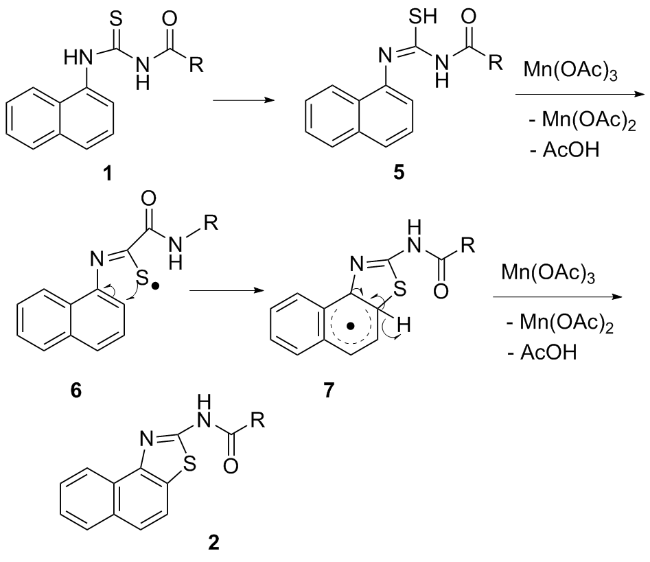
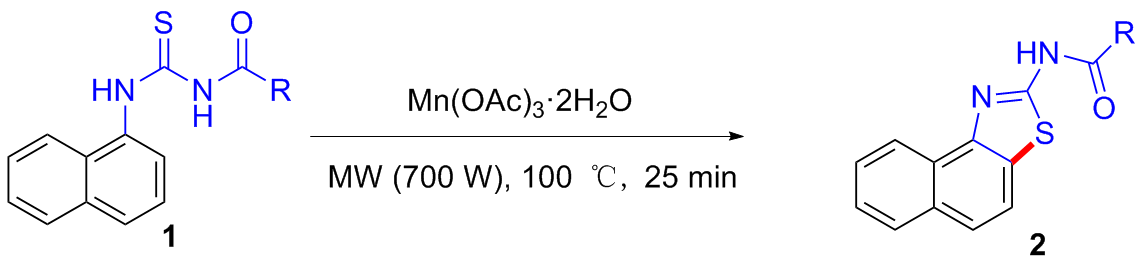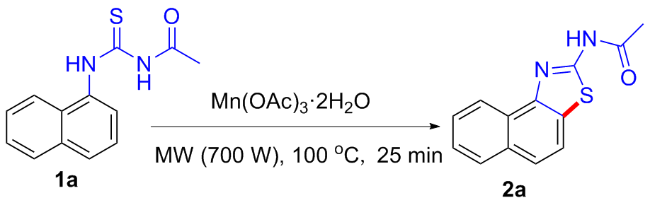将1a (300 mg, 1.23 mmol)、醋酸锰(III) (825 mg, 3.08 mmol)和冰醋酸(15 mL)依次加入到50 mL圆底烧瓶中后, 放入微波合成仪, 设定反应参数(温度100 ℃, 功率700 W, 时间25 min), 打开微波反应开关. 25 min后, 将圆底烧瓶从微波合成仪取出, 冷至室温, 然后将去离子水(15 mL)加入到反应液中, 立即有大量灰白色固体出现. 用二氯甲烷(15 mL×3)萃取, 合并有机相, 用无水硫酸钠干燥, 过滤, 滤液在真空下减压除去溶剂得灰白色固体. 粗产品用石油醚/二氯甲烷(V∶V=4∶2)的混合液为洗脱剂进行柱层析, 得260 mg白色固体2a. 化合物2b~2o以相同方法进行合成.
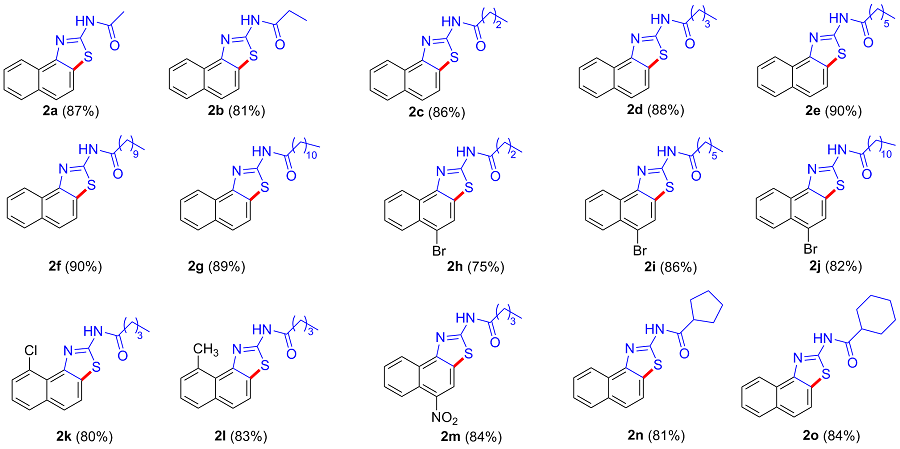
2-乙酰氨基萘并[1,2-
d]噻唑(
2a)
[16]: 白色固体, 260 mg, 产率87%. m.p. 280~282 ℃;
1H NMR (400 MHz, CDCl
3)
δ: 9.74 (s, 1H), 8.57 (d,
J=8.00 Hz, 1H), 7.95 (d,
J=8.40 Hz, 1H), 7.86 (d,
J=8.80 Hz, 1H), 7.77 (d,
J=8.80 Hz, 1H), 7.65~7.61 (m, 1H), 7.58~7.55 (m, 1H), 2.29 (s, 3H);
13C NMR (100 MHz, DMSO-
d6)
δ: 169.7, 158.5, 144.7, 132.3, 128.7, 127.6, 127.3, 127.0, 126.2, 124.2, 123.2, 120.0, 23.2; HRMS (ESI) calcd for C
13H
11N
2OS [M+H]
+ 243.0592, found 243.0612.
2-丙酰氨基萘并[1,2-d]噻唑(2b): 白色固体, 271 mg, 产率81%. m.p. 154~155 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.52 (s, 1H), 8.57 (d, J=8.00 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.80 Hz, 1H), 7.78 (d, J=8.80 Hz, 1H), 7.65~7.61 (m, 1H), 7.58~7.54 (m, 1H), 2.41(q, J=7.60 Hz, 2H), 1.18(t, J=7.60 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 172.6, 159.1, 144.0, 132.4, 128.5, 127.9, 127.1, 126.7, 126.0, 124.7, 122.9, 119.1, 29.5, 8.9; HRMS (ESI) calcd for C14H13N2OS [M+H]+ 257.0748, found 257.0741.
2-丁酰氨基萘并[1,2-d]噻唑(2c): 白色固体, 286 mg, 产率86%. m.p. 139~140 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.75 (s, 1H), 8.59 (d, J=8.40 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.80 Hz, 1H), 7.78 (d, J=8.80 Hz, 1H), 7.65~7.61 (m, 1H), 7.59~7.55 (m, 1H), 2.29 (t, J=7.40 Hz, 2H), 1.69~1.60 (m, 2H), 0.75 (t, J=7.40 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.9, 159.1, 144.0, 132.3, 128.5, 127.9, 127.1, 126.7, 126.0, 124.7, 123.0, 119.0, 38.2, 18.5, 13.3; HRMS (ESI) calcd for C15H15N2OS [M+H]+ 271.0905, found 271.0900.
2-戊酰氨基萘并[1,2-d]噻唑(2d): 白色固体, 308 mg, 产率88%. m.p. 80~82 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.35 (s, 1H), 8.59 (d, J=8.00 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.40 Hz, 1H), 7.78 (d, J=8.80 Hz, 1H), 7.65~7.55 (m, 2H), 2.36 (t, J=7.60 Hz, 2H), 1.67~1.60 (m, 2H), 1.24~1.14 (m, 2H), 0.78 (t, J=7.40 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.5, 158.2, 144.2, 132.3, 128.4, 127.9, 127.3, 126.7, 126.0, 124.6, 123.0, 119.0, 36.2, 27.0, 22.1, 13.5; HRMS (ESI) calcd for C16H17N2OS [M+H]+ 285.1061, found 285.1062.
2-庚酰氨基萘并[1,2-d]噻唑(2e): 白色固体, 331 mg, 产率90%. m.p. 99~100 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.73 (s, 1H), 8.59 (d, J=8.00 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.80 Hz, 1H), 7.78 (d, J=8.40 Hz, 1H), 7.65~7.55 (m, 2H), 2.31 (t, J=7.60 Hz, 2H), 1.61~1.58 (m, 2H), 1.26~1.06 (m, 6H), 0.80 (t, J=7.20 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.9, 159.0, 144.0, 132.3, 128.5, 127.9, 127.2, 126.7, 126.0, 124.7, 123.0, 119.0, 36.4, 31.1, 28.6, 25.0, 22.4, 13.9; HRMS (ESI) calcd for C18H21N2OS [M+H]+ 313.1394, found 313.1387.
2-十一烷酰氨基萘并[1,2-d]噻唑(2f): 白色固体, 409 mg, 产率90%. m.p. 76~77 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.80 (s, 1H), 8.59 (d, J=8.00 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.40 Hz, 1H), 7.78 (d, J=8.80 Hz, 1H), 7.65~7.54 (m, 2H), 2.29 (t, J=7.40 Hz, 2H), 1.73~1.70 (m, 2H), 1.29~1.05 (m, 14H), 0.88 (t, J=7.00 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 172.0, 159.2, 144.0, 132.4, 128.5, 127.9, 127.1, 126.7, 126.0, 124.7, 123.0, 119.0, 36.4, 31.9, 29.5, 29.4, 29.3, 29.0, 28.9, 25.0, 22.7, 14.2; HRMS (ESI) calcd for C22H29N2OS [M+H]+ 369.2000, found 369.1998.
2-十二烷酰氨基萘并[1,2-d]噻唑(2g): 白色固体, 419 mg, 产率89%. m.p. 80~81 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.56 (s, 1H), 8.59 (d, J=8.40 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.40 Hz, 1H), 7.78 (d, J=8.80 Hz, 1H), 7.65~7.61 (m, 1H), 7.59~7.55 (m, 1H), 2.34~2.30 (m, 2H), 1.62~1.61 (m, 2H), 1.32~1.08 (m, 16H), 0.88 (t, J=6.80 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.9, 159.1, 144.0, 132.3, 128.5, 127.9, 127.1, 126.7, 126.0, 124.7, 123.0, 119.0, 36.4, 31.9, 29.6, 29.5, 29.3, 28.9, 28.9, 25.0, 22.7, 14.1; HRMS (ESI) calcd for C23H31N2OS [M+H]+ 383.2157, found 383.2137.
2-丁酰氨基-5-溴萘并[1,2-d]噻唑(2h): 白色固体, 322 mg, 产率75%. m.p. 200~202 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 12.63 (s, 1H), 8.57 (d, J=7.60 Hz, 1H), 8.52 (s, 1H), 8.23 (d, J=6.00 Hz, 1H), 7.77~7.75 (m, 2H), 2.53 (t, J=7.20 Hz, 2H), 1.71~1.63 (m, 2H), 0.94 (t, J=7.20 Hz, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 172.7, 159.2, 144.9, 130.0, 128.1, 128.0, 127.9, 127.8, 127.5, 123.9, 116.5, 37.4, 18.6, 14.0; HRMS (ESI) calcd for C15H14BrN2OS [M+H]+ 349.0010, found 349.0008.
2-庚酰氨基-5-溴萘并[1,2-d]噻唑(2i): 白色固体, 400 mg, 产率86%. m.p. 136~138 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.79 (s, 1H), 8.61~8.59 (m, 1H), 8.35~8.33 (m, 1H), 8.18 (s, 1H), 7.69~7.66 (m, 2H), 2.43~2.39 (m, 2H), 1.71~1.64 (m, 2H), 1.28~1.17 (m, 6H), 0.85 (t, J=7.00 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.5, 158.4, 144.1, 130.5, 128.1, 128.0, 127.4, 127.2, 123.5, 122.6, 118.3, 36.5, 31.2, 28.9, 24.9, 22.4, 14.0; HRMS (ESI) calcd for C18H20BrN2OS [M+H]+ 391.0479, found 391.0472.
2-十二烷酰氨基-5-溴萘并[1,2-d]噻唑(2j): 白色固体, 465 mg, 产率82%. m.p. 100~101 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 12.62 (s, 1H), 8.59~8.56 (m, 1H), 8.53 (s, 1H), 8.25~8.22 (m, 1H), 7.79~7.73 (m, 2H), 1.66~1.63 (m, 2H), 1.29~1.22 (m, 18H), 0.82 (t, J=6.60 Hz, 3H); HRMS (ESI) calcd for C23H30BrN2OS [M+H]+ 461.1262, found 461.1255. 2j溶解度较差, 达不到13C NMR的测试浓度.
2-戊酰氨基-9-氯萘并[1,2-d]噻唑(2k): 313 mg, 浅黄色固体, 产率80%. m.p. 160~161 ℃; 1H NMR (400 MHz, DMSO-d6) δ 12.38 (s, 1H), 8.18 (d, J=8.40 Hz, 1H), 8.04 (d, J=8.00 Hz, 1H), 7.90 (d, J=8.40 Hz, 1H), 7.74 (d, J=7.60 Hz, 1H), 7.54 (t, J=7.80 Hz, 1H), 2.57 (t, J=7.40 Hz, 2H), 1.68~1.61 (m, 2H), 1.40~1.31 (m, 2H), 0.92 (t, J=7.40 Hz, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 173.0, 156.7, 142.9, 134.8, 130.9, 129.5, 129.2, 128.7, 126.3, 125.0, 124.3, 121.2, 35.2, 27.3, 22.2, 14.2; HRMS (ESI) calcd for C16H16ClN2OS [M+H]+ 319.0672, found 319.0669.
9-甲基-2-戊酰氨基萘并[1,2-d]噻唑(2l): 304 mg, 白色固体, 产率83%. m.p. 162~164 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.20 (s, 1H), 7.85 (d, J=8.40 Hz, 1H), 7.80~7.78 (m, 1H), 7.75 (d, J=8.80 Hz, 1H), 7.45~7.40 (m, 2H), 3.16 (s, 3H), 2.43 (t, J=7.60 Hz, 2H), 1.75~1.67 (m, 2H), 1.38~1.29 (m, 2H), 0.89 (t, J=7.20 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.0, 155.0, 144.8, 135.1, 133.6, 129.7, 129.0, 126.9, 126.8, 125.6, 125.5, 118.8, 36.4, 27.1, 24.8, 22.3, 13.7; HRMS (ESI) calcd for C17H19N2OS [M+H]+ 299.1218, found 299.1215.
2-戊酰氨基-5-硝基萘并[1,2-d]噻唑(2m): 340 mg, 黄色固体, 产率84%. m.p. 182~184 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 12.91 (s, 1H), 9.12 (s, 1H), 8.67~8.65 (m, 1H), 8.58~8.56 (m, 1H), 7.85~7.80 (m, 2H), 2.59 (t, J=7.40 Hz, 2H), 1.69~1.62 (m, 2H), 1.41~1.32 (m, 2H), 0.92 (t, J=7.40 Hz, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 173.3, 163.6, 149.7, 141.8, 129.3, 128.2, 126.9, 125.5, 124.2, 124.1, 123.8, 121.1, 35.3, 27.0, 22.2, 14.1; HRMS (ESI) calcd for C16H16N3O3S [M+H]+ 330.0912, found 330.0908.
2-环戊基甲酰氨基萘并[1,2-d]噻唑(2n): 白色固体, 296 mg, 产率81%. m.p. 170~172 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.06 (s, 1H), 8.59 (d, J=8.00 Hz, 1H), 7.95 (d, J=8.40 Hz, 1H), 7.87 (d, J=8.40 Hz, 1H), 7.77 (d, J=8.80 Hz, 1H), 7.65~7.60 (m, 1H), 7.58~7.54 (m, 1H), 2.73~2.67 (m, 1H), 1.90~1.79 (m, 4H), 1.73~1.69 (m, 2H), 1.45~1.44 (m, 2H); 13C NMR (100 MHz, CDCl3) δ: 175.1, 158.8, 144.1, 132.3, 128.4, 127.9, 127.2, 126.7, 126.0, 124.6, 123.0, 119.0, 45.5, 30.3, 25.8; HRMS (ESI) calcd for C17H17N2OS [M+H]+ 297.1061, found 297.1060.
2-环己基甲酰氨基萘并[1,2-
d]噻唑(
2o)
[16]: 白色固体, 321 mg, 产率84%. m.p. 154~156 ℃;
1H NMR (400 MHz, CDCl
3)
δ: 10.36 (s, 1H), 8.63 (d,
J=8.00 Hz, 1H), 7.97 (d,
J=8.00 Hz, 1H), 7.88 (d,
J=8.80 Hz, 1H), 7.78 (d,
J=8.80 Hz, 1H), 7.66~7.62 (m, 1H), 7.59~7.55 (m, 1H), 2.24~2.12 (m, 1H), 1.70~1.25 (m, 7H), 1.09~ 0.71 (m, 3H);
13C NMR (100 MHz, CDCl
3)
δ: 174.9, 159.3, 144.0, 132.3, 128.5, 127.9, 127.2, 126.7, 126.1, 124.7, 123.0, 119.1, 44.9, 29.1, 25.2, 24.8; HRMS (ESI) calcd for C
18H
19N
2OS [M+H]
+ 311.1218, found 311.1211.
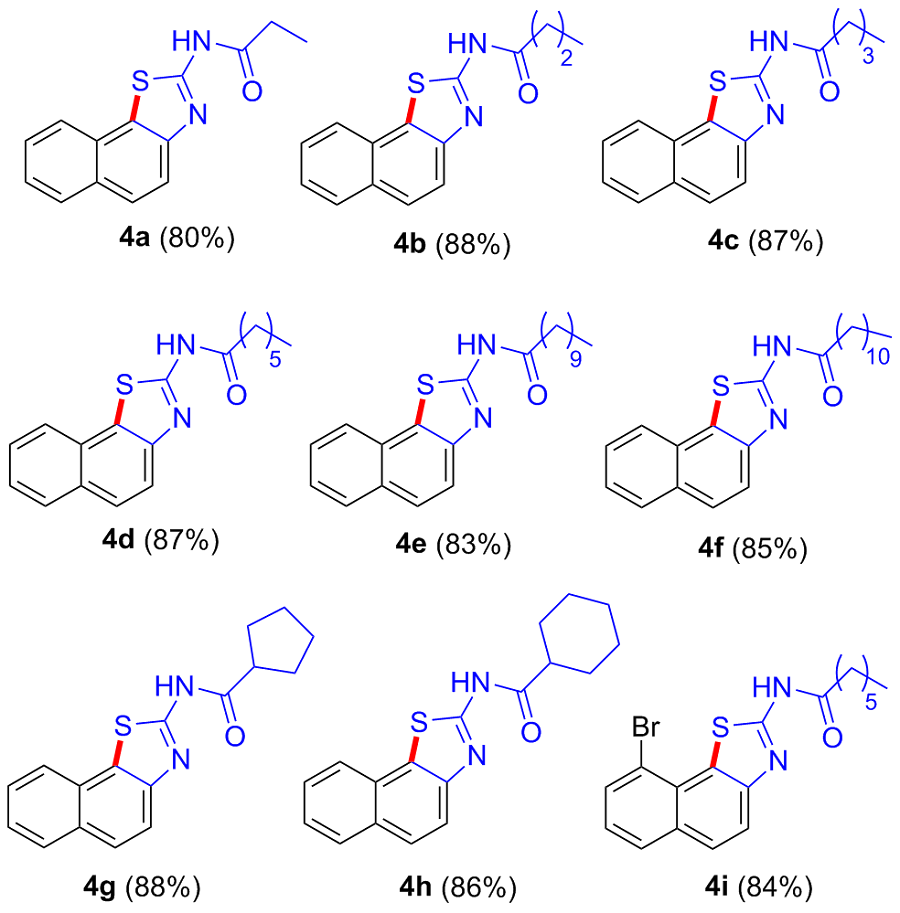
将3a (300 mg, 1.16 mmol)、醋酸锰(III)(777 mg, 2.90 mmol)和冰醋酸(15 mL)依次加入到50 mL圆底烧瓶中后, 放入微波合成仪, 然后按照3.2.1的步骤进行反应和后处理, 得238 mg白色固体4a. 化合物4b~4i以相同方法进行合成.
2-丙酰氨基萘并[2,1-d]噻唑(4a): 白色固体, 238 mg, 产率80%. m.p. 206~208 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.77 (s, 1H), 8.04 (d, J=8.40 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.80 Hz, 1H), 7.84 (d, J=8.80 Hz, 1H), 7.64~7.60 (m, 1H), 7.55~7.51 (m, 1H), 2.57 (q, J=7.60 Hz, 2H), 1.29 (t, J=7.60 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 172.3, 158.7, 145.8, 130.5, 129.0, 128.3, 127.9, 127.5, 127.2, 125.5, 124.1, 119.7, 29.7, 9.1; HRMS (ESI) calcd for C14H13N2OS [M+H]+ 257.0748, found 257.0744.
2-丁酰氨基萘并[2,1-
d]噻唑(
4b)
[12]: 白色固体, 276 mg, 产率88%. m.p. 181~183 ℃;
1H NMR (400 MHz, CDCl
3)
δ: 11.04 (s, 1H), 8.04 (d,
J=8.00 Hz, 1H), 7.96 (d,
J=8.00 Hz, 1H), 7.88 (d,
J=8.80 Hz, 1H), 7.85 (d,
J=8.80 Hz, 1H), 7.64~7.60 (m, 1H), 7.55~7.51 (m, 1H), 2.51 (t,
J=7.40 Hz, 2H), 1.84~1.75 (m, 2H), 0.97 (t,
J=7.40 Hz, 3H);
13C NMR (100 MHz, CDCl
3)
δ: 171.8, 159.1, 145.7, 130.5, 129.0, 128.3, 127.8, 127.5, 127.3, 125.5, 124.1, 119.6, 38.4, 18.6, 13.7.
2-戊酰氨基萘并[2,1-d]噻唑(4c): 白色固体, 287 mg, 产率87%. m.p. 154~155 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.22 (s, 1H), 8.04 (d, J=8.40 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.80 Hz, 1H), 7.85 (d, J=8.80 Hz, 1H), 7.61 (t, J=7.00 Hz, 1H), 7.53 (t, J=7.00 Hz, 1H), 2.52 (t, J=7.60 Hz, 2H), 1.77~1.69 (m, 3H), 1.38~1.29 (m, 2H), 0.87 (t, J=7.30 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 172.0, 159.3, 145.7, 130.5, 129.0, 128.3, 127.8, 127.4, 127.2, 125.5, 124.1, 119.5, 36.3, 27.1, 22.3, 13.7; HRMS (ESI) calcd for C16H17N2OS [M+H]+ 285.1061, found 285.1062.
2-庚酰氨基萘并[2,1-d]噻唑(4d): 淡黄色固体, 301 mg, 产率87%. m.p. 124~125 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.28 (s, 1H), 8.04 (d, J=8.00 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.80 Hz, 1H), 7.85 (d, J=8.80 Hz, 1H), 7.64~7.59 (m, 1H), 7.55~7.51 (m, 1H), 2.50 (t, J=7.60 Hz, 2H), 1.75~1.68 (m, 2H), 1.31~1.20 (m, 6H), 0.80 (t, J=7.00 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 172.0, 159.2, 145.7, 130.5, 129.0, 128.3, 127.8, 127.5, 127.3, 125.5, 124.1, 119.6, 36.6, 31.4, 28.8, 25.1, 22.4, 13.9; HRMS (ESI) calcd for C18H21N2OS [M+H]+ 313.1394, found 313.1395.
2-十一烷酰氨基萘并[2,1-d]噻唑(4e): 白色固体, 355 mg, 产率83%. m.p. 127~129 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.99 (s, 1H), 8.04 (d, J=8.00 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.80 Hz, 1H), 7.85 (d, J=8.80 Hz, 1H), 7.64~7.59 (m, 1H), 7.55~7.51 (m, 1H), 2.51 (t, J=7.60 Hz, 2H), 1.76~1.69 (m, 2H), 1.29-1.19 (m, 14H), 0.87 (t, J=7.00 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.8, 158.7, 145.8, 130.5, 129.0, 128.3, 127.9, 127.4, 127.2, 125.5, 124.1, 119.7, 36.6, 31.9, 29.5, 29.4, 29.3, 29.2, 29.1, 25.1, 22.7, 14.1; HRMS (ESI) calcd for C22H29N2OS [M+H]+ 369.2000, found 369.1999.
2-十二烷酰氨基萘并[2,1-d]噻唑(4f): 淡黄色固体, 377 mg, 产率85%. m.p. 130~131 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.06 (s, 1H), 8.04 (d, J=8.00 Hz, 1H), 7.96 (d, J=8.00 Hz, 1H), 7.88 (d, J=8.80 Hz, 1H), 7.85 (d, J=8.80 Hz, 1H), 7.63~7.59 (m, 1H), 7.55~7.51 (m, 1H), 2.50 (t, J=7.60 Hz, 2H), 1.76~1.69 (m, 2H), 1.28~1.18 (m, 16H), 0.87 (t, J=7.00 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 171.7, 158.6, 145.8, 130.5, 129.0, 128.3, 127.9, 127.4, 127.2, 125.4, 124.1, 119.7, 36.6, 31.9, 29.6, 29.6, 29.4, 29.3, 29.2, 29.1, 25.1, 22.7, 14.1; HRMS (ESI) calcd for C23H31N2OS [M+H]+ 383.2157, found 383.2152.
2-环戊基甲酰氨基萘并[2,1-d]噻唑(4g): 白色固体, 303 mg, 产率88%. m.p. 137~139 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.56 (s, 1H), 8.03 (d, J=8.40 Hz, 1H), 7.95 (d, J=8.40 Hz, 1H), 7.87 (d, J=8.80 Hz, 1H), 7.84 (d, J=8.80 Hz, 1H), 7.60 (t, J=7.20 Hz, 1H), 7.52 (t, J=7.40 Hz, 1H), 2.85 (p, J=8.00 Hz, 1H), 2.00~1.87 (m, 4H), 1.81~1.71 (m, 2H), 1.57~1.46 (m, 2H); 13C NMR (100 MHz, CDCl3) δ: 174.7, 158.1, 146.0, 130.5, 129.0, 128.3, 128.0, 127.3, 127.1, 125.4, 124.0, 119.8, 45.7, 30.4, 26.0; HRMS (ESI) calcd for C17H17N2OS [M+H]+ 297.1061, found 297.1054.
2-环己基甲酰氨基萘并[2,1-d]噻唑(4h): 白色固体, 309 mg, 产率86%. m.p. 144~146 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.33 (s, 1H), 8.03 (d, J=8.40 Hz, 1H), 7.96 (d, J=8.40 Hz, 1H), 7.87 (d, J=8.80 Hz, 1H), 7.84 (d, J=8.40 Hz, 1H), 7.63~7.59 (m, 1H), 7.54~7.50 (m, 1H), 2.47~2.39 (m, 1H), 2.00~1.97 (m, 2H), 1.81~1.80 (m, 2H), 1.65~1.56 (m, 3H), 1.27~1.20 (m, 3H); 13C NMR (100 MHz, CDCl3) δ: 174.6, 158.6, 145.8, 130.5, 129.0, 128.3, 127.9, 127.4, 127.2, 125.4, 124.1, 119.8, 45.1, 29.3, 25.5, 25.3; HRMS (ESI) calcd for C18H19N2OS [M+H]+ 311.1218, found 311.1212.
2-庚酰氨基-9-溴萘并[2,1-d]噻唑(4i): 369 mg, 白色固体, 产率84%. m.p. 168~169 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 12.41 (s, 1H), 8.14 (d, J=7.60 Hz, 1H), 8.06 (d, J=8.80 Hz, 1H), 8.03 (dd, J=7.60, 0.80 Hz, 1H), 7.96 (d, J=8.40 Hz, 1H), 7.45 (t, J=7.80 Hz, 1H), 1.67 (t, J=7.00 Hz, 2H), 1.37~1.18 (m, 8H), 0.87 (t, J=6.60 Hz, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 172.7, 159.7, 149.2, 132.8, 132.5, 129.8, 128.5, 128.3, 126.0, 125.5, 122.2, 118.4, 35.4, 31.5, 28.7, 25.0, 22.4, 14.4; HRMS (ESI) calcd for C18H20BrN2OS [M+H]+ 391.0479, found 391.0477.