

硒催化环己烯氧化制备1,2-环己二醇进展
收稿日期: 2025-01-13
修回日期: 2025-02-13
网络出版日期: 2025-03-13
基金资助
江苏省自然科学基金(BK20191220)
江苏省高校优秀科技创新团队(2021)
江苏省国际合作(BZ2023038)
江苏省高等学校自然科学基金(19KJB210020)
Progress in Selenium Catalyzed Oxidation of Cyclohexene to Produce 1,2-Cyclohexanediol
Received date: 2025-01-13
Revised date: 2025-02-13
Online published: 2025-03-13
Supported by
Natural Science Foundation of Jiangsu Province(BK20191220)
Jiangsu Provincial Excellent Science and Technology Innovation Team(2021)
Jiangsu Province International Cooperation Project(BZ2023038)
Natural Science Foundation of the Jiangsu Higher Education Institutions(19KJB210020)
Copyright
雍达明 , 袁广树 , 刘永红 , 张旭 . 硒催化环己烯氧化制备1,2-环己二醇进展[J]. 有机化学, 2025 , 45(8) : 2767 -2772 . DOI: 10.6023/cjoc202501008
Cyclohexene is an easily available chemical raw material that can be prepared by selective hydrogenation of benzene. Compared with cyclohexane, the introduction of the alkene functional group makes cyclohexene more active and can be oxidized with better selectivity. Therefore, the oxidation reaction of cyclohexene is the key step for the synthesis of nylon-66 series products and their intermediates. In recent years, selenium catalysis technology has developed rapidly. Compared with metal catalysis, selenium is easily metabolized and ecologically safe, and its catalytic oxidation process usually uses hydrogen peroxide and molecular oxygen as clean oxidants, which are environmentally friendly. The selenium-catalyzed oxidation technology of cyclohexene is expected to be applied in the synthesis of nylon intermediates. The relevant progresses including both homogeneous and heterogeneous catalysis are reviewed, the principle from a scientific perspective is expounded and prospects for its industrial application are provided.
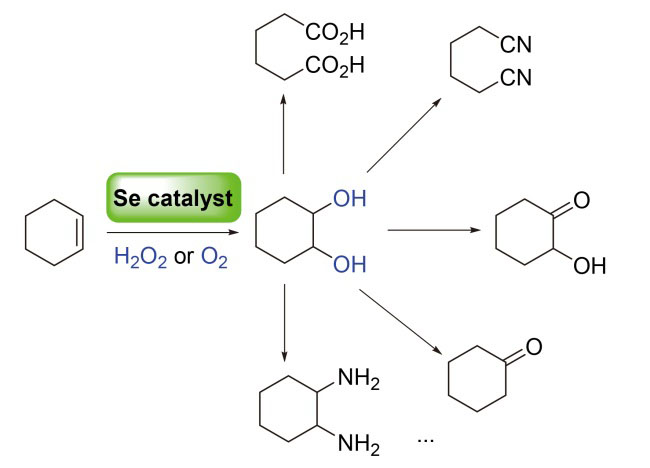
Key words: selenium; catalysis; oxidation; cyclohexene; nylon
| [1] |
(a)
(b)
(c)
|
| [2] |
|
| [3] |
(a)
(b)
|
| [4] |
(a)
(b)
(c)
(d)
(e)
|
| [5] |
(a)
(b)
(c)
(d)
(e)
(f)
(g)
(h)
|
| [6] |
(a)
(b)
(c)
(d)
(周文见, 肖欣蕊, 刘永红, 张旭, 有机化学, 2022, 42, 1849.)
(e)
|
| [7] |
(a)
(b)
(c)
(d)
|
| [8] |
(a)
(辛翠, 蒋俊, 邓紫微, 欧丽娟, 何卫民, 化学学报, 2024, 82, 1109.)
(b)
(c)
(d)
(e)
|
| [9] |
|
| [10] |
(a)
(景旋, 胡利如, 孙玉洁, 王余伟, 夏伟峰, 王金堂, 合成技术及应用, 2024, 39, 41.)
(b)
(郭颖, 王树霞, 黄珞玮, 司虎, 合成技术及应用, 2024, 39, 10.)
|
| [11] |
|
| [12] |
|
| [13] |
(俞磊, 王俊, 陈天, 丁克鸿, 潘毅, 有机化学, 2013, 33, 1096.)
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
(a)
(b)
(c)
(d)
|
| [21] |
(a)
(雍达明, 田杰, 杨瑞洪, 吴启超, 张旭, 有机化学, 2024, 44, 1343.)
(b)
(c)
(d)
(曹洪恩, 李培梓, 景崤壁, 周宏伟, 有机化学, 2022, 42, 3890.)
(e)
(f)
(g)
(h)
|
| [22] |
|
| [23] |
(黄杰军, 钱蓉蓉, 王爽, 曹洪恩, 有机化学, 2021, 41, 1639.)
|
| [24] |
(a)
(b)
(c)
|
| [25] |
(a)
(b)
(c)
(d)
(e)
|
| [26] |
(a)
(b)
|
| [27] |
(a)
(孟祥坤, 祁正源, 俞磊, 张亦旸, 有机化学, 2023, 43, 112.)
(b)
|
| [28] |
(a)
(b)
|
| [29] |
(a)
(b)
(c)
(d)
|
| [30] |
(a)
(b)
(c)
(d)
(刘峰, 詹杰, 孙扬阳, 景崤壁, 有机化学, 2021, 41, 2099.)
|
| [31] |
(a)
(b)
(b)
(c)
(d)
(e)
|
/
| 〈 |
|
〉 |