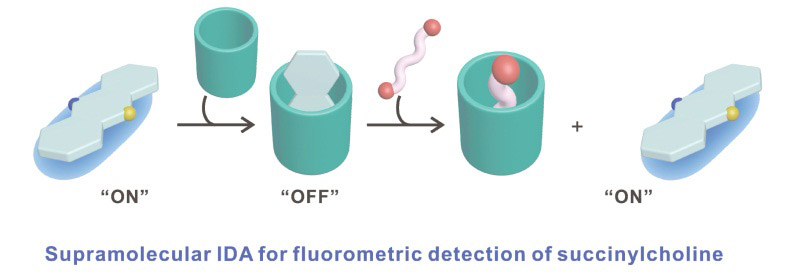
To develop an efficient detection method for
SC, it is imperative to identify a host capable of high-affinity specificity toward
SC.
[34-36] SC is highly hydrophilic as its oil- water partition coefficient (
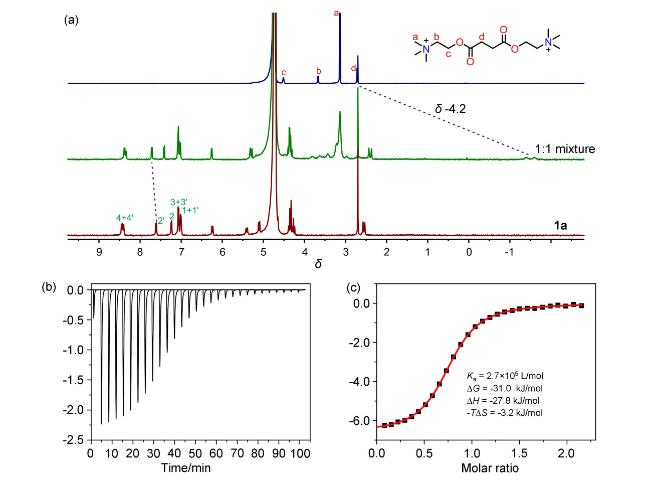
Kow) is less than 0.01 at pH 7.4. The recognition of hydrophilic molecules in aqueous environments presents a considerable challenge in molecular recognition. In recent years, our research has concentrated on the aqueous phase molecular recognition and application of amide naphthotubes, which feature amide nitrogen- hydrogen bonds buried in their cavities.
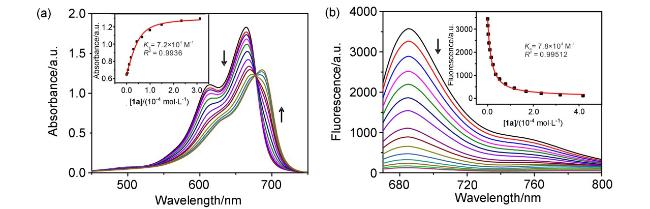
[37] By harnessing hydrogen bonding interactions shielded by the hydrophobic cavity, we have achieved selective recognition of hydrophilic molecules.
[38] This approach has led to applications across various domains, including optical sensing,
[39] drug reversal,
[40] biorthogonal chemistry,
[41] and pollutant adsorption removal.
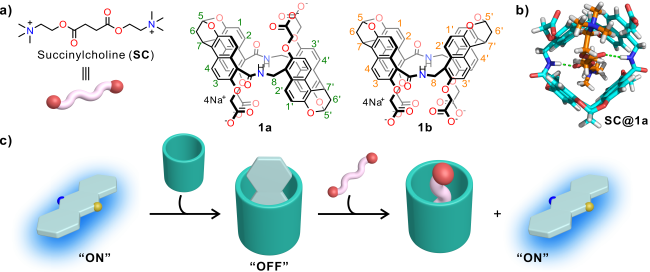
[42] Structurally, anti-amide naphthotube (
1a,
Figure 1a) represents ideal candidates for recognizing
SC (
Figure 1b): the elongated linear structure of
SC aligns well with the binding preferences exhibited by the hosts. The central region of
SC linear structure possesses relatively hydrophobic characteristics that complement the deep hydrophobic cavity of the amide naphthotube. Moreover, its ester bonds form complementary double hydrogen bonds with the amide groups present in the host cavity. Additionally, the water-soluble quaternary ammonium salts at two
SC ends enhance binding through electrostatic attraction to the peripheral carboxylate ions of
1a. Therefore, in this study, we exploited the specific binding between amide naphthotube and
SC to construct a fluorescence-based analytical method employing the IDA strategy for rapid and accurate detection of
SC (
Figure 1c). This methodology successfully facilitated the precise quantification of
SC in pharmaceutical formulations and plasma samples.