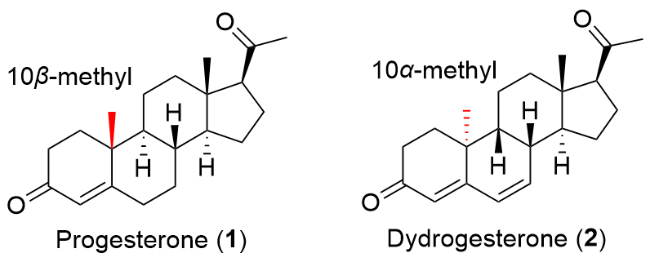
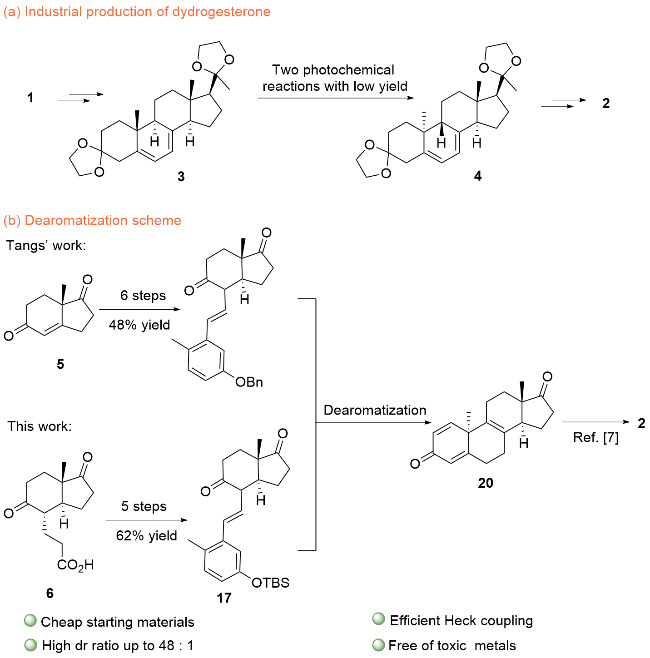
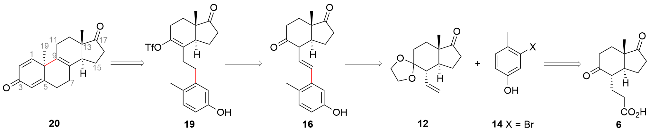
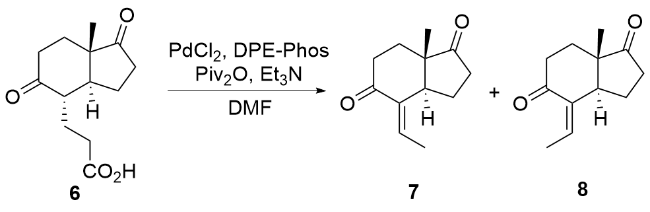
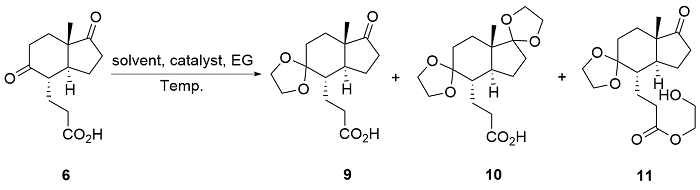
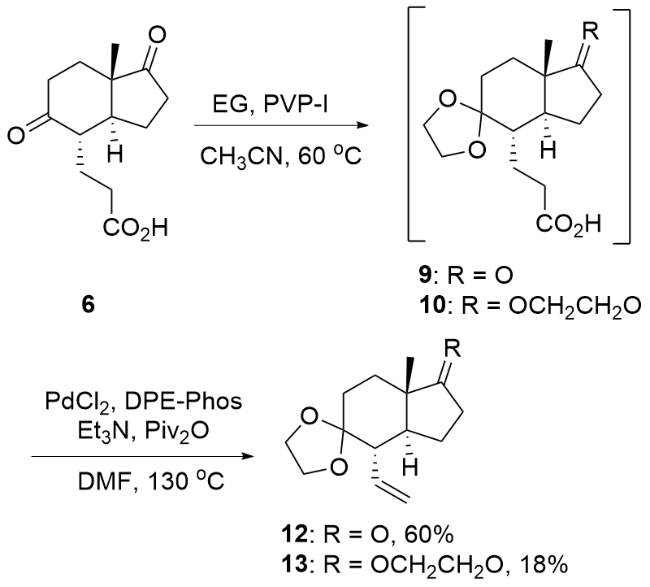
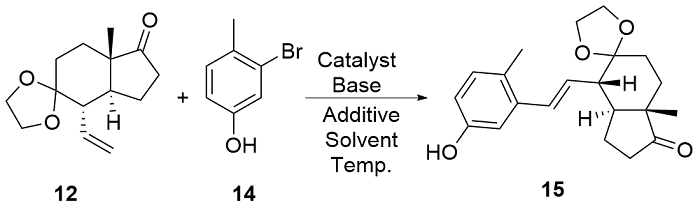
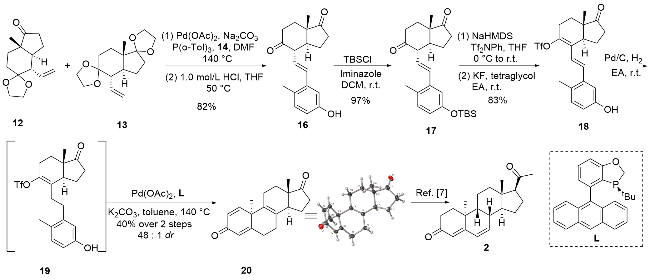
| [8] |
(a) Samir, N.; Suresh, V.; Sachin, S.; Abraham, T.; Pramod, P.; Bhaskar, B. S. WO 2018109622, 2018.
(b) Rappoldt, M. P.; Westerhof, P. Recl. Trav. Chim. Pays-Bas 1961, 80, 43.
(c) Engbert, H. R.; Westerhof, P.; Frederik, L. S. H. US 3198792, 1965.
(d) Rappoldt, M. P.; Rix, T. H. R. Recl. Trav. Chim. Pays-Bas 1971, 90, 27.
(e) Bout, B.; Voorhaar, R.EP 0558119, 1993.
(f) de Lignieres, B.; Dennerstein, L.; Backstrom, T. Maturitas 1995, 21, 251.
PMID
(g) Schindler, A. E.; Campagnoli, C.; Druckmann, R.; Huber, J.; Pasqualini, J. R.; Schweppe, K. W.; Thijssen, J. H. H. Maturitas 2003, 46, 7.
(h) Zhang, J.; Wang, C.; Chen, J.WO 2013078575, 2013.
(i) Dong, J.; Wu, Z.; Chen, J.WO 2016154772, 2016.
(j) Pan, C.; Su, G.; Yu, C.; Qin, J.; Li, Y.; Yang, Q.; Fang, S.WO 2021093494, 2021.
(k) Li, X.; Zhang, R.; Li, J.; Liu, N.; Chen, X.; Liu, Y.; Zhao, G.; Ding, K.; Yao, P.; Feng, J.; Wu, Q.; Zhu, D.; Ma, Y. JACS Au 2024, 4, 1356.
|