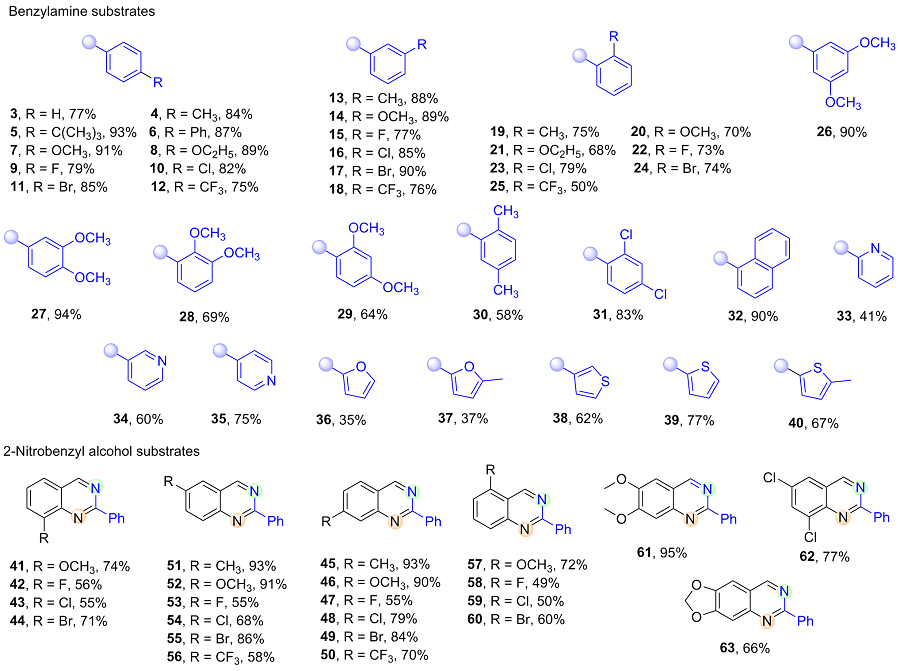
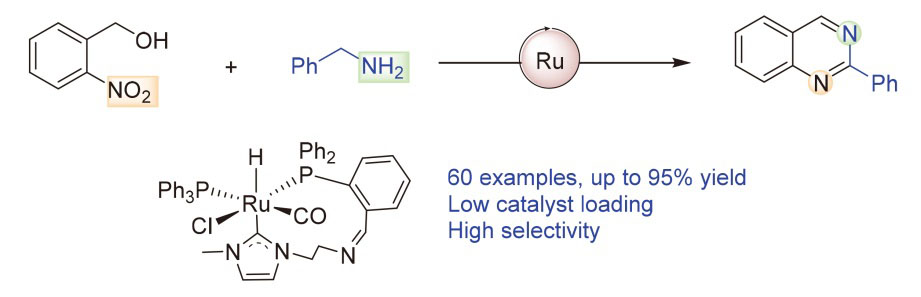
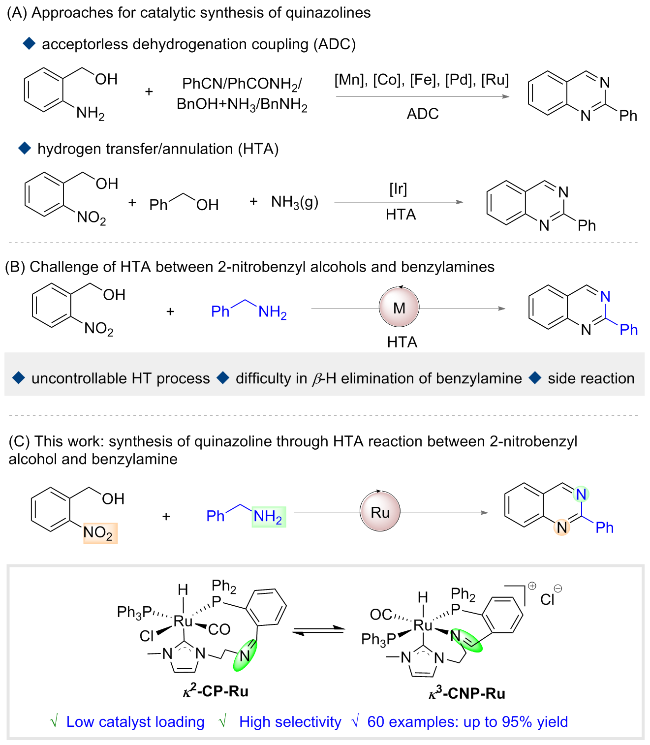
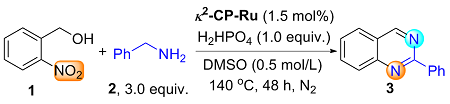
2-Nitrobenzyl alcohol (0.5 mmol), κ2-CP-Ru (1.5 mmol%, 6.3 mg) and K2HPO4 (0.5 mmol, 87 mg) were added into an oven-dried Schlenk tube equipped with a N2 balloon, and it was subject to three cycles of vacuum and refilled with nitrogen. Then another alcohol (1.5 mmol) and 1 mL of PhCl were sequentially injected and the mixture was stirred at 140 ℃. After reacting 48 h, the mixture was filtered, and the sample was washed with dichloromethane. The crude residue was purified by flash column chromatography [unless mentioned otherwise, the mixture of petroleum (PE)/ethyl acetate (EA) with V∶V=30∶1 was used as eluent) to obtain the pure product.
2-Phenylquinazoline (3): Light yellow solid, 79 mg, 77% yield. m.p. 103~105 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.50 (s, 1H), 8.69~8.60 (m, 2H), 8.18~8.07 (m, 1H), 8.01~7.87 (m, 2H), 7.71~7.61 (m, 1H), 7.60~7.49 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.0, 160.5, 150.7, 138.0, 134.1, 130.6, 128.6, 128.6, 127.2, 127.1, 123.6; HRMS (ESI+) calcd for C14H11N2 [M+H]+ 207.1917, found 207.1922.
2-(p-Tolyl)quinazoline (4): Light yellow solid, 88 mg, 84% yield. m.p. 97~99 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.46 (s, 1H), 8.64~8.46 (m, 2H), 8.20~7.99 (m, 1H), 8.00~7.83 (m, 2H), 7.65~7.52 (m, 1H), 7.37 (d, J=8.0 Hz, 2H), 2.47 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.1, 160.4, 150.8, 140.8, 135.3, 134.0, 129.4, 128.5, 127.1, 127.0, 123.5, 21.5; HRMS (ESI+) calcd for C15H13N2 [M+H]+ 211.1073, found 211.1068.
2-(4-(tert-Butyl)phenyl)quinazoline (5): Light yellow solid, 122 mg, 93% yield. m.p. 82~84 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.47 (s, 1H), 8.66~8.52 (m, 2H), 8.10 (dd, J=8.4, 0.8 Hz, 1H), 7.97~7.85 (m, 2H), 7.67~7.53 (m, 3H), 1.42 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 161.1, 160.4, 153.9, 150.8, 135.3, 134.0, 128.6, 128.3, 127.1, 127.0, 125.6, 123.5, 34.8, 31.3; HRMS (ESI+) calcd for C18H19N2 [M+H]+ 263.1543, found 263.1543.
2-([1'-Viphenyl]-4-yl)quinazoline (6): Light yellow solid, 123 mg, 87% yield. m.p. 117~119 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.50 (s, 1H), 8.73 (d, J=8.4 Hz, 2H), 8.13 (d, J=8.4 Hz, 1H), 8.00~7.90 (m, 2H), 7.86~7.78 (m, 2H), 7.74 (dd, J=5.2, 3.3 Hz, 2H), 7.68~7.58 (m, 1H), 7.51 (dd, J=10.3, 4.8 Hz, 2H), 7.47~7.38 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.8, 160.5, 150.8, 143.2, 140.6, 137.0, 134.1, 129.0, 128.8, 128.6, 127.6, 127.3, 127.2, 127.2, 127.1, 123.6; HRMS (ESI+) calcd for C20H15N2 [M+H]+ 283.1230, found 283.1231.
2-(4-Methoxyphenyl)quinazoline (7): Light yellow solid, 107 mg, 91% yield. m.p. 91~93 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.42 (s, 1H), 8.74~8.50 (m, 2H), 8.05 (d, J=9.0 Hz, 1H), 8.00~7.83 (m, 2H), 7.65~7.48 (m, 1H), 7.16~6.98 (m, 2H), 3.91 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.8, 160.8, 160.3, 150.8, 134.0, 130.7, 130.2, 128.3, 127.1, 126.7, 123.3, 113.9, 55.3; HRMS (ESI+) calcd for C15H13N2O [M+H]+ 237.1022, found 237.1015.
2-(4-Ethoxyphenyl)quinazoline (8): Light yellow solid, 111 mg, 89% yield. m.p. 123~125 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.42 (s, 1H), 8.78~8.48 (m, 2H), 8.05 (d, J=9.0 Hz, 1H), 7.93~7.80 (m, 2H), 7.70~7.43 (m, 1H), 7.24~6.85 (m, 2H), 4.14 (q, J=7.0 Hz, 2H), 1.48 (t, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.2, 160.8, 160.3, 150.8, 134.0, 130.5, 130.2, 128.3, 127.1, 126.7, 123.2, 114.4, 63.5, 14.8; HRMS (ESI+) calcd for C16H15N2O [M+H]+ 251.1179, found 251.1173.
2-(4-Fluorophenyl)quinazoline (9): Yellow solid, 88 mg, 79% yield. m.p. 136~138 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.46 (s, 1H), 8.82~8.53 (m, 2H), 8.09 (d, J=8.3 Hz, 1H), 8.02~7.84 (m, 2H), 7.71~7.51 (m, 1H), 7.27~7.15 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 164.7 (d, J=250.4 Hz), 160.5, 160.0, 150.6, 134.2, 134.1, 134.1, 130.7 (d, J=8.7 Hz), 128.5, 127.2 (d, J=15.9 Hz), 123.4, 115.5 (d, J=21.6 Hz); HRMS (ESI+) calcd for C14H17FN2 [M+H]+ 225.0823, found 225.0824.
2-(4-Chlorophenyl)quinazoline (10): Yellow solid, 98 mg, 82% yield. m.p. 137~139 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.46 (s, 1H), 8.68~8.52 (m, 2H), 8.16~8.02 (m, 1H), 8.00~7.89 (m, 2H), 7.72~7.58 (m, 1H), 7.58~7.45 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 160.5, 159.9, 150.6, 136.8, 136.4, 134.3, 129.9, 128.8, 128.5, 127.4, 127.1, 123.6; HRMS (ESI+) calcd for C14H17ClN2 [M+ H]+ 241.0527, found 241.0531.
2-(4-Bromophenyl)quinazoline (11): yellow solid, 121 mg, 85% yield. m.p. 122~124 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.45 (s, 1H), 8.55~8.45 (m, 2H), 8.23~8.03 (m, 1H), 7.99~7.86 (m, 2H), 7.73~7.58 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 160.5, 160.0, 150.6, 136.9, 134.3, 131.7, 130.1, 128.5, 127.5, 127.1, 125.4, 123.6; HRMS (ESI+) calcd for C14H10BrN2 [M+H]+ 285.0022, found 285.0017.
2-(4-(Trifluoromethyl)phenyl)quinazoline (12): Brown solid, 103 mg, 75% yield. m.p. 144~146 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.50 (s, 1H), 8.76 (d, J=8.1 Hz, 2H), 8.13 (dd, J=8.3, 0.8 Hz, 1H), 7.95 (dd, J=12.2, 4.5 Hz, 2H), 7.80 (d, J=8.2 Hz, 2H), 7.70~7.59 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.6, 159.6, 150.6, 141.2, 134.3, 132.1 (q, J=32.3 Hz), 128.8, 128.7, 127.8, 127.1, 125.5 (q, J=3.8 Hz), 125.5(q, J=272.4 Hz), 123.8; HRMS (ESI+) calcd for C15H10F3N2 [M+H]+ 275.0791, found 275.0785.
2-(m-Tolyl)quinazoline (13): Light yellow solid, 92 mg, 88% yield. m.p. 101~103 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.48 (s, 1H), 8.45 (dd, J=6.4, 5.3 Hz, 2H), 8.16~8.07 (m, 1H), 8.03~7.83 (m, 2H), 7.68~7.54 (m, 1H), 7.50~7.42 (m, 1H), 7.35 (d, J=7.5 Hz, 1H), 2.52 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.2, 160.4, 150.7, 138.2, 137.9, 134.1, 131.4, 129.1, 128.6, 128.6, 127.2, 127.1, 125.8, 123.5, 21.5; HRMS (ESI+) calcd for C15H13N2 [M+H]+ 211.1073, found 211.1080.
2-(3-Methoxyphenyl)quinazoline (14): Light yellow solid, 105 mg, 89% yield. m.p. 82~84 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.48 (s, 1H), 8.29~8.23 (m, 1H), 8.21 (dd, J=2.5, 1.5 Hz, 1H), 8.11 (dd, J=8.4, 0.8 Hz, 1H), 7.99~7.88 (m, 2H), 7.68~7.58 (m, 1H), 7.51~7.43 (m, 1H), 7.17~7.04 (m, 1H), 3.98 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 160.8, 160.4, 160.0, 150.7, 139.5, 134.1, 129.6, 128.6, 127.3, 127.1, 123.6, 121.1, 117.2, 113.0, 55.4; HRMS (ESI+) calcd for C15H13N2O [M+H]+ 237.1022, found 237.1027.
2-(3-Fluorophenyl)quinazoline (15): Light yellow solid, 86 mg, 77% yield. m.p. 95~97 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.49 (s, 1H), 8.44 (d, J=7.8 Hz, 1H), 8.39~8.28 (m, 1H), 8.18~8.06 (m, 1H), 8.02~7.88 (m, 2H), 7.74~7.61 (m, 1H), 7.60~7.46 (m, 1H), 7.26~7.18 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 163.2 (d, J=244.8 Hz), 160.5, 159.8 (d, J=3.4 Hz), 150.6, 140.4 (d, J=7.9 Hz), 134.3, 130.0 (d, J=8.0 Hz), 128.7, 127.6, 127.1, 124.1 (d, J=2.8 Hz), 123.7, 117.4 (d, J=21.5 Hz), 115.4 (d, J=23.3 Hz); HRMS (ESI+) calcd for C14H17FN2 [M+H]+ 225.0823, found 225.0821.
2-(3-Chlorophenyl)quinazoline (16): Yellow solid, 102 mg, 85% yield. m.p. 148~150 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.49 (s, 1H), 8.66 (dd, J=2.2, 1.3 Hz, 1H), 8.61~8.48 (m, 1H), 8.12 (dd, J=8.3, 0.8 Hz, 1H), 8.02~7.90 (m, 2H), 7.73~7.60 (m, 1H), 7.54~7.44 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 160.5, 159.7, 150.6, 139.8, 134.7, 134.3, 130.5, 129.8, 128.6, 128.6, 127.6, 127.1, 126.6, 123.7; HRMS (ESI+) calcd for C14H17ClN2 [M+ H]+ 241.0527, found 241.0529.
2-(3-Bromophenyl)quinazoline (17): Yellow solid, 128 mg, 90% yield. m.p. 149~151 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.47 (s, 1H), 8.81 (t, J=1.8 Hz, 1H), 8.65~8.50 (m, 1H), 8.10 (d, J=9.0 Hz, 1H), 8.65~8.50 (m, 1H), 8.10 (d, J=9.0 Hz, 1H), 8.00~7.87 (m, 2H), 7.71~7.60 (m, 2H), 7.46~7.32 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.5, 159.6, 150.6, 140.0, 134.3, 133.4, 131.5, 130.1, 128.7, 127.6, 127.1, 127.1, 123.7, 122.9; HRMS (ESI+) calcd for C14H10BrN2 [M+H]+ 285.0022, found 285.0029.
2-(3-(Trifluoromethyl)phenyl)quinazoline (18): Brown solid, 104 mg, 76% yield. m.p. 146~148 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.50 (s, 1H), 8.96 (s, 1H), 8.85 (d, J=7.8 Hz, 1H), 8.20~8.10 (m, 1H), 8.02~7.92 (m, 2H), 7.78 (d, J=7.7 Hz, 1H), 7.67 (dd, J=11.6, 4.2 Hz, 2H); 13C NMR (101 MHz, CDCl3) δ: 160.6, 159.5, 150.6, 138.8, 134.3, 131.6, 131.1 (q, J=32.4 Hz), 129.0, 128.7, 127.7, 127.1, 127.0 (q, J=3.7 Hz), 125.5 (q, 3.9 Hz), 124.0 (q, J=274.1 Hz), 123.8; HRMS (ESI+) calcd for C15H10F3N2 [M+H]+ 275.0791, found 275.0797.
2-(o-Tolyl)quinazoline (19): Light yellow solid, 79 mg, 75% yield. m.p. 44~46 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.52 (s, 1H), 8.12 (dd, J=8.5, 0.7 Hz, 1H), 8.00~7.90 (m, 3H), 7.73~7.61 (m, 1H), 7.44~7.31 (m, 3H), 2.64 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 164.0, 160.0, 150.4, 138.6, 137.3, 134.1, 131.3, 130.6, 129.3, 128.5, 127.5, 127.0, 125.9, 122.9, 21.0; HRMS (ESI+) calcd for C15H13N2 [M+H]+ 211.1073, found 211.1068.
2-(2-Methoxyphenyl)quinazoline (20): Light yellow solid, 83 mg, 70% yield. m.p. 118~120 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.52 (s, 1H), 8.13 (dd, J=8.5, 0.7 Hz, 1H), 8.03~7.88 (m, 2H), 7.80 (dd, J=7.6, 1.8 Hz, 1H), 7.70~7.61 (m, 1H), 7.53~7.41 (m, 1H), 7.15~7.10 (m, 1H), 7.08 (d, J=8.4 Hz, 1H), 3.89 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 162.4, 160.0, 157.6, 150.6, 134.0, 131.7, 130.8, 128.9, 128.5, 127.5, 127.0, 123.1, 120.8, 111.9, 56.0; HRMS (ESI+) calcd for C15H13N2O [M+H]+ 237.1022, found 237.1015.
2-(2-Ethoxyphenyl)quinazoline (21): Light yellow solid, 85 mg, 68% yield. m.p. 127~129 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.50 (s, 1H), 8.19~8.08 (m, 1H), 8.01~7.87 (m, 2H), 7.79 (dd, J=7.6, 1.8 Hz, 1H), 7.70~7.60 (m, 1H), 7.50~7.37 (m, 1H), 7.20~7.00 (m, 2H), 4.14 (q, J=7.0 Hz, 2H), 1.34 (t, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 162.5, 159.8, 157.2, 150.5, 133.9, 131.6, 130.7, 129.3, 128.5, 127.4, 127.0, 123.0, 120.7, 113.5, 64.6, 14.7; HRMS (ESI+) calcd for C16H15N2O [M+H]+ 251.1179, found 251.1173.
2-(2-Fluorophenyl)quinazoline (22): Light yellow solid, 81 mg, 73% yield. m.p. 88~90 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 9.52 (s, 1H), 8.22~8.15 (m, 1H), 8.15~8.10 (m, 1H), 7.98~7.89 (m, 2H), 7.70~7.63 (m, 1H), 7.53~7.44 (m, 1H), 7.35~7.29 (m, 1H), 7.29~7.20 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 161.2 (d, J=254.6 Hz), 160.4, 159.8 (d, J=4.4 Hz), 150.5, 134.3, 132.1 (d, J=1.9 Hz), 131.6 (d, J=8.6 Hz), 128.6, 127.9, 127.1,127.0 (d, J=9.8 Hz), 124.2 (d, J=3.8 Hz), 123.2, 116.8 (d, J=22.4 Hz); HRMS (ESI+) calcd for C14H17FN2 [M+H]+ 225.0823, found 225.0824.
2-(2-Chlorophenyl)quinazoline (23): Light yellow solid, 95 mg, 79% yield. m.p. 66~68 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.55 (s, 1H), 8.15 (dd, J=8.5, 0.7 Hz, 1H), 8.05~7.92 (m, 2H), 7.90~7.79 (m, 1H), 7.79~7.64 (m, 1H), 7.63~7.50 (m, 1H), 7.50~7.36 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 161.9, 160.2, 150.3, 138.2, 134.4, 132.9, 131.8, 130.5, 130.3, 128.6, 128.0, 127.1, 126.9, 123.2; HRMS (ESI+) calcd for C14H17ClN2 [M+H]+ 241.0527, found 241.0531.
2-(2-Bromophenyl)quinazoline (24): Yellow solid, 105 mg, 74% yield. m.p. 71~73 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.54 (s, 1H), 8.15 (dd, J=8.5, 0.6 Hz, 1H), 8.07~7.92 (m, 2H), 7.81 (dd, J=7.6, 1.7 Hz, 1H), 7.78~7.66 (m, 2H), 7.50~7.44 (m, 1H), 7.38~7.30 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 162.7, 160.2, 150.2, 140.1, 134.4, 133.7, 131.6, 130.4, 128.6, 128.1, 127.4, 127.1, 123.3, 121.9; HRMS (ESI+) calcd for C14H10BrN2 [M+ H]+ 285.0022, found 285.0017.
2-(2-(Trifluoromethyl)phenyl)quinazoline (25): Brown solid, 69 mg, 50% yield. Purified by flash column chromatography using PE/EA (V∶V=20∶1) as eluent. m.p. 97~99 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.53 (s, 1H), 8.14 (d, J=8.4 Hz, 1H), 8.07~7.95 (m, 2H), 7.86 (dd, J=7.6, 3.4 Hz, 2H), 7.79~7.66 (m, 2H), 7.66~7.58 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 162.4, 160.1, 150.1, 138.7 (q, J=4.0 Hz), 134.5, 131.6, 131.5, 129.1, 128.8 (q, J=31.3 Hz), 128.6, 128.1, 127.1, 126.8 (q, J=5.2 Hz), 124.0 (q, J=273.7 Hz), 123.2; HRMS (ESI+) calcd for C15H10F3N2 [M+H]+ 275.0791, found 275.0785.
2-(3,5-Dimethoxyphenyl)quinazoline (26): Light yellow solid, 120 mg, 90% yield. m.p. 106~108 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.47 (s, 1H), 8.12 (dd, J=8.4, 0.8 Hz, 1H), 7.98~7.89 (m, 2H), 7.86 (d, J=2.4 Hz, 2H), 7.69~7.59 (m, 1H), 6.69~6.63 (m, 1H), 3.95 (s, 6H); 13C NMR (101 MHz, CDCl3) δ: 161.0, 160.6, 160.3, 150.6, 140.0, 134.1, 128.6, 127.3, 127.1, 123.7, 106.2, 103.8, 55.6; HRMS (ESI+) calcd for C16H15N2O2 [M+H]+ 267.1128, found 267.1123.
2-(3,4-Dimethoxyphenyl)quinazoline (27): Light yellow solid, 125 mg, 94% yield. m.p. 110~112 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.41 (s, 1H), 8.27 (dd, J=8.4, 2.0 Hz, 1H), 8.21 (d, J=1.9 Hz, 1H), 8.06 (d, J=9.0 Hz, 1H), 7.96~7.82 (m, 2H), 7.63~7.50 (m, 1H), 7.02 (d, J=8.5 Hz, 1H), 4.07 (s, 3H), 3.98 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 160.6, 160.3, 151.3, 150.7, 149.0, 134.0, 130.8, 128.3, 127.1, 126.8, 123.3, 122.0, 111.1, 110.8, 56.0, 55.9; HRMS (ESI+) calcd for C16H15N2O2 [M+H]+ 267.1128, found 267.1127.
2-(2,3-Dimethoxyphenyl)quinazoline (28): Light yellow solid, 92 mg, 69% yield. m.p. 113~115 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.53 (s, 1H), 8.13 (d, J=8.5 Hz, 1H), 8.02~7.87 (m, 2H), 7.68 (dd, J=11.1, 3.9 Hz, 1H), 7.44 (dd, J=7.8, 1.5 Hz, 1H), 7.25~7.13 (m, 1H), 7.08 (dd, J=8.2, 1.4 Hz, 1H), 3.96 (s, 3H), 3.95 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 162.0, 160.1, 153.4, 150.5, 148.0, 134.3, 134.0, 128.6, 127.5, 127.0, 124.0, 123.2, 123.1, 113.9, 61.6, 56.1; HRMS (ESI+) calcd for C16H15N2O2 [M+H]+ 267.1128, found 267.1123.
2-(2,4-Dimethoxyphenyl)quinazoline (29): Light yellow solid, 85 mg, 64% yield. m.p. 90~92 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.50 (s, 1H), 8.11 (dd, J=8.5, 0.6 Hz, 1H), 7.99~7.79 (m, 3H), 7.71~7.58 (m, 1H), 6.72~6.58 (m, 2H), 3.91 (s, 3H), 3.90 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 162.2, 162.0, 159.9, 159.2, 150.6, 133.9, 133.2, 128.4, 127.2, 127.0, 122.8, 121.7, 105.0, 99.4, 56.1, 55.5; HRMS (ESI+) calcd for C16H15N2O2 [M+H]+ 267.1128, found 267.1130.
2-(2,5-Dimethylphenyl)quinazoline (30): Light yellow solid, 68 mg, 58% yield. m.p. 93~95 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.52 (s, 1H), 8.13 (d, J=8.4 Hz, 1H), 8.04~7.91 (m, 2H), 7.74 (s, 1H), 7.71~7.65 (m, 1H), 7.27~7.16 (m, 2H), 2.58 (s, 3H), 2.43 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 164.1, 160.0, 150.3, 138.2, 135.4, 134.2, 134.1, 131.2, 131.1, 130.1, 128.5, 127.4, 127.0, 122.8, 20.9, 20.5; HRMS (ESI+) calcd for C16H15N2 [M+H]+ 235.1210, found 235.1216.
2-(2,4-Dichlorophenyl)quinazoline (31): Light yellow solid, 114 mg, 83% yield. m.p. 109~111 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.54 (s, 1H), 8.14 (d, J=9.5 Hz, 1H), 8.10~7.94 (m, 2H), 7.84 (d, J=8.3 Hz, 1H), 7.77~7.69 (m, 1H), 7.69~7.29 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 160.9, 160.3, 150.3, 136.6, 135.7, 134.5, 133.8, 132.8, 130.4, 128.6, 128.2, 127.2, 127.1, 123.3; HRMS (ESI+) calcd for C14H9Cl2N2 [M+H]+ 275.0137, found 275.0141.
2-(Naphthalen-1-yl)quinazoline (32): Light yellow solid, 115 mg, 90% yield. m.p. 124~126 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.63 (s, 1H), 8.74 (dd, J=8.0, 6.3 Hz, 1H), 8.21 (dd, J=5.1, 4.0 Hz, 2H), 8.08~7.89 (m, 4H), 7.79~7.70 (m, 1H), 7.69~7.62 (m, 1H), 7.62~7.53 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 163.4, 160.4, 150.5, 136.3, 134.3, 134.2, 131.2, 130.4, 129.6, 128.6, 128.5, 127.7, 127.1, 126.8, 125.9, 125.3, 123.1; HRMS (ESI+) calcd for C18H13N2 [M+H]+ 257.1073, found 257.1073.
2-(Pyridin-2-yl)quinazoline (33): Light brown oil, 42 mg, 41% yield. Purified by flash column chromatography using PE/EA (V∶V=5∶1) as eluent. 1H NMR (400 MHz, CDCl3) δ: 9.56~9.48 (m, 1H), 8.87 (s, 1H), 8.69~8.62 (m, 1H), 8.23~8.16 (m, 1H), 8.01~7.78 (m, 3H), 7.62 (q, J=7.4 Hz, 1H), 7.43~7.34 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.9, 159.8, 155.0, 150.7, 150.1, 136.9, 134.3, 129.1, 128.0, 127.1, 124.7, 124.0, 124.0; HRMS (ESI+) calcd for C13H10N3 [M+H]+ 208.0869, found 208.0865.
2-(Pyridin-3-yl)quinazoline (34): Light brown oil, 62 mg, 60% yield. Purified by flash column chromatography using PE/EA (V∶V=10∶1) as eluent. 1H NMR (400 MHz, CDCl3) δ: 9.83 (s, 1H), 9.51~9.47 (m, 1H), 8.92~8.84 (m, 1H), 8.75 (dd, J=4.8, 1.7 Hz, 1H), 8.15~8.08 (m, 1H), 7.99~7.90 (m, 2H), 7.71~7.62 (m, 1H), 7.51~7.43 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.6, 159.1, 151.1, 150.6, 150.2, 135.8, 134.4, 133.5, 128.6, 127.8, 127.2, 123.8, 123.4; HRMS (ESI+) calcd for C13H10N3 [M+H]+ 208.0869, found 208.0863.
2-(Pyridin-4-yl)quinazoline (35): Light brown solid, 78 mg, 75% yield. Purified by flash column chromatography using PE/EA (V∶V=10∶1) as eluent. m.p. 104~106 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.53 (s, 1H), 8.86~8.82 (m, 2H), 8.49 (d, J=5.7 Hz, 2H), 8.16 (d, J=8.4 Hz, 1H), 8.04~7.95 (m, 2H), 7.76~7.68 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.7, 158.9, 150.5, 150.3, 145.4, 134.5, 128.9, 128.3, 127.2, 124.1, 122.4; HRMS (ESI+) calcd for C13H10N3 [M+H]+ 208.0869, found 208.0866.
2-(Furan-2-yl)quinazoline (36): Light yellow oil, 34 mg, 35% yield. Purified by flash column chromatography using PE/EA (V∶V=20∶1) as eluent. 1H NMR (400 MHz, CDCl3) δ: 9.39 (s, 1H), 8.19~8.05 (m, 1H), 7.95~7.85 (m, 2H), 7.70 (dd, J=1.7, 0.8 Hz, 1H), 7.67~7.53 (m, 1H), 7.47 (dd, J=3.4, 0.8 Hz, 1H), 6.63 (dd, J=3.4, 1.7 Hz, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.7, 154.0, 152.5, 150.4, 145.3, 134.5, 128.3, 127.2, 127.2, 123.3, 114.1, 112.3; HRMS (ESI+) calcd for C12H9N2O [M+H]+ 197.0709, found 197.0714.
2-(5-Methylfuran-2-yl)quinazoline (37): Light yellow solid, 39 mg, 37% yield. Purified by flash column chromatography using PE/EA (V∶V=20∶1) as eluent. m.p. 114~116 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.34 (s, 1H), 8.11~8.04 (m, 1H), 7.92~7.83 (m, 2H), 7.60~7.52 (m, 1H), 7.39 (d, J=3.3 Hz, 1H), 6.26~6.21 (m, 1H), 2.51 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 160.7, 156.1, 154.2, 151.1, 150.5, 134.4, 128.3, 127.3, 126.9, 123.2, 115.7, 108.9, 14.3; HRMS (ESI+) calcd for C13H11N2O [M+H]+ 211.0866, found 211.0869.
2-(Thiophen-3-yl)quinazoline (38): Light yellow oil, 66 mg, 62% yield. Purified by flash column chromatography using PE/EA (V∶V=20∶1) as eluent. 1H NMR (400 MHz, CDCl3) δ: 9.41 (s, 1H), 8.47 (dd, J=3.1, 1.1 Hz, 1H), 8.26~8.00 (m, 2H), 8.00~7.84 (m, 2H), 7.71~7.55 (m, 1H), 7.44 (dd, J=5.1, 3.1 Hz, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.5, 158.2, 150.6, 141.9, 134.1, 128.3, 128.3, 127.7, 127.1, 127.0, 126.1, 123.3; HRMS (ESI+) calcd for C12H9N2S [M+H]+ 213.0481, found 213.0489.
2-(Thiophen-2-yl)quinazoline (39): Light yellow oil, 82 mg, 77% yield. Purified by flash column chromatography using PE/EA (V∶V=20∶1) as eluent. 1H NMR (400 MHz, CDCl3) δ: 9.36 (s, 1H), 8.18 (dd, J=3.7, 1.1 Hz, 1H), 8.09~7.96 (m, 1H), 7.92~7.83 (m, 2H), 7.65~7.46 (m, 2H), 7.21 (dd, J=4.9, 3.8 Hz, 1H); 13C NMR (101 MHz, CDCl3) δ: 160.5, 157.8, 150.6, 143.8, 134.3, 129.8, 129.2, 128.4, 128.1, 127.2, 127.0, 123.3; HRMS (ESI+) calcd for C12H9N2S [M+H]+ 213.0481, found 213.0479.
2-(5-Methylthiophen-2-yl)quinazoline (40): Light yellow solid, 76 mg, 67% yield. Purified by flash column chromatography using PE/EA (V∶V=20∶1) as eluent. m.p. 137~139 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.32 (s, 1H), 8.02~7.94 (m, 2H), 7.90~7.82 (m, 2H), 7.58~7.50 (m, 1H), 6.90~6.84 (m, 1H), 2.58 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 160.5, 157.9, 150.6, 145.2, 141.2, 134.2, 129.6, 128.0, 127.2, 126.9, 126.7, 123.2, 15.8; HRMS (ESI+) calcd for C13H10N2S [M+H]+ 227.0637, found 227.0639.
8-Methoxy-2-phenylquinazoline (41): Light yellow solid, 87 mg, 74% yield. m.p. 99~101 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.46 (s, 1H), 8.91~8.33 (m, 2H), 7.62~7.33 (m, 5H), 7.25 (dd, J=7.3, 1.7 Hz, 1H), 4.14 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 160.4, 160.3, 154.9, 142.9, 138.0, 130.5, 128.6, 128.5, 127.5, 124.4, 118.6, 112.0, 56.3; HRMS (ESI+) calcd for C15H13N2O [M+H]+ 237.1022, found 237.1028.
8-Fluoro-2-phenylquinazoline (42): Light yellow solid, 63 mg, 56% yield. m.p. 137~139 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.53 (s, 1H), 8.74~8.62 (m, 2H), 7.82~7.72 (m, 1H), 7.68~7.51 (m, 5H); 13C NMR (101 MHz, CDCl3) δ: 160.2, 160.2, 159.9 (d, J=259.0 Hz), 156.0, 141.2 (d, J=12.7 Hz), 137.5, 131.0, 128.7, 128.6, 127.0 (d, J=7.3 Hz), 122.7 (d, J=4.9 Hz), 118.1 (d, J=18.3 Hz); HRMS (ESI+) calcd for C14H17FN2 [M+H]+ 225.0823, found 225.0823.
8-Chloro-2-phenylquinazoline (43): Light yellow solid, 66 mg, 55% yield. m.p. 111~113 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.50 (s, 1H), 8.89~8.55 (m, 2H), 8.03 (dd, J=7.5, 1.3 Hz, 1H), 7.88 (dd, J=8.1, 1.3 Hz, 1H), 7.72~7.39 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 161.5, 160.7, 147.3, 137.6, 133.9, 133.2, 131.0, 128.8, 128.7, 127.1, 125.9, 124.6; HRMS (ESI+) calcd for C14H17ClN2 [M+ H]+ 241.0527, found 241.0529.
8-Bromo-2-phenylquinazoline (44): Light yellow solid, 101 mg, 71% yield. m.p. 107~109 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.46 (s, 1H), 8.79~8.70 (m, 2H), 8.24 (dd, J=7.5, 1.2 Hz, 1H), 7.92 (dd, J=8.1, 1.2 Hz, 1H), 7.64~7.53 (m, 3H), 7.50~7.45 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 161.7, 160.9, 148.1, 137.5, 137.4, 131.0, 128.9, 128.7, 127.6, 126.7, 124.7, 124.3; HRMS (ESI+) calcd for C14H10BrN2 [M+H]+ 285.0022, found 285.0015.
7-Methyl-2-phenylquinazoline (45): Light yellow solid, 98 mg, 93% yield. m.p. 130~132 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.40 (s, 1H), 8.82~8.32 (m, 2H), 7.88 (d, J=0.7 Hz, 1H), 7.81 (d, J=8.3 Hz, 1H), 7.61~7.50 (m, 3H), 7.44 (dd, J=8.3, 1.4 Hz, 1H), 2.62 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.1, 159.8, 151.0, 145.1, 138.2, 130.4, 129.5, 128.6, 128.5, 127.5, 126.7, 121.8, 22.3; HRMS (ESI+) calcd for C15H13N2 [M+H]+ 211.1073, found 211.1077.
7-Methoxy-2-phenylquinazoline (46): Light yellow solid, 106 mg, 90% yield. m.p. 119~121 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.31 (s, 1H), 8.71~8.43 (m, 2H), 7.80 (d, J=8.9 Hz, 1H), 7.58~7.48 (m, 3H), 7.38 (d, J=2.4 Hz, 1H), 7.24 (dd, J=8.9, 2.4 Hz, 1H), 4.02 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 164.3, 161.5, 158.9, 153.1, 138.2, 130.5, 128.6, 128.5, 128.3, 120.7, 119.2, 106.2, 55.8; HRMS (ESI+) calcd for C15H13N2O [M+H]+ 237.1022, found 237.1024.
7-Fluoro-2-phenylquinazoline (47): Light yellow solid, 62 mg, 55% yield. m.p. 137~139 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.45 (s, 1H), 8.71~8.55 (m, 2H), 7.97 (dd, J=8.9, 5.9 Hz, 1H), 7.73 (dd, J=9.8, 2.4 Hz, 1H), 7.65~7.48 (m, 3H), 7.46~7.33 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 165.9 (d, J=255.9 Hz), 161.8, 159.9, 152.4 (d, J=14.0 Hz), 137.6, 130.9, 129.7 (d, J=10.8 Hz), 128.7, 128.67, 120.8, 117.9 (d, J=25.7 Hz), 112.4 (d, J=20.6 Hz); HRMS (ESI+) calcd for C14H17FN2 [M+H]+ 225.0823, found 225.0818.
7-Chloro-2-phenylquinazoline (48): Light yellow solid, 95 mg, 79% yield. m.p. 200~202 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.43 (s, 1H), 8.67~8.43 (m, 2H), 8.09 (d, J=1.9 Hz, 1H), 7.85 (d, J=8.6 Hz, 1H), 7.68~7.49 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 161.8, 160.1, 151.2, 140.3, 137.5, 130.9, 128.7, 128.6, 128.4, 128.3, 127.7, 121.9; HRMS (ESI+) calcd for C14H17ClN2 [M+H]+ 241.0527, found 241.0526.
7-Bromo-2-phenylquinazoline (49): Light yellow solid, 119 mg, 84% yield. m.p. 167~169 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.43 (s, 1H), 8.70~8.51 (m, 2H), 8.29 (d, J=1.7 Hz, 1H), 7.78 (d, J=8.6 Hz, 1H), 7.70 (dd, J=8.6, 1.8 Hz, 1H), 7.62~7.49 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.7, 160.3, 151.3, 137.5, 131.1, 130.9, 130.9, 128.9, 128.7, 128.6, 128.2, 122.1; HRMS (ESI+) calcd for C14H10- BrN2 [M+H]+ 285.0022, found 285.0026.
2-Phenyl-7-(trifluoromethyl)quinazoline (50): Yellow solid, 96 mg, 70% yield. m.p. 149~151 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.56 (s, 1H), 8.76~8.55 (m, 2H), 8.42 (d, J=0.7 Hz, 1H), 8.07 (d, J=8.5 Hz, 1H), 7.81 (d, J=1.5 Hz, 1H), 7.68~7.53 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 162.1, 160.6, 150.1, 137.2, 135.5 (q, J=32.9 Hz), 131.2, 128.7, 128.7, 128.3, 126.6 (q, J=4.4 Hz), 124.5, 123.4 (q, J=273.0 Hz), 122.9 (q, J=3.1 Hz); HRMS (ESI+) calcd for C15H10F3N2 [M+H]+ 275.0791, found 275.0794.
6-Methyl-2-phenylquinazoline (51): Light yellow solid, 98 mg, 93% yield. m.p. 106~108 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.39 (s, 1H), 8.70~8.54 (m, 2H), 8.00 (d, J=8.6 Hz, 1H), 7.79~7.70 (m, 1H), 7.71~7.64 (m, 1H), 7.62~7.45 (m, 3H), 2.58 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 160.4, 159.7, 149.3, 138.1, 137.4, 136.4, 130.4, 128.6, 128.4, 128.2, 125.8, 123.6, 21.6; HRMS (ESI+) calcd for C15H13N2 [M+H]+ 211.1073, found 211.1080.
6-Methoxy-2-phenylquinazoline (52): Light yellow solid, 107 mg, 91% yield. m.p. 84~86 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.37 (s, 1H), 8.73~8.50 (m, 2H), 8.01 (d, J=9.2 Hz, 1H), 7.59~7.44 (m, 4H), 7.15 (d, J=2.8 Hz, 1H), 3.97 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 159.4, 158.8, 158.2, 147.0, 138.1, 130.1, 130.1, 128.6, 128.2, 127.1, 124.4, 103.9, 55.7; HRMS (ESI+) calcd for C15H13- N2O [M+H]+ 237.1022, found 237.1027.
6-Fluoro-2-phenylquinazoline (53): Light yellow solid, 62 mg, 55% yield. m.p. 149~151 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.47 (s, 1H), 8.69~8.59 (m, 2H), 8.15 (dd, J=9.2, 5.0 Hz, 1H), 7.78~7.63 (m, 1H), 7.62~7.46 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 160.4 (d, J=251.1 Hz), 159.8, 159.7, 147.9, 137.7, 131.3 (d, J=8.5 Hz), 130.7, 128.6, 128.4, 124.5 (d, J=25.9 Hz), 123.9 (d, J=9.3 Hz), 110.1 (d, J=21.9 Hz); HRMS (ESI+) calcd for C14H17FN2 [M+H]+ 225.0823, found 225.0820.
6-Chloro-2-phenylquinazoline (54): Light yellow solid, 82 mg, 68% yield. m.p. 135~137 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.42 (s, 1H), 8.62 (dd, J=7.6, 2.2 Hz, 2H), 8.05 (d, J=9.0 Hz, 1H), 7.92 (d, J=2.1 Hz, 1H), 7.85 (dd, J=9.0, 2.3 Hz, 1H), 7.61~7.49 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.3, 159.5, 149.2, 137.6, 135.0, 132.7, 130.8, 130.4, 128.7, 128.5, 125.8, 123.9; HRMS (ESI+) calcd for C14H17ClN2 [M+H]+ 241.0527, found 241.0528.
6-Bromo-2-phenylquinazoline (55): Yellow solid, 122 mg, 86% yield. m.p. 133~135 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.41 (s, 1H), 8.74~8.54 (m, 2H), 8.12~8.07 (m, 1H), 7.98 (d, J=1.2 Hz, 2H), 7.69~7.43 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.3, 159.4, 149.4, 137.6, 137.5, 130.9, 130.4, 129.2, 128.7, 128.6, 124.4, 120.7; HRMS (ESI+) calcd for C14H10BrN2 [M+H]+ 285.0022, found 285.0029.
2-Phenyl-6-(trifluoromethyl)quinazoline (56): Light yellow solid, 79 mg, 58% yield. m.p. 154~156 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.58 (s, 1H), 8.67 (dd, J=6.8, 3.0 Hz, 2H), 8.34~8.18 (m, 2H), 8.09 (dd, J=8.9, 1.9 Hz, 1H), 7.63~7.52 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 162.8, 161.2, 151.9, 137.2, 131.3, 129.9, 129.7 (q, J=2.9 Hz), 129.6~129.1 (m), 128.9, 128.7, 125.2 (q, J=4.3 Hz), 123.7 (q, J=272.5 Hz), 122.4; HRMS (ESI+) calcd for C15H10F3N2 [M+H]+ 275.0791, found 275.0797.
5-Methoxy-2-phenylquinazoline (57): Light yellow solid, 85 mg, 72% yield. m.p. 151~153 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.82 (s, 1H), 8.63 (dd, J=7.9, 1.6 Hz, 2H), 7.84~7.76 (m, 1H), 7.65 (d, J=8.5 Hz, 1H), 7.61~7.47 (m, 3H), 6.89 (d, J=7.8 Hz, 1H), 4.06 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.4, 156.1, 156.1, 151.6, 138.1, 134.6, 130.5, 128.6, 120.4, 115.9, 105.1, 55.8; HRMS (ESI+) calcd for C15H13N2O [M+H]+ 237.1022, found 237.1024.
5-Fluoro-2-phenylquinazoline (58): Light yellow solid, 55 mg, 49% yield. m.p. 101~103 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.78 (s, 1H), 8.70~8.58 (m, 2H), 7.95 (d, J=8.5 Hz, 1H), 7.92~7.82 (m, 1H), 7.63~7.53 (m, 3H), 7.36~7.27 (m, 1H); 13C NMR (101 MHz, CDCl3) δ: 161.6, 158.3 (d, J=254.5 Hz), 154.9 (d, J=3.6 Hz), 151.3, 137.3, 134.3 (d, J=9.1 Hz), 131.1, 128.8, 128.7, 124.5 (d, J=4.5 Hz), 116.6 (d, J=33.0 Hz), 111.1 (d, J=18.6 Hz); HRMS (ESI+) calcd for C14H17FN2 [M+H]+ 225.0823, found 225.0819.
5-Chloro-2-phenylquinazoline (59): Light yellow solid, 60 mg, 50% yield. m.p. 99~101 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.86 (s, 1H), 8.73~8.57 (m, 2H), 8.08~7.99 (m, 1H), 7.82 (dd, J=8.5, 7.6 Hz, 1H), 7.64 (dd, J=7.5, 0.9 Hz, 1H), 7.61~7.52 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.6, 157.8, 151.8, 137.4, 133.9, 131.9, 131.0, 128.7, 128.7, 127.8, 127.2, 121.4; HRMS (ESI+) calcd for C14H17ClN2 [M+H]+ 241.0527, found 241.0526.
5-Bromo-2-phenylquinazoline (60): Light yellow solid, 85 mg, 60% yield. m.p. 105~107 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 9.79 (s, 1H), 8.69~8.62 (m, 2H), 8.07 (dd, J=8.5, 1.2 Hz, 1H), 7.84 (dd, J=7.5, 1.1 Hz, 1H), 7.80~7.71 (m, 1H), 7.62~7.44 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.6, 160.2, 152.0, 137.3, 134.3, 131.0, 130.8, 128.7, 128.7, 128.5, 122.6, 121.7; HRMS (ESI+) calcd for C14H10BrN2 [M+H]+ 285.0022, found 285.0026.
6,7-Dimethoxy-2-phenylquinazoline (61): Light yellow solid, 126 mg, 95% yield. m.p. 176~178 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.23 (s, 1H), 8.65~8.45 (m, 2H), 7.61~7.45 (m, 3H), 7.39 (s, 1H), 7.11 (s, 1H), 4.09 (s, 3H), 4.05 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 159.9, 157.1, 156.2, 150.3, 148.6, 138.3, 130.1, 128.5, 128.1, 119.4, 106.8, 103.9, 56.4, 56.2; HRMS (ESI+) calcd for C16H15N2O2 [M+H]+ 267.1128, found 267.1129.
6,8-Dichloro-2-phenylquinazoline (62): Light yellow solid, 105 mg, 77% yield. m.p. 132~134 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.40 (s, 1H), 8.78~8.59 (m, 2H), 7.97 (d, J=2.2 Hz, 1H), 7.83 (d, J=2.2 Hz, 1H), 7.66~7.48 (m, 3H); 13C NMR (101 MHz, CDCl3) δ: 161.6, 159.8, 146.0, 137.1, 134.6, 134.4, 132.2, 131.3, 128.8, 128.7, 124.7, 124.5; HRMS (ESI+) calcd for C14H9Cl2N2 [M+H]+ 275.0137, found 275.0134.
6-Phenyl-[1,3]dioxolo[5-g]quinazoline (63): Light yellow solid, 83 mg, 66% yield. m.p. 174~176 ℃; 1H NMR (400 MHz, CDCl3) δ: 9.17 (s, 1H), 8.66~8.37 (m, 2H), 7.72~7.39 (m, 3H), 7.35 (s, 1H), 7.11 (s, 1H), 6.15 (s, 2H); 13C NMR (101 MHz, CDCl3) δ: 160.0, 157.4, 154.1, 150.2, 148.2, 138.1, 130.2, 128.5, 128.1, 120.7, 104.9, 102.1, 101.8; HRMS (ESI+) calcd for C15H11N2O2 [M+H]+ 251.0815, found 251.0811.
Supporting Information Detailed experimental procedures, characterization data, and NMR spectra of products. The Supporting Information is available free of charge via the Internet at
http://sioc-journal.cn.