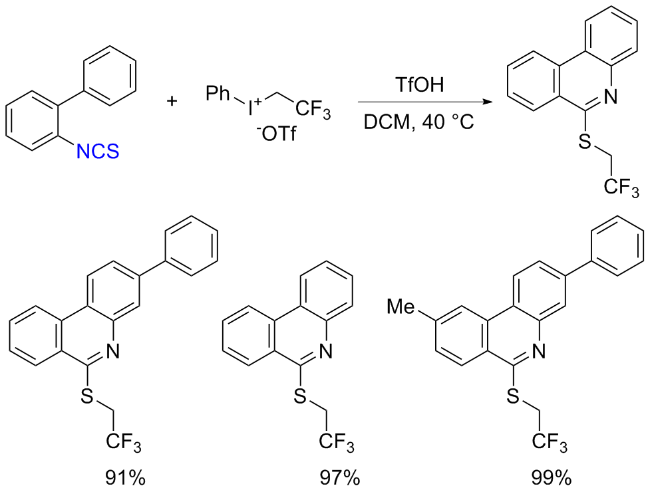
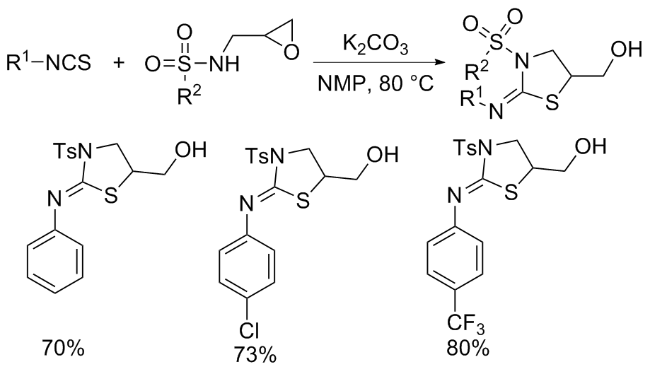
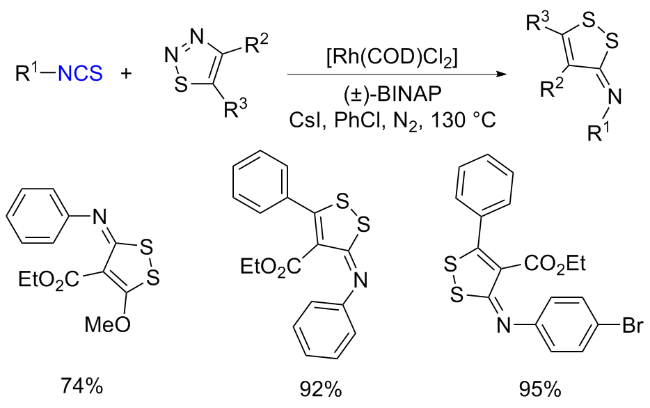
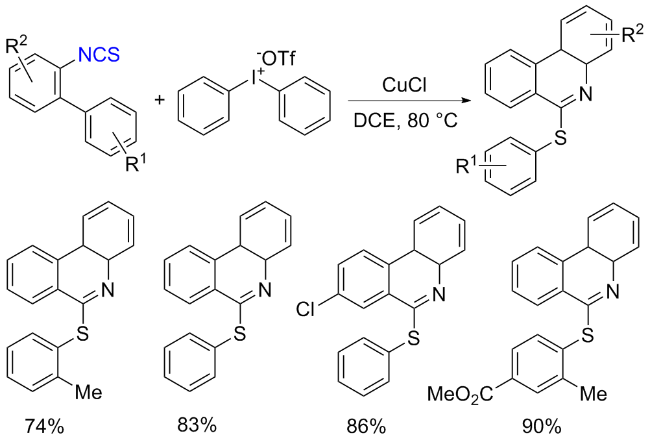
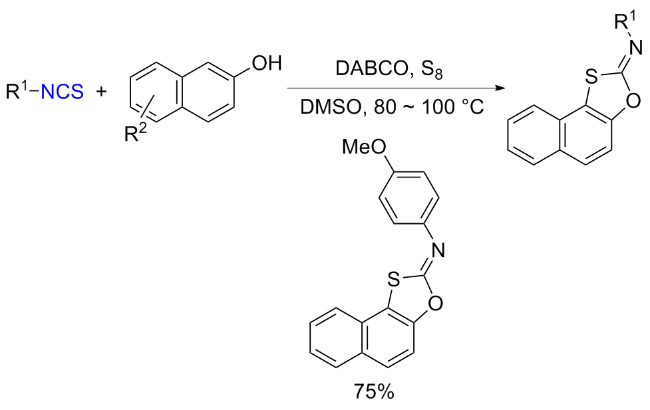
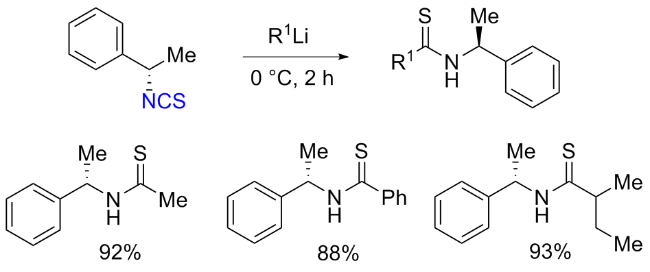
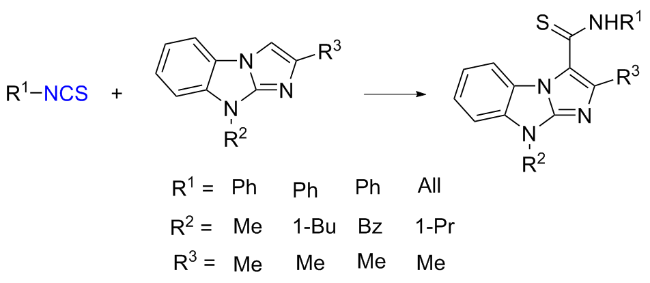
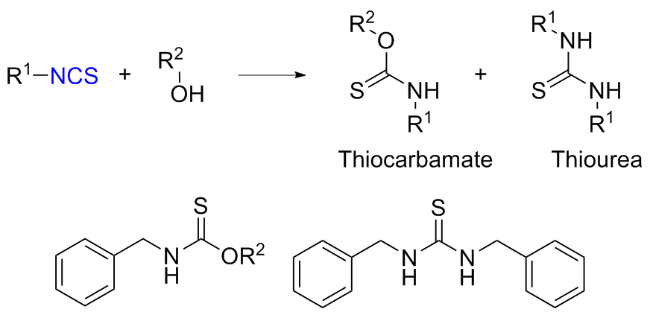
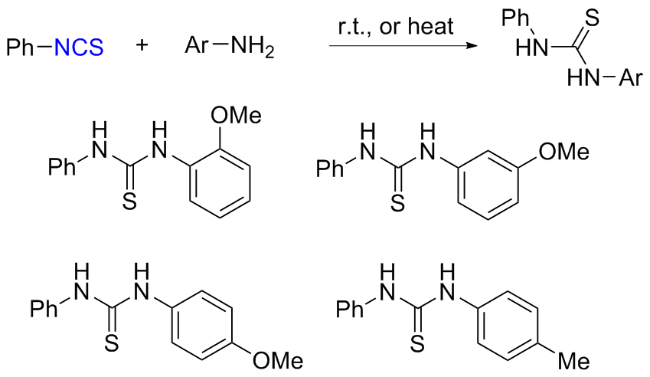
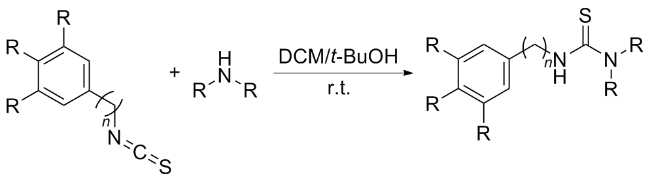
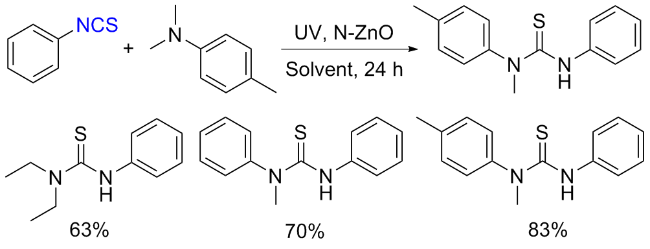
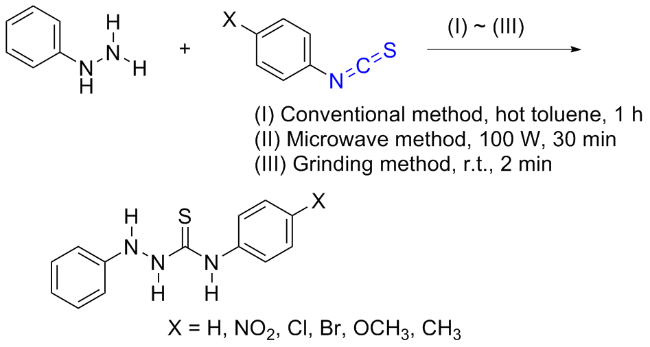
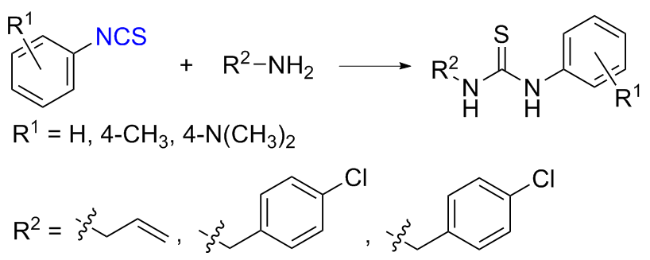
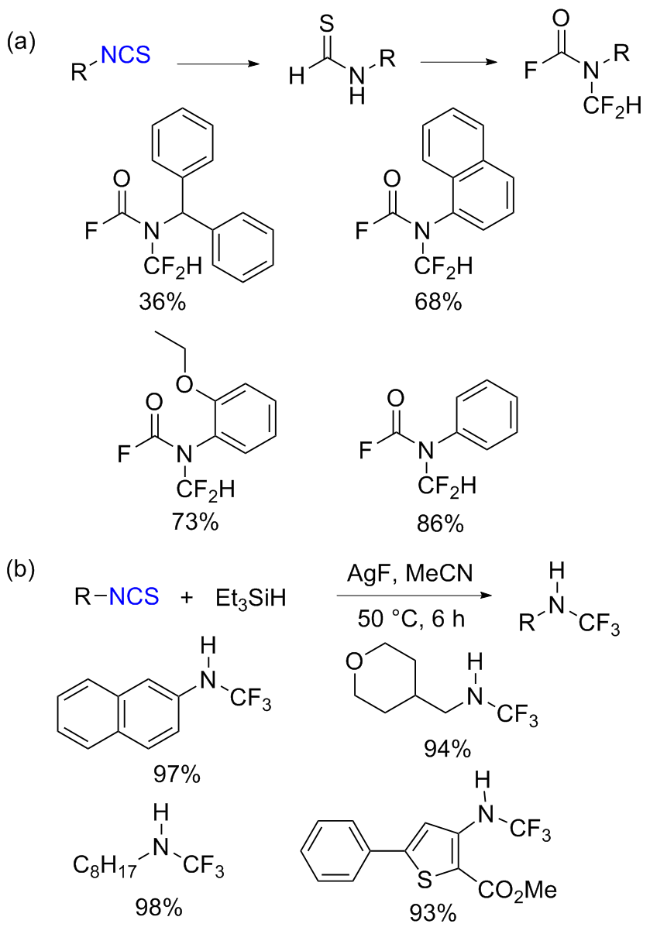
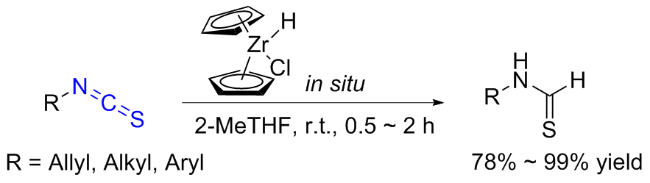The sulfuration of isocyanides directly leads to ITCs (
Scheme 11). Aromatic isocyanides react with sulfur to form isothiocyanates (ITCs) upon refluxing in benzene for 3 d with moderate yields obtained.
[36] On the other hand, aliphatic isocyanides do not undergo any reaction.
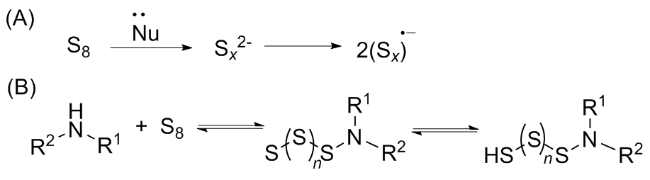
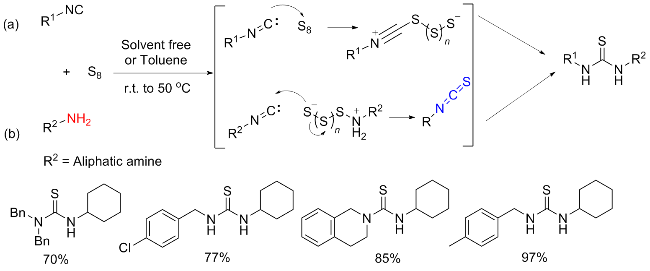
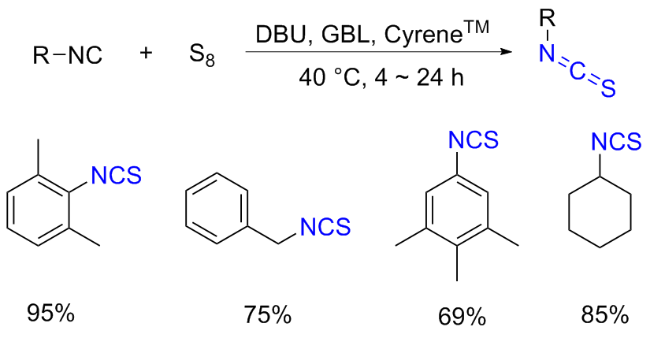
[37] This has led researchers to discover that catalysis or other types of activation, particularly nucleophilic additives, are necessary for an efficient, valuable, and comprehensive methodology. The most effective substitute for integrating the sulphur atom into the product is elemental sulphur. Sulphur, which acts as an electrophile because of its vacant d-orbitals, is attacked nucleophilically by
in situ produced carbene functionalities (
a). Using this method, primary amines (
c) combine with thiocarbonyl surrogates (
b), which are often dihalogenides to produce ITCs (
d). Otherwise, ITCs are produced by reacting with sulphur under heat conditions or with external additions in isocyanides (
e), where the terminal carbon atom may function as a carbene (
Scheme 11a). Notably, it was also claimed that adding sulphur to formaldimines produced ITCs. However, this technique is hardly ever employed nowadays. The idea that a switched mechanism also exists, involving a nucleophilic sulphur anion (S
x-) and the carbene of the isocyanide (
e) acting as an electrophile, is supported by the easy activation of sulphur by nucleophilic additives, such as aliphatic amines and hydroxyl, sulphide, and cyanide anions, and the correspondingly milder conditions compared to thermal activation (
Scheme 11b).