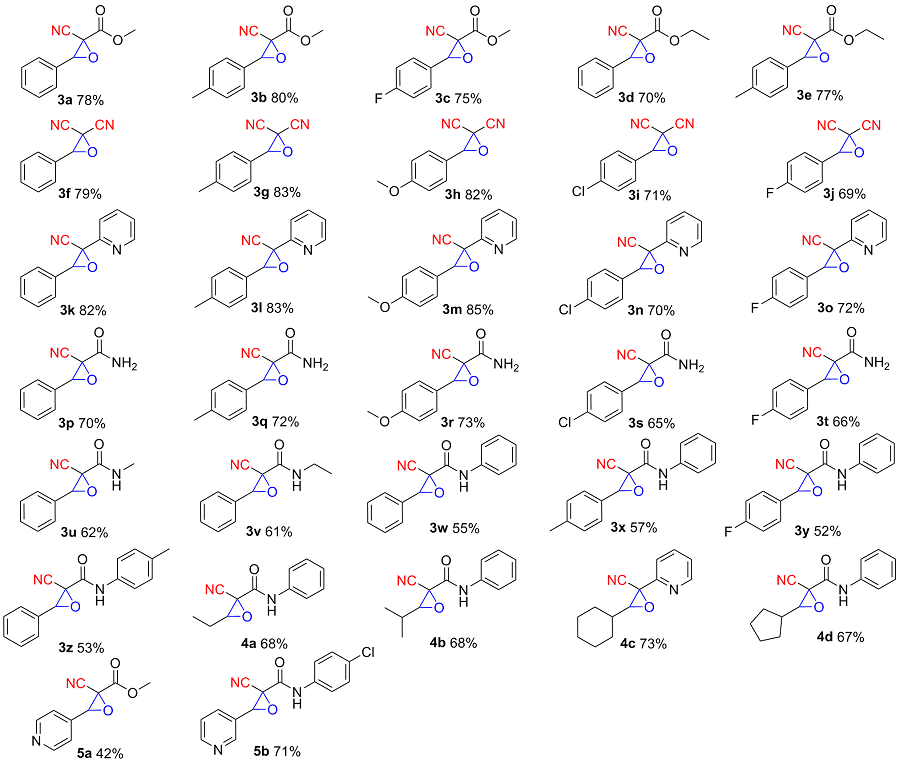
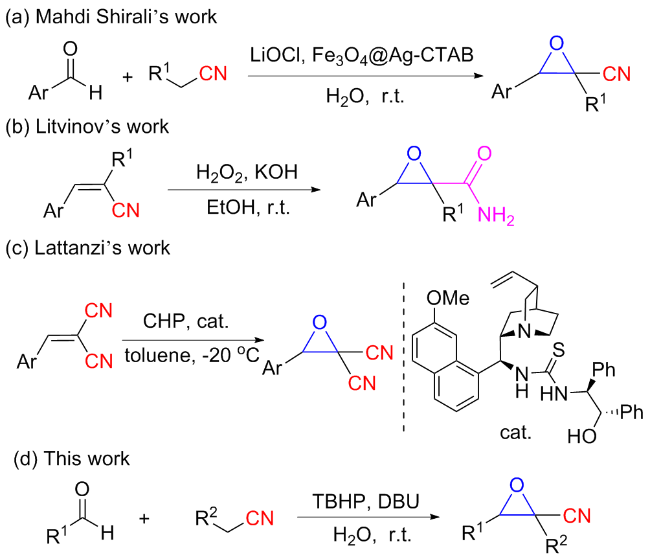
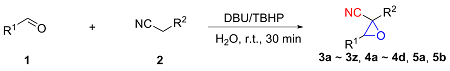
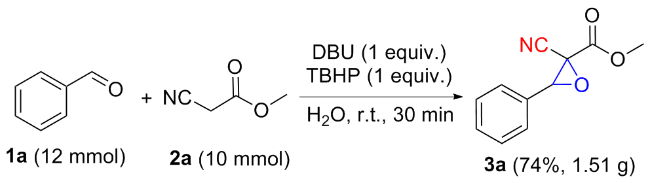
以2-氰基-3-苯基环氧乙烷-2-羧酸甲酯(3a)的合成为例. 在10 mL干燥的圆底烧瓶中加入磁力搅拌子, 依次加入H2O (5 mL)、DBU (1 mmol)、苯甲醛(1.2 mmol)、氰基乙酸甲酯(1 mmol)和叔丁醇过氧化氢(1 mmol), 在室温条件下搅拌反应30 min, 薄层色谱(TLC)监测至反应完成. 向反应混合物中依次加入乙酸乙酯萃取(30 mL×3), 再用水进行洗涤(30 mL×3), 收集有机相后减压除去溶剂, 粗产物以石油醚/乙酸乙酯(V∶V=15∶1)为洗脱液进行柱层析纯化, 得到2-氰基-3-苯基环氧乙烷-2-羧酸甲酯(3a), 白色固体, 产率78% (159 mg). m.p. 77.3~78.9 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.05~7.99 (m, 1H), 7.83 (s, 1H), 7.46~7.41 (m, 2H), 7.07~7.02 (m, 2H), 4.71 (s, 1H), 3.80 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 164.10, 160.92, 128.59, 123.05, 115.00, 114.55, 63.63, 56.11, 55.72; HRMS (TOF ES+) calcd for C11H10NO3 [M+H]+ 204.0655, found 204.0661.
2-氰基-3-(对甲苯基)环氧乙烷-2-羧酸甲酯(3b): 白色固体, 产率80% (174 mg). m.p. 85.8~86.6 ℃; 1H NMR (DMSO-d6, 400 MHz) δ: 7.39~7.35 (m, 2H), 7.30 (d, J=8.1 Hz, 2H), 4.78 (s, 1H), 3.86 (s, 3H), 2.35 (s, 3H); 13C NMR (DMSO-d6, 100 MHz) δ: 163.32, 140.17, 129.57, 128.00, 127.20, 114.34, 64.32, 54.55, 54.01, 21.36; HRMS (TOF ES+) calcd for C12H12NO3 [M+H]+ 218.0812, found 218.0816.
2-氰基-3-(4-氟苯基)环氧乙烷-2-羧酸甲酯(3c): 白色固体, 产率75% (166 mg). m.p. 88.2~89.7 ℃; 1H NMR (DMSO-d6, 400 MHz) δ: 7.54 (dd, J=8.7, 5.5 Hz, 2H), 7.34 (t, J=8.8 Hz, 2H), 4.85 (s, 1H), 3.86 (s, 3H); 13C NMR (DMSO-d6, 100 MHz) δ: 163.45 (d, J=245 Hz), 163.18, 129.67 (d, J=9 Hz), 127.38 (d, J=3 Hz), 116.09 (d, J=22 Hz), 114.25, 63.75, 54.55, 53.99; HRMS (TOF ES+) calcd for C11H9NO3 [M+H]+ 222.0561, found 222.0568.
2-氰基-3-苯基环氧乙烷-2-羧酸乙酯(3d): 无色油状, 产率70% (153 mg). 1H NMR (DMSO-d6, 400 MHz) δ: 8.40 (s, 1H), 8.05 (d, J=7.4 Hz, 2H), 7.61 (dt, J=14.6, 6.8 Hz, 3H), 4.32 (q, J=7.1 Hz, 2H), 1.31 (t, J=7.1 Hz, 3H); 13C NMR (DMSO-d6, 100 MHz) δ: 162.25, 155.53, 133.86, 131.82, 131.26, 129.78, 116.03, 103.08, 62.84, 14.43; HRMS (TOF ES+) calcd for C12H12NO3 [M+H]+ 218.0812, found 218.0817.
2-氰基-3-(对甲苯基)环氧乙烷-2-羧酸乙酯(3e): 白色固体, 产率77% (179 mg). m.p. 162~164 ℃; 1H NMR (DMSO-d6, 400 MHz) δ: 7.38 (d, J=8.2 Hz, 2H), 7.29 (d, J=8.0 Hz, 2H), 4.79 (s, 1H), 4.31 (qd, J=7.1, 1.3 Hz, 2H), 2.35 (s, 3H), 1.30 (t, J=7.1 Hz, 3H); 13C NMR (DMSO-d6, 100 MHz) δ: 162.25, 155.53, 133.86, 131.82, 131.26, 129.78, 116.03, 103.08, 62.84, 14.43; HRMS (TOF ES+) calcd for C13H14NO3 [M+H]+ 232.0968, found 232.0961.
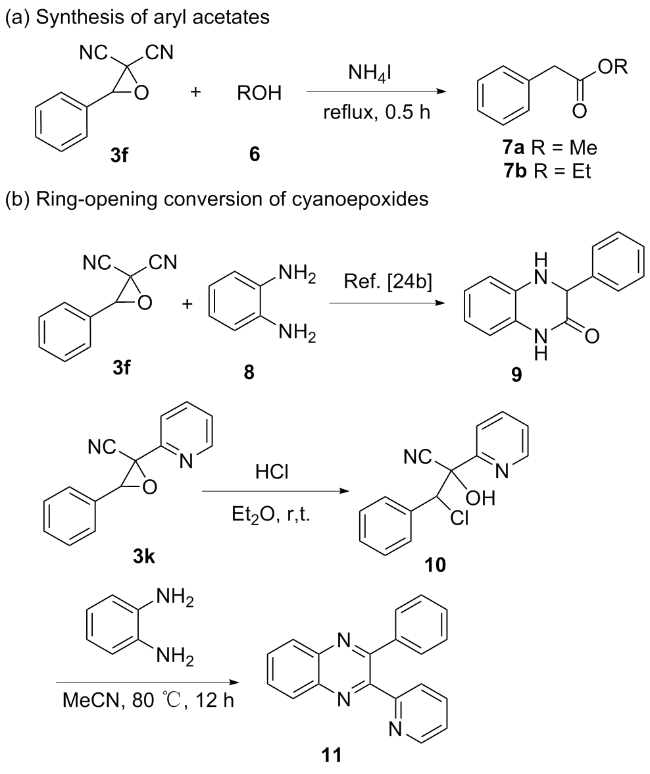
3-苯基环氧乙烷-2,2-二腈(3f): 白色固体, 产率79% (135 mg). m.p. 50.8~52.3 ℃; 1H NMR (DMSO-d6, 400 MHz) δ: 7.85~7.78 (m, 2H), 7.56~7.47 (m, 3H), 5.32 (d, J=1.1 Hz, 1H); 13C NMR (DMSO-d6, 100 MHz) δ: 131.01, 130.60, 129.98, 127.95, 112.04, 110.47, 42.82, 24.19; HRMS (TOF ES+) calcd for C10H7N2O [M+H]+ 171.0553, found 171.0553.
3-(对甲苯基)环氧乙烷-2,2-二腈(3g): 白色固体, 产率83% (153 mg). m.p. 78.0~79.2 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.69 (d, J=8.2 Hz, 2H), 7.31 (d, J=8.3 Hz, 2H), 5.24 (s, 1H), 2.34 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 140.19, 129.97, 129.89, 124.35, 111.50, 109.93, 42.24, 23.62, 21.28; HRMS (TOF ES+) calcd for C11H9N2O [M+H]+ 185.0709, found 185.0702.
3-(4-甲氧基苯基)环氧乙烷-2,2-二腈(3h): 白色固体, 产率82% (164 mg). m.p. 85.6~86.4 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.39 (s, 1H), 8.02~7.93 (m, 2H), 7.24~7.15 (m, 2H), 3.89 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 164.83, 160.91, 133.85, 124.58, 115.67, 115.29, 114.39, 77.31, 56.40, 56.38; HRMS (TOF ES+) calcd for C11H9N2O2 [M+H]+ 201.0659, found 201.0653.
3-(4-氯苯基)环氧乙烷-2,2-二腈(3i): 白色固体, 产率71% (145 mg). m.p. 127.0~129.2 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.55 (s, 1H), 7.96 (d, J=8.7 Hz, 2H), 7.75~7.67 (m, 2H); 13C NMR (100 MHz, DMSO-d6) δ: 160.58, 139.51, 132.61, 130.54, 130.20, 114.52, 113.48, 82.71; HRMS (TOF ES+) calcd for C10H6ClN2O [M+H]+ 205.0163, found 205.0169.
3-(4-氟苯基)环氧乙烷-2,2-二腈(3j): 白色固体, 产率69% (130 mg). m.p. 78.3~80.9 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.55 (s, 1H), 8.10~7.98 (m, 2H), 7.56~7.45 (m, 2H); 13C NMR (100 MHz, DMSO-d6) δ: 166.54 (d, J=255 Hz), 160.63, 134.04 (d, J=10 Hz) 132.72, 132.63, 128.51 (d, J=3 Hz), 117.41 (d, J=22 Hz), 116.59, 116.37, 114.63, 113.64, 111.44, 109.92, 81.64, 81.62, 23.83; HRMS (TOF ES+) calcd for C10H6FN2O [M+H]+ 189.0459, found 189.0453.
3-苯基-2-(吡啶-2-基)环氧乙烷-2-甲腈(3k): 无色油状物, 产率82% (183 mg). 1H NMR (400 MHz, DMSO- d6) δ: 8.70 (dt, J=4.6, 1.6 Hz, 1H), 8.01 (td, J=7.8, 1.8 Hz, 1H), 7.56 (ddt, J=7.4, 5.6, 1.7 Hz, 4H), 7.51 (dtd, J=6.6, 4.6, 1.8 Hz, 3H), 4.94 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δ: 151.41, 149.96, 138.58, 132.31, 130.13, 129.01, 127.05, 125.59, 119.96, 116.06, 67.15, 59.00; HRMS (TOF ES+) calcd for C14H11N2O [M+H]+ 223.0866, found 223.0858.
3-(对甲苯基)-2-(吡啶-2-基)环氧乙烷-2-甲腈(3l): 白色固体, 产率83% (196 mg). m.p. 150.3~151.9 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.72~8.64 (m, 1H), 8.00 (td, J=7.8, 1.8 Hz, 1H), 7.59~7.49 (m, 2H), 7.44 (d, J=8.2 Hz, 2H), 7.31 (d, J=8.2 Hz, 2H), 4.87 (s, 1H), 2.36 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 151.48, 149.96, 139.75, 138.62, 129.60, 129.33, 126.98, 125.57, 119.91, 116.10, 67.20, 58.99, 21.37; HRMS (TOF ES+) calcd for C15H13N2O [M+H]+ 237.1022, found 237.1029.
3-(4-甲氧基苯基)-2-(吡啶-2-基)环氧丙烷-2-甲腈(3m): 白色固体, 产率85% (215 mg). m.p. 158.5~159.3 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.68 (d, J=4.8 Hz, 1H), 7.99 (td, J=7.8, 1.6 Hz, 1H), 7.58~7.46 (m, 4H), 7.07 (d, J=8.8 Hz, 2H), 4.84 (s, 1H), 3.81 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 160.75, 151.55, 149.93, 138.61, 128.54, 125.53, 124.03, 119.80, 116.23, 114.47, 67.28, 59.07, 55.71, 55.69; HRMS (TOF ES+) calcd for C15H13N2O2 [M+H]+ 253.0972, found 253.0977.
3-(4-氯苯基)-2-(吡啶-2-基)环氧丙烷-2-甲腈(3n): 白色固体, 产率70% (180 mg). m.p. 165.9~166.9 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.69 (d, J=4.9 Hz, 1H), 8.00 (td, J=7.8, 1.8 Hz, 1H), 7.57 (d, J=12.5 Hz, 6H), 4.99 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δ: 151.16, 149.97, 138.59, 134.89, 131.40, 129.13, 128.96, 125.66, 120.07, 115.90, 66.39, 58.98; HRMS (TOF ES+) calcd for C14H10ClN2O [M+H]+ 257.0476, found 257.0471.
3-(4-氟苯基)-2-(吡啶-2-基)环氧丙烷-2-甲腈(3o): 白色固体, 产率72% (174 mg). m.p. 149.9~152.3 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.72~8.65 (m, 1H), 8.00 (td, J=7.7, 1.6 Hz, 1H), 7.68~7.51 (m, 4H), 7.36 (t, J=8.8 Hz, 2H), 4.97 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δ: 163.27 (d, J=244 Hz), 151.25, 149.96, 138.63, 129.39 (d, J=8 Hz), 128.61 (d, J=3 Hz), 125.66, 119.98, 116.21, 115.99, 66.52, 58.99; HRMS (TOF ES+) calcd for C14H10- FN2O [M+H]+ 241.0772, found 241.0768.
2-氰基-3-苯基环氧乙烷-2-酰胺(3p): 白色固体, 产率70% (132 mg). m.p. 173.8~174.6 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.07 (s, 1H), 7.88 (s, 1H), 7.52 (d, J=2.6 Hz, 5H), 4.81 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δ: 163.96, 131.50, 130.41, 129.12, 127.08, 114.86, 63.55, 56.01; HRMS (TOF ES+) calcd for C10H9N2O2 [M+H]+ 189.0659, found 189.0652.
2-氰基-3-(对甲苯基)环氧乙烷-2-甲酰胺(3q): 白色固体, 产率72% (146 mg). m.p. 177.3~178.9 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 8.06 (s, 1H), 7.86 (s, 1H), 7.38 (d, J=8.3 Hz, 2H), 7.30 (d, J=8.2 Hz, 2H), 4.74 (s, 1H), 2.35 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 164.05, 140.06, 129.66, 128.42, 126.99, 114.90, 63.62, 56.03, 21.35; HRMS (TOF ES+) calcd for C11H11N2O2 [M+H]+ 203.0815, found 203.0807.
2-氰基-3-(4-甲氧基苯基)环氧乙烷-2-甲酰胺(3r): 白色固体, 产率73% (160 mg). m.p. 183.3~185.0 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.05~7.99 (m, 1H), 7.83 (s, 1H), 7.47~7.40 (m, 2H), 7.08~7.02 (m, 2H), 4.71 (s, 1H), 3.80 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 164.10, 160.92, 128.59, 123.05, 115.00, 114.55, 63.63, 56.11, 55.72; HRMS (TOF ES+) calcd for C11H11N2O3 [M+H]+ 219.0764, found 219.0769.
3-(4-氯苯基)-2-氰基环氧乙烷-2-甲酰胺(3s): 白色固体, 产率65% (146 mg). m.p. 189.1~191.2 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.09 (s, 1H), 7.90 (s, 1H), 7.57 (d, J=8.5 Hz, 2H), 7.52 (d, J=4.3 Hz, 2H), 4.83 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δ: 163.71, 135.10, 130.54, 129.21, 128.97, 114.71, 62.87, 55.91; HRMS (TOF ES+) calcd for C10H8ClN2O2 [M+H]+ 223.0269, found 223.0261.
3-(4-氟苯基)-2-氰基环氧乙烷-2-甲酰胺(3t): 白色固体, 产率66% (137 mg). m.p. 180.9~181.9 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.08 (s, 1H), 7.88 (s, 1H), 7.61~7.48 (m, 2H), 7.39~7.32 (m, 2H), 4.83 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δ: 163.81, 163.39 (d, J=244 Hz), 129.45 (d, J=10 Hz), 127.75 (d, J=3 Hz), 116.19 (d, J=22 Hz), 114.79, 62.97, 55.96; HRMS (TOF ES+) calcd for C10H8FN2O2 [M+H]+ 207.0564, found 207.0571.
2-氰基-N-甲基-3-苯基环氧乙烷-2-甲酰胺(3u): 白色固体, 产率62% (126 mg). m.p. 157.7~159.0 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.40 (d, J=4.6 Hz, 1H), 7.50 (s, 5H), 4.79 (s, 1H), 2.70 (d, J=4.6 Hz, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 162.07, 131.34, 130.43, 129.11, 127.03, 114.77, 63.77, 56.02, 26.51; HRMS (TOF ES+) calcd for C11H11N2O2 [M+H]+ 203.0815, found 203.0819.
2-氰基-N-乙基-3-苯基环氧乙烷-2-甲酰胺(3v): 白色固体, 产率61% (132 mg). m.p. 160.3~162.2 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.46 (t, J=5.8 Hz, 1H), 7.50 (s, 5H), 4.77 (s, 1H), 3.37 (s, 2H), 1.09 (t, J=7.3 Hz, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 161.39, 131.34, 130.42, 129.10, 127.06, 114.81, 63.72, 56.06, 34.59, 14.76; HRMS (TOF ES+) calcd for C12H13N2O2 [M+H]+ 217.0972, found 217.0979.
2-氰基-N,3-二苯基环氧乙烷-2-甲酰胺(3w): 白色固体, 产率55% (146 mg). m.p. 140.4~141.9 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 10.17 (s, 1H), 7.77~7.70 (m, 2H), 7.63~7.49 (m, 5H), 7.44~7.36 (m, 2H), 7.22~7.16 (m, 1H), 5.05 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δ: 160.62, 137.78, 131.30, 130.54, 129.23, 129.19, 127.18, 125.30, 121.30, 114.70, 63.97, 56.71; HRMS (TOF ES+) calcd for C16H13N2O2 [M+H]+ 265.0972, found 265.0966.
2-氰基-N-苯基-3-(对甲苯基)环氧乙烷-2-甲酰胺(3x): 白色固体, 产率57% (159 mg). m.p. 155.5~156.1 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 10.15 (s, 1H), 7.72 (d, J=8.5 Hz, 2H), 7.51~7.29 (m, 6H), 7.17 (t, J=7.4 Hz, 1H), 4.99 (s, 1H), 2.37 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 160.71, 140.21, 137.75, 129.72, 129.19, 128.22, 127.10, 125.23, 121.22, 114.74, 64.06, 56.77, 21.40; HRMS (TOF ES+) calcd for C17H15N2O2 [M+H]+ 279.1128, found 279.1121.
2-氰基-3-(4-氟苯基)-N-苯基环氧乙烷-2-甲酰胺(3y): 白色固体, 产率52% (147 mg). m.p. 145.2~146.8 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 10.15 (s, 1H), 7.72 (d, J=8.5 Hz, 2H), 7.51~7.29 (m, 6H), 7.17 (t, J=7.4 Hz, 1H), 4.99 (s, 1H), 2.37 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 163.47(d, J=246 Hz), 160.49, 137.72, 129.56(d, J=9 Hz), 129.21, 127.55(d, J=3 Hz), 125.26, 121.20, 116.28(d, J=22 Hz), 114.64, 63.41, 56.70; HRMS (TOF ES+) calcd for C16H12FN2O2 [M+H]+ 283.0877, found 283.0869.
2-氰基-3-苯基-N-(对甲苯基)环氧乙烷-2-甲酰胺(3z): 白色固体, 产率53% (148 mg). m.p. 177.7~178.7 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 10.15 (s, 1H), 7.72 (d, J=8.5 Hz, 2H), 7.51~7.29 (m, 6H), 7.17 (t, J=7.4 Hz, 1H), 4.99 (s, 1H), 2.37 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 160.71, 140.21, 137.75, 129.72, 129.19, 128.22, 127.10, 125.23, 121.22, 114.74, 64.06, 56.77, 21.40; HRMS (TOF ES+) calcd for C17H15N2O2 [M+H]+ 279.1128, found 279.1121.
2-氰基-3-乙基-N-苯基环氧乙烷-2-甲酰胺(4a): 白色固体, 产率68% (147 mg). m.p. 86.2~87.7 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 9.93 (s, 1H), 7.72~7.58 (m, 2H), 7.56~7.34 (m, 2H), 7.28~7.08 (m, 1H), 3.82 (dd, J=6.7, 5.4 Hz, 1H), 1.96~1.73 (m, 2H), 1.16 (t, J=7.5 Hz, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 161.39, 137.68, 129.17, 125.24, 121.30, 115.41, 65.51, 53.91, 23.71, 10.08; HRMS (TOF ES+) calcd for C12H13N2O2 [M+H]+ 217.0972, found 217.0978.
2-氰基-3-异丙基-N-苯基环氧乙烷-2-甲酰胺(4b): 白色固体, 产率68% (157 mg). m.p. 94.3~96.7 ℃; 1H NMR (600 MHz, DMSO-d6) δ: 9.94 (s, 1H), 7.67~7.63 (m, 2H), 7.38~7.31 (m, 2H), 7.14 (tt, J=7.5, 1.2 Hz, 1H), 3.61 (d, J=8.9 Hz, 1H), 1.66 (dp, J=9.0, 6.8 Hz, 1H), 1.15 (dd, J=37.7, 6.8 Hz, 6H); 13C NMR (100 MHz, DMSO-d6) δ: 161.30, 137.65, 129.12, 125.19, 121.24, 115.32, 68.95, 53.86, 30.67, 19.55, 17.93; HRMS (TOF ES+) calcd for C13H15N2O2 [M+H]+ 231.1128, found 231.1121.
3-环己基-2-(吡啶-2-基)环氧丙烷-2-甲腈(4c): 白色固体, 产率73% (167 mg). m.p. 128.6~129.9 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.66 (dt, J=4.8, 1.5 Hz, 1H), 7.93 (td, J=7.8, 1.8 Hz, 1H), 7.51 (ddd, J=7.7, 4.8, 1.2 Hz, 1H), 7.38 (dd, J=8.0, 1.2 Hz, 1H), 3.51 (d, J=8.8 Hz, 1H), 1.96~1.87 (m, 1H), 1.76 (td, J=8.0, 7.2, 4.2 Hz, 3H), 1.66 (dt, J=13.8, 2.6 Hz, 1H), 1.53 (tdt, J=12.1, 8.7, 3.6 Hz, 1H), 1.42~1.16 (m, 5H); 13C NMR (100 MHz, DMSO-d6) δ: 151.94, 149.90, 138.48, 125.35, 119.81, 116.85, 70.72, 55.53, 39.84, 29.60, 28.05, 25.95, 25.25, 25.09; HRMS (TOF ES+) calcd for C14H17N2O [M+H]+ 229.1335, found 229.1342.
2-氰基-3-环戊基-N-苯基环氧乙烷-2-甲酰胺(4d): 白色固体, 产率67% (172 mg). m.p. 126.1~128.3 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 9.91 (s, 1H), 7.76~7.66 (m, 2H), 7.41~7.29 (m, 2H), 7.19~7.09 (m, 1H), 3.71 (d, J=8.1 Hz, 1H), 1.94 (tdd, J=17.0, 12.3, 8.5 Hz, 3H), 1.77~1.52 (m, 6H); 13C NMR (DMSO-d6, 100 MHz) δ: 161.36, 137.77, 129.07, 125.12, 121.24, 115.44, 67.98, 53.89, 40.74, 30.48, 28.22, 25.60, 25.45; HRMS (TOF ES+) calcd for C15H17N2O2 [M+H]+ 257.1285, found 257.1292.
2-氰基-3-(吡啶-4-基)环氧乙烷-2-羧酸甲酯(5a): 白色液体, 产率42% (86 mg); 1H NMR (400 MHz, DMSO- d6) δ: 8.73~8.67 (m, 2H), 7.52~7.48 (m, 2H), 4.92 (s, 1H), 3.89 (s, 3H); 13C NMR (DMSO-d6, 100 MHz) δ: 162.93, 150.35, 140.23, 122.05, 113.98, 62.79, 54.71, 53.79; HRMS (TOF ES+) calcd for C10H9N2O3 [M+H]+ 205.0608, found 205.0601.
N-(4-氯苯基)-2-氰基-3-(吡啶-3-基)环氧乙烷-2-甲酰胺(5b): 白色固体, 产率71% (213 mg). m.p. 136.4~138.9 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.45 (d, J=4.9 Hz, 1H), 8.23~8.17 (m, 2H), 7.99~7.88 (m, 4H), 7.47~7.30 (m, 6H); 13C NMR (100 MHz, DMSO-d6) δ: 160.51, 151.54, 148.83, 136.77, 134.80, 129.21, 129.18, 129.05, 129.01, 127.47, 124.13, 122.83, 122.29, 114.46, 62.35, 56.69; HRMS (TOF ES+) calcd for C15H11ClN3O2 [M+H]+ 300.0534, found 300.0539.