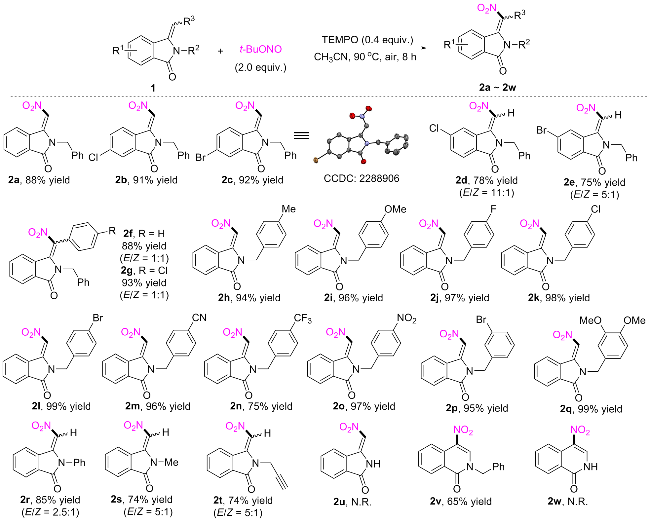
(E)-2-Benzyl-3-(nitromethylene)isoindolin-1-one (2a): Light yellow solid (123.2 mg, 88% yield). m.p. 102~104 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.74~8.67 (m, 1H), 7.98 (dt, J=6.7, 2.0 Hz, 1H), 7.77~7.67 (m, 2H), 7.38~7.33 (m, 2H), 7.33~7.28 (m, 1H), 7.23 (dd, J=7.0, 1.8 Hz, 2H), 7.13 (s, 1H), 5.00 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 166.8, 144.6, 134.4, 134.3, 133.0, 131.7, 129.7, 129.3, 128.8, 128.3, 126.9, 124.4, 121.3, 44.0. HRMS (ESI) calcd for C16H13N2O3 [M+H]+ 281.0921, found 281.0913.
(E)-2-Benzyl-6-chloro-3-(nitromethylene)isoindolin-1-one (2b): Light yellow solid (143.1 mg, 91% yield). m.p. 139~141 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.66 (d, J=8.5 Hz, 1H), 7.95 (d, J=2.1 Hz, 1H), 7.70 (dd, J=8.5, 2.1 Hz, 1H), 7.40~7.35 (m, 2H), 7.34~7.30 (m, 1H), 7.24~7.19 (m, 2H), 7.12 (s, 1H), 5.00 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 165.7, 143.8, 139.8, 134.22, 134.16, 131.6, 130.2, 129.9, 129.5, 128.5, 126.9, 124.7, 121.7, 44.3. HRMS (ESI) calcd for C16H12ClN2O3 [M+ H]+ 315.0531, 317.0501, found 315.0524, 317.0495
(E)-2-Benzyl-6-bromo-3-(nitromethylene)isoindolin-1- one (2c): Light yellow solid (164.6 mg, 92% yield). m.p. 149~151 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.58 (dd, J=8.5, 2.4 Hz, 1H), 8.11 (t, J=2.3 Hz, 1H), 7.86 (dt, J=8.6, 2.2 Hz, 1H), 7.40~7.35 (m, 2H), 7.34~7.29 (m, 1H), 7.24~7.20 (m, 2H), 7.13 (s, 1H), 4.99 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 165.6, 143.9, 137.2, 134.1, 131.6, 130.3, 130.2, 129.4, 128.5, 128.0, 127.7, 126.9, 121.8, 44.3. HRMS (ESI) calcd for C16H12BrN2O3 [M+H]+ 359.0026, 361.0005, found 359.0034, 361.0012.
2-Benzyl-5-chloro-3-(nitromethylene)isoindolin-1-one (2d): A mixture of the (E) and (Z) isomers, E/Z=11∶1. Light yellow solid (122.5 mg, 78% yield). m.p. 140~142 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.73 (d, J=1.7 Hz, 1H), 7.93~7.89 (m, 1+0.09H), 7.70 (dd, J=8.1, 1.7 Hz, 1H), 7.65 (dd, J=8.1, 1.7 Hz, 0.09H), 7.59 (s, 0.09H), 7.36 (dd, J=8.0, 6.3 Hz, 2H), 7.32 (t, J=7.2 Hz, 1H), 7.24 (d, J=5.0 Hz, 0.2H), 7.23~7.19 (m, 2H), 7.12 (s, 1H), 7.08 (s, 0.09H), 7.06~7.03 (m, 0.18H), 5.42 (s, 0.18H), 4.99 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 165.9, 143.4, 141.0, 134.1, 133.2, 133.1, 129.4, 129.2, 128.5, 128.0, 126.9, 125.4, 121.9, 44.3. HRMS (ESI) calcd for [M+H]+ C16H12ClN2O3 315.0531, 317.0501, found 315.0522, 317.0492.
2-Benzyl-5-bromo-3-(nitromethylene)isoindolin-1-one (2e): A mixture of the (E) and (Z) isomers, E/Z=5∶1. Light yellow solid (134.1 mg, 75% yield). m.p. 139~142 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.89 (d, J=1.5 Hz, 1H), 7.90~7.80 (m, 2+0.4H), 7.75 (d, J=5.0 Hz, 0.2H), 7.40~7.34 (m, 2H), 7.34~7.30 (m, 1H), 7.24 (t, J=1.5 Hz, 0.38H), 7.23~7.19 (m, 2H), 7.11 (s, 1H), 7.07 (s, 0.2H), 7.06~7.03 (m, 0.39H), 5.42 (s, 0.4H), 4.99 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 166.0, 143.4, 136.2, (135.7), (135.6), 134.1, 133.3, 132.0, 129.5, 129.0, 128.55, (128.47), (128.0), 127.2, 126.9, (126.3), 125.5, (124.0), 121.9, (46.3), 44.3. HRMS (ESI) calcd for C16H12BrN2O3 [M+H]+ 359.0026, 361.0005, found 359.0034, 361.0012.
2-Benzyl-3-(nitro(phenyl)methylene)isoindolin-1-one (2f): A mixture of the (E) and (Z) isomers, E/Z=1∶1. Yellow solid (156.6 mg, 88% yield). m.p. 131~134 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.02~7.97 (m, 1H), 7.95 (d, J=7.5 Hz, 1.17H), 7.68~7.62 (m, 3H), 7.56~7.48 (m, 2.47H), 7.47~7.41 (m, 3.43H), 7.31~7.20 (m, 9H), 7.19~7.09 (m, 7.34H), 6.53 (d, J=7.1 Hz, 2H), 6.11 (d, J=8.0 Hz, 1.15H), 5.24 (s, 2.20H), 4.68 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 168.8, 167.9, 137.0, 136.5, 135.68, 135.67, 135.1, 134.2, 133.7, 133.5, 133.4, 133.0, 132.1, 131.41, 131.39, 131.3, 131.1, 130.9, 130.8, 129.9, 129.7, 128.9, 128.7, 128.6, 128.53, 128.48, 128.1, 127.3, 125.8, 125.0, 124.6, 124.3, 123.4, 46.0, 45.3. HRMS (ESI) calcd for C22H17N2O3 [M+H]+ 357.1234, found 357.1244.
2-Benzyl-3-((4-chlorophenyl)(nitro)methylene)isoin-dolin-1-one (2g): A mixture of the (E) and (Z) isomers, E/Z=1∶1. Yellow solid (181.3 mg, 93% yield). m.p. 123~126 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.02~7.98 (m, 0.83H), 7.96 (d, J=7.5 Hz, 1H), 7.69~7.61 (m, 2.6H), 7.53 (t, J=7.5 Hz, 1H), 7.44~7.39 (m, 2H), 7.33~7.24 (m, 2+1.66H), 7.24~7.19 (m, 2H), 7.19~7.12 (m, 2+1.66+0.86H), 7.11~7.05 (m, 2+1.67H), 6.56 (d, J=6.9 Hz, 1.66H), 6.22 (d, J=8.0 Hz, 1H), 5.23 (s, 2H), 4.69 (s, 1.66H); 13C NMR (126 MHz, Chloroform-d) δ: 168.7, 167.9, 137.4, 137.3, 137.2, 136.8, 135.5, 135.4, 135.1, 133.8, 133.7, 133.4, 133.2, 132.9, 132.2, 132.0, 131.6, 131.5, 130.5, 130.1, 128.9, 128.8, 128.61, 128.59, 128.5, 128.20, 128.15, 127.5, 127.3, 125.6, 124.9, 124.8, 124.4, 123.5, 46.1, 45.4. HRMS (ESI) calcd for C22H16ClN2O3 [M+H]+ 391.0844, 393.0814, found 391.0851, 393.0822.
(E)-2-(4-Methylbenzyl)-3-(nitromethylene)isoindolin-1-one (2h). Light yellow solid (138.2 mg, 94% yield). m.p. 137~139 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.71 (dd, J=7.2, 1.2 Hz, 1H), 8.02~7.96 (m, 1H), 7.77~7.69 (m, 2H), 7.18~7.10 (m, 5H), 4.96 (s, 2H), 2.33 (s, 3H); 13C NMR (126 MHz, Chloroform-d) δ: 166.9, 144.7, 138.2, 134.3, 133.0, 132.4, 131.8, 131.4, 130.0, 129.9, 128.9, 127.0, 124.5, 121.4, 43.9, 21.2. HRMS (ESI) calcd for C17H15N2O3 [M+H]+ 295.1077, found 295.1086.
(E)-2-(4-Methoxybenzyl)-3-(nitromethylene)isoindolin-1-one (2i): Light yellow solid (148.5 mg, 96% yield). m.p. 148~149 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.71 (d, J=7.8 Hz, 1H), 7.98 (dd, J=7.4, 1.4 Hz, 1H), 7.77~7.68 (m, 2H), 7.20~7.14 (m, 3H), 6.91~6.85 (m, 2H), 4.94 (s, 2H), 3.79 (s, 3H); 13C NMR (126 MHz, Chloroform-d) δ: 166.9, 159.6, 144.7, 134.3, 133.0, 131.8, 129.9, 128.9, 128.4, 126.4, 124.4, 121.4, 114.7, 55.5, 43.6. HRMS (ESI) calcd for C17H15N2O4 [M+H]+ 311.1026, found 311.1018.
(E)-2-(4-Fluorobenzyl)-3-(nitromethylene)isoindolin-1-one (2j): Light yellow solid (144.5 mg, 97% yield). m.p. 169~171 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.71 (dd, J=7.4, 1.2 Hz, 1H), 7.99 (dd, J=7.0, 1.5 Hz, 1H), 7.79~7.70 (m, 2H), 7.26~7.19 (m, 2H), 7.10 (s, 1H), 7.09~7.01 (m, 2H), 4.97 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 166.8, 162.6 (d, JC-F=248.2 Hz), 144.5, 134.4, 133.1, 131.7, 130.3 (d, JC-F=2.5 Hz), 129.7, 128.9, 128.8 (d, JC-F=8.8 Hz), 124.5, 121.3, 116.4 (d, JC-F=21.4 Hz), 43.4. HRMS (ESI) calcd for C16H11FN2O3Na [M+Na]+ 321.0646, found 321.0638.
(E)-2-(4-Chlorobenzyl)-3-(nitromethylene)isoindolin-1-one (2k): Light yellow solid (153.6 mg, 98% yield). m.p. 145~147 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.74~8.68 (m, 1H), 7.99 (dd, J=7.5, 1.3 Hz, 1H), 7.79~7.70 (m, 2H), 7.36~7.30 (m, 2H), 7.20~7.15 (m, 2H), 7.07 (s, 1H), 4.96 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 166.8, 144.5, 134.5, 134.4, 133.2, 133.0, 131.7, 129.7, 129.6, 128.9, 128.3, 124.6, 121.3, 43.4. HRMS (ESI) calcd for C16H11ClN2O3Na [M+Na]+ 337.0350, 339.0321, found 337.0341, 339.0313.
(E)-2-(4-Bromobenzyl)-3-(nitromethylene)isoindolin-1-one (2l): Light yellow solid (177.2 mg, 99% yield). m.p. 134~135 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 8.50 (d, J=7.9 Hz, 1H), 8.01~7.95 (m, 1H), 7.90~7.80 (m, 2H), 7.62 (s, 1H), 7.55 (dd, J=8.1, 2.4 Hz, 2H), 7.27 (d, J=8.1 Hz, 2H), 5.04 (s, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 166.1, 143.9, 134.9, 134.3, 133.3, 131.7, 131.1, 129.3, 129.2, 127.7, 124.1, 121.5, 120.8, 42.1. HRMS (ESI) calcd for C16H11BrN2O3Na [M+Na]+ 380.9845, 382.9825, found 380.9837, 382.9817.
(E)-4-((1-(Nitromethylene)-3-oxoisoindolin-2-yl)meth- yl)benzonitrile (2m): Light yellow solid (146.4 mg, 96% yield). m.p. 186~188 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 8.53~8.49 (m, 1H), 8.00~7.96 (m, 1H), 7.91~7.80 (m, 4H), 7.63 (s, 1H), 7.49 (d, J=8.1 Hz, 2H), 5.17 (s, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 166.2, 144.0, 141.2, 134.3, 133.3, 132.7, 131.2, 129.3, 127.80, 127.77, 124.1, 121.6, 118.6, 110.4, 42.5. HRMS (ESI) calcd for [M+H]+ C17H12N3O3 306.0873, found 306.0863.
(E)-3-(Nitromethylene)-2-(4-(trifluoromethyl)benzyl)-isoindolin-1-one (2n): Light yellow solid (130.5 mg, 75% yield). m.p. 115~117 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.73 (d, J=7.8 Hz, 1H), 8.01 (dd, J=7.3, 1.4 Hz, 1H), 7.81~7.72 (m, 2H), 7.63 (d, J=8.0 Hz, 2H), 7.36 (d, J=8.0 Hz, 2H), 7.05 (s, 1H), 5.06 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 166.8, 144.4, 138.5, 134.6, 133.3, 131.7, 130.8 (q, JC-F=32.8 Hz), 129.6, 129.0, 127.3, 126.4 (q, JC-F=3.8 Hz), 126.2 (q, JC-F=270.2 Hz), 124.7, 121.3, 43.6. HRMS (ESI) calcd for C17H12F3N2O3
[M+H]+ 349.0795, found 349.0802.(E)-2-(4-Nitrobenzyl)-3-(nitromethylene)isoindolin-1-one (2o): Yellow solid (157.6 mg, 97% yield). m.p. 181~183 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.74 (dd, J=7.5, 1.2 Hz, 1H), 8.27~8.21 (m, 2H), 8.05~8.00 (m, 1H), 7.83~7.73 (m, 2H), 7.42 (d, J=8.7 Hz, 2H), 7.01 (s, 1H), 5.11 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 166.7, 148.0, 144.3, 141.7, 134.7, 133.4, 131.6, 129.4, 129.1, 127.7, 124.75, 124.66, 121.2, 43.4. HRMS (ESI) calcd for C16H12N3O5 [M+H]+ 326.0771, found 326.0760.
(E)-2-(3-Bromobenzyl)-3-(nitromethylene)isoindolin-1-one (2p): Light yellow solid (170.0 mg, 95% yield). m.p. 117~119 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.73 (dd, J=7.5, 1.2 Hz, 1H), 8.03~7.97 (m, 1H), 7.80~7.71 (m, 2H), 7.45 (dt, J=8.0, 1.4 Hz, 1H), 7.37 (t, J=1.8 Hz, 1H), 7.27~7.21 (m, 1H), 7.17 (dt, J=7.8, 1.3 Hz, 1H), 7.07 (s, 1H), 4.97 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 166.8, 144.5, 136.8, 134.5, 133.2, 131.70, 131.67, 130.9, 129.9, 129.6, 129.0, 125.5, 124.6, 123.6, 121.3, 43.4. HRMS (ESI) calcd for C16H12BrN2O3 [M+H]+ 359.0026, 361.005, found 359.0034, 361.0014.
(E)-2-(3,4-Dimethoxybenzyl)-3-(nitromethylene)isoin-dolin-1-one (2q): Yellow solid (168.3 mg, 99% yield). m.p. 148~150 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.75~8.69 (m, 1H), 8.02~7.97 (m, 1H), 7.79~7.69 (m, 2H), 7.20 (s, 1H), 6.85~6.78 (m, 2H), 6.76 (d, J=2.0 Hz, 1H), 4.93 (s, 2H), 3.87 (s, 3H), 3.84 (s, 3H); 13C NMR (126 MHz, Chloroform-d) δ: 167.0, 149.8, 149.2, 144.7, 134.4, 133.1, 131.8, 129.8, 128.9, 126.9, 124.5, 121.5, 119.6, 111.6, 110.3, 56.14, 56.09, 44.0. HRMS (ESI) calcd for C18H17N2O5 [M+H]+ 341.1132, found 341.1140.
3-(Nitromethylene)-2-phenylisoindolin-1-one (2r): A mixture of the (E) and (Z) isomers, E/Z=2.5∶1. Light yellow solid (113.0 mg, 85% yield). m.p. 133~135 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.82 (d, J=7.8 Hz, 1H), 8.04~7.98 (m, 1+0.4H), 7.85~7.70 (m, 2+3×0.4H), 7.62~7.52 (m, 2+3×0.4H), 7.47 (t, J=7.6 Hz, 0.76H), 7.40 (t, J=7.5 Hz, 0.4H), 7.32 (dd, J=7.2, 1.8 Hz, 2H), 7.27 (t, J=3.7 Hz, 1H), 6.94 (s, 1H); 13C NMR (126 MHz, Chloroform-d) δ: (167.8), 166.4, 146.9, (139.5), (136.1), (135.6), 134.4, (134.0), 133.4, (133.0), (132.8), 131.6, 130.4, 130.1, (129.9), 129.3, 128.9, 128.64, (128.58), (127.5), 126.0, (125.3), 124.6, 122.2, (120.6), (115.1). HRMS (ESI) calcd for C15H11N2O3 [M+H]+ 267.0764, found 267.0757.
2-Methyl-3-(nitromethylene)isoindolin-1-one (2s): A mixture of the (E) and (Z) isomers, E/Z=5∶1. Light yellow solid (75.5 mg, 74% yield). m.p. 167~169 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.74 (d, J=7.9 Hz, 1H), 7.95~7.88 (m, 1+0.19H), 7.74 (td, J=7.7, 1.4 Hz, 1H), 7.71~7.62 (m, 1+0.2×3H), 7.25 (s, 0.19H), 7.21 (s, 1H), 3.53 (s, 0.6H), 3.29 (s, 3H); 13C NMR (126 MHz, Chloro- form-d) δ: (168.7), 166.6, 146.2, (140.7), (136.2), 134.2, (133.6), 133.0, (132.4), 131.6, 130.2, 128.8, (127.7), (124.8), 124.2, 120.42, (120.39), (115.0), (31.0), 27.0. HRMS (ESI) calcd for [M+H]+ C10H9N2O3 205.0608, found 205.0616.
3-(Nitromethylene)-2-(prop-2-yn-1-yl)isoindolin-1-one (2t): A mixture of the (E) and (Z) isomers, E/Z=5∶1. Yellow solid (84.4 mg, 74% yield). m.p. 138~140 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.75 (d, J=7.9 Hz, 1H), 7.98~7.92 (m, 1H), 7.77 (td, J=7.7, 1.4 Hz, 1H), 7.74~7.66 (m, 1+3×0.2H), 7.46 (s, 1H), 7.36 (s, 0.18H), 5.03 (d, J=2.5 Hz, 0.39H), 4.60 (d, J=2.5 Hz, 2H), 2.39 (t, J=2.5 Hz, 1H), 2.26 (t, J=2.5 Hz, 0.16H); 13C NMR (126 MHz, Chloroform-d) δ: 165.5, 143.8, (138.0), (136.5), 134.5, (134.0), 133.1, (132.6), 131.7, 129.6, 129.0, (127.1), (125.2), 124.6, 121.5, (120.7), (116.3), 75.7, 74.4, (73.8), (32.9), 29.7. HRMS (ESI) calcd for C12H9N2O3 [M+H]+ 229.0608, found 229.0600.
2-Benzyl-4-nitroisoquinolin-1(2H)-one (2v): Light yellow solid (91.0 mg, 65% yield). m.p. 139~141 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 8.69 (d, J=8.4 Hz, 1H), 8.63 (s, 1H), 8.52 (d, J=8.1 Hz, 1H), 7.87~7.82 (m, 1H), 7.63 (t, J=7.6 Hz, 1H), 7.41~7.31 (m, 5H), 5.29 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 161.7, 136.9, 135.1, 134.5, 129.6, 129.4, 129.2, 129.1, 128.9, 128.8, 128.5, 124.6, 123.9, 53.0. HRMS (ESI) calcd for [M+H]+ C16H13N2O3 281.0921, found 281.0912.
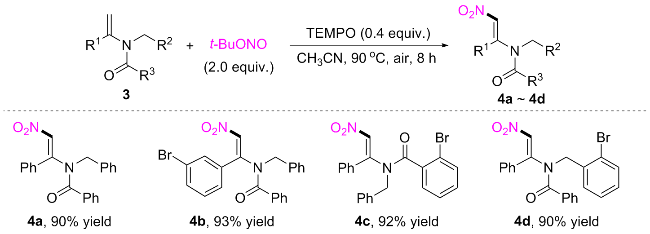
(E)-N-Benzyl-N-(2-nitro-1-phenylvinyl)benzamide (4a): Light yellow solid (161.2 mg, 90% yield). m.p. 179~181 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 7.69~7.63 (m, 2H), 7.59 (t, J=7.4 Hz, 1H), 7.52~7.46 (m, 4H), 7.44~7.38 (m, 2H), 7.32 (t, J=7.6 Hz, 2H), 7.28~7.23 (m, 3H), 7.18 (dd, J=7.1, 2.5 Hz, 2H), 4.93 (d, J=137.2 Hz, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 170.5, 148.5, 135.8, 134.4, 132.3, 131.9, 131.4, 131.1, 129.4, 129.2, 128.9, 128.2, 128.0, 127.7, 127.6, 51.5. HRMS (ESI) calcd forC22H19N2O3 [M+H]+ 359.1390, found 359.1382.
(E)-N-Benzyl-N-(1-(3-bromophenyl)-2-nitrovinyl)ben-zamide (4b): Light yellow solid (102.7 mg, 93% yield). m.p. 120~122 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 7.58 (dt, J=7.8, 1.5 Hz, 1H), 7.48~7.43 (m, 2H), 7.38~7.34 (m, 1H), 7.32 (t, J=1.9 Hz, 1H), 7.30~7.19 (m, 8H), 7.16 (dt, J=7.8, 1.4 Hz, 1H), 6.85 (s, 1H), 4.97 (s, 2H); 13C NMR (126 MHz, Chloroform-d) δ: 171.3, 147.8, 135.6, 135.5, 135.0, 134.8, 131.6, 131.4, 130.9, 129.7, 128.7, 128.5, 128.3, 127.9, 127.2, 123.5, 52.1. HRMS (ESI) calcd for C22H18BrN2O3 [M+H]+ 437.0495, 439.0475, found 437.0504, 439.0484.
(E)-N-Benzyl-2-bromo-N-(2-nitro-1-phenylvinyl)ben-zamide (4c): Light yellow solid (200.5 mg, 92% yield). m.p. 118~120 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 7.69 (s, 1H), 7.50 (dd, J=7.9, 1.2 Hz, 1H), 7.43~7.38 (m, 4H), 7.37~7.33 (m, 3H), 7.32~7.25 (m, 2H), 7.25~7.18 (m, 3H), 6.89 (d, J=7.4 Hz, 2H), 4.99 (s, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 168.9, 148.9, 137.1, 136.1, 134.7, 132.8, 132.1, 131.4, 130.6, 128.8, 128.6, 128.5, 128.4, 128.2, 128.0, 127.7, 119.2, 51.9. HRMS (ESI) calcd for C22H18BrN2O3 [M+H]+ 437.0495, 439.0475, found 437.0504, 439.0483.
(E)-N-(2-Bromobenzyl)-N-(2-nitro-1-phenylvinyl)ben-zamide (4d): Light yellow solid (196.2 mg, 90% yield). m.p. 119~121 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 7.75~7.68 (m, 3H), 7.58 (t, J=7.4 Hz, 1H), 7.55~7.51 (m, 2H), 7.51~7.47 (m, 3H), 7.47~7.41 (m, 2H), 7.36 (q, J=8.3 Hz, 3H), 7.19 (t, J=7.7 Hz, 1H), 4.97 (s, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 170.8, 148.7, 135.2, 134.0, 132.40, 132.37, 131.7, 131.6, 131.3, 130.9, 129.6, 129.33, 129.25, 128.1, 127.8, 127.7, 122.7, 51.7. HRMS (ESI) calcd for C22H18BrN2O3 [M+H]+ 437.0495, 439.0475; found 437.0503, 439.0482.
Supporting Information The
1H NMR,
13C NMR, and
19F NMR spectra of products
2a~
2w and
4a~
4d. The Supporting Information is available free of charge via the Internet at
http://sioc-journal.cn.