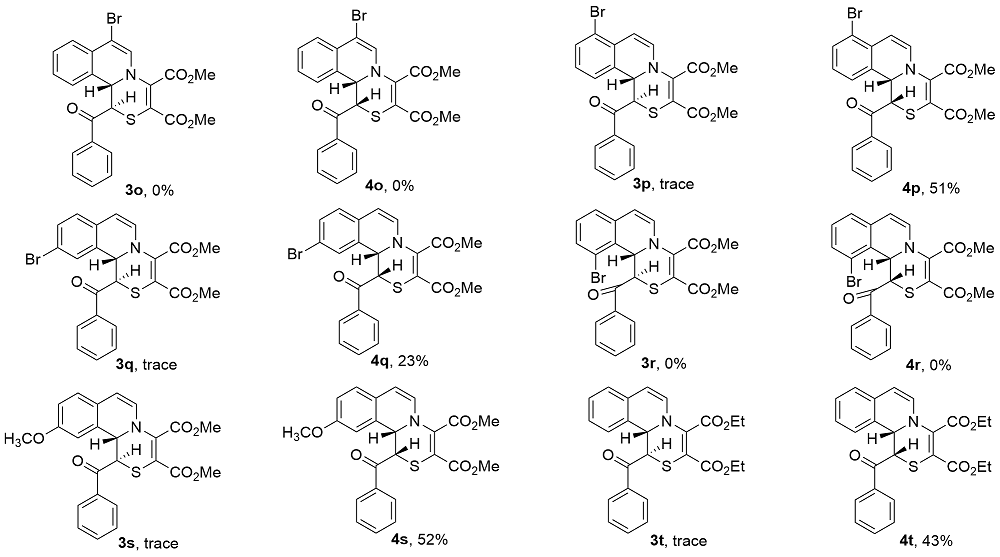
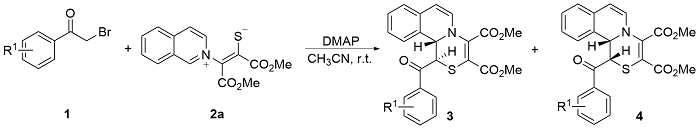
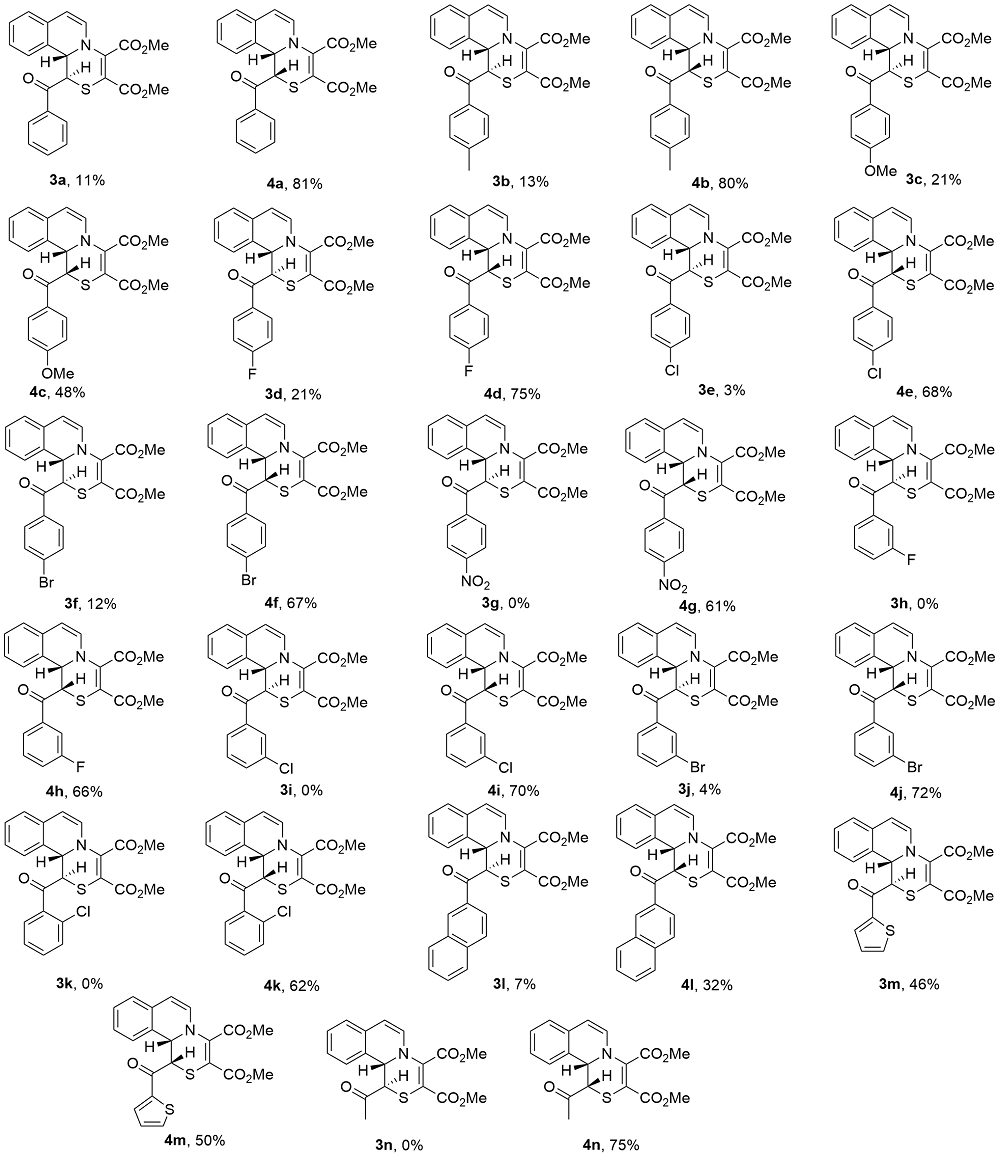
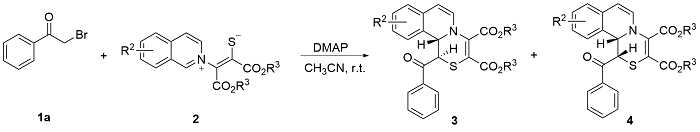
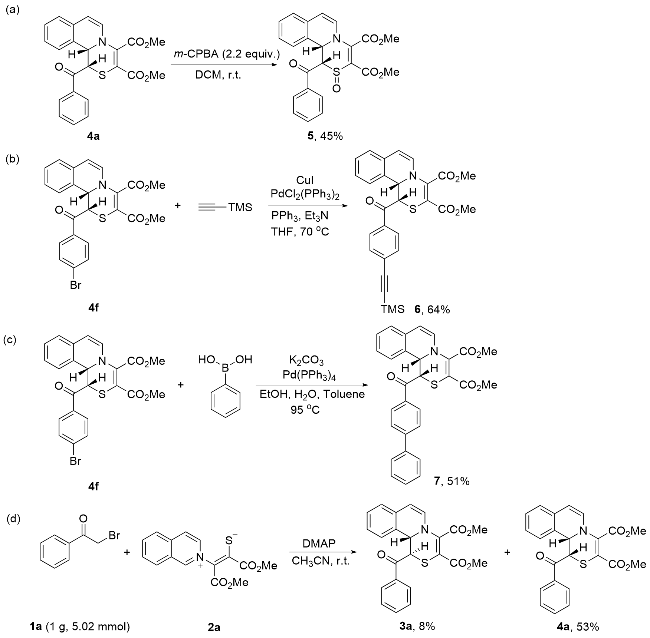
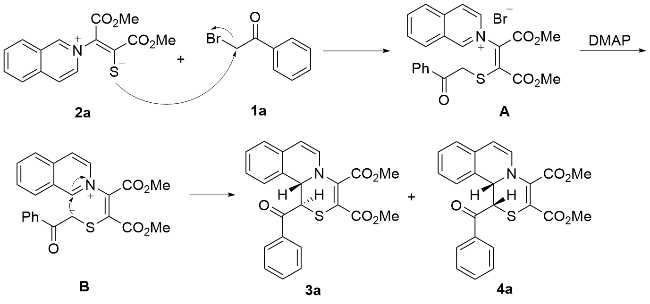
Under Ar atmosphere, substrates 1 (0.25 mmol) and 2 (0.75 mmol) were added to an oven-dried round bottle flask with a magnetic stirring bar, followed by the addition of CH3CN (2 mL) and DMAP (0.50 mmol). The stirring was maintained at room temperature until completion of the reaction (the reaction was monitored by TLC plate). The residue was purified by a flash column chromatography on silica gel (ethyl acetate/petroleum ether, V:V=1:2) to yield 3 and 4.
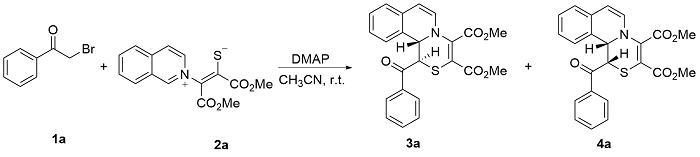
Dimethyl-1-benzoyl-1,11b-dihydro[1,4]thiazino[3,4-a]- isoquinoline-3,4-dicarboxylate (3a): Yellow solid, 11 mg, 11% yield. m.p. 189~190 ℃; 1H NMR (400 MHz, DMSO- d6) δ: 8.02 (d, J=7.6 Hz, 2H), 7.59 (t, J=7.2 Hz, 1H), 7.53 (d, J=7.6 Hz, 1H), 7.45 (t, J=8.0 Hz, 2H), 7.23~7.14 (m, 3H), 6.65 (d, J=7.2 Hz, 1H), 6.24 (d, J=7.6 Hz, 1H), 5.59 (d, J=3.2 Hz, 1H), 4.71 (d, J=3.2 Hz, 1H), 3.83 (s, 3H), 3.59 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 191.9, 163.1, 140.5, 134.9, 133.4, 131.9, 129.8, 129.2, 128.9, 128.5, 128.1, 126.8, 125.1, 124.5, 110.5, 58.4, 53.3, 52.5, 42.4; IR (KBr) ν: 2951, 1741, 1703, 1663, 1593, 1553, 1432, 1358, 1303, 1240, 1204, 1074, 1002, 928, 840, 777, 689 cm-1; HRMS (ESI-TOF) calcd for C23H20NO5S [M+H]+ 422.1057, found 422.1065.
Dimethyl-1-benzoyl-1,11b-dihydro[1,4]thiazino[3,4-a]- isoquinoline-3,4-dicarboxylate (4a): Yellow solid, 85 mg, 81% yield. m.p. 192~193 ℃; 1H NMR (400 MHz, DMSO- d6) δ: 7.78 (d, J=7.6 Hz, 2H), 7.52 (t, J=7.2 Hz, 2H), 7.37 (t, J=8.0 Hz, 2H), 7.11 (t, J=7.2 Hz, 1H), 7.05 (t, J=7.2 Hz, 1H), 6.82 (d, J=7.2 Hz, 1H), 6.27 (d, J=8.0 Hz, 1H), 6.01 (s, 1H), 5.45 (d, J=7.6 Hz, 1H), 5.33 (s, 1H), 3.83 (s, 3H), 3.67 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 193.9, 164.6, 164.3, 139.0, 136.5, 133.7, 130.0, 129.3, 129.0, 128.9, 128.8, 128.8, 127.2, 127.0, 125.2, 103.9, 102.4, 59.2, 53.7, 53.0, 44.7; IR (KBr) ν: 2951, 1733, 1698, 1665, 1638, 1555, 1432, 1420, 1339, 1239, 1202, 1137, 1072, 1002, 839, 778, 699 cm-1; HRMS (ESI-TOF) calcd for C23H20NO5S [M+H]+ 422.1057, found 422.1075.
Dimethyl-1-(4-methylbenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (3b): Purified by flash chromatography on silica gel (ethyl acetate/petroleum ether, V:V=1:2). Yellow solid, 13 mg, 13% yield. m.p. 135~136 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.91 (d, J=8.0 Hz, 2H), 7.49 (d, J=7.6 Hz, 1H), 7.25 (d, J=8.0 Hz, 2H), 7.19~7.12 (m, 3H), 6.63 (d, J=7.6 Hz, 1H), 6.21 (d, J=7.6 Hz, 1H), 5.53 (d, J=3.6 Hz, 1H), 4.69 (d, J=3.2 Hz, 1H), 3.82 (s, 3H), 3.59 (s, 3H), 2.34 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 191.6, 164.3, 163.1, 144.0, 140.1, 132.5, 131.8, 129.8, 129.1, 129.1, 128.1, 126.8, 125.3, 124.5, 110.1, 58.5, 53.3, 52.5, 42.5, 21.2; IR (KBr) ν: 2950, 1739, 1699, 1663, 1640, 1551, 1433, 1420, 1304, 1228, 1203, 1075, 1003, 929, 846, 774, 696 cm-1; HRMS (ESI- TOF) calcd for C24H22NO5S [M+H]+ 436.1213, found 436.1233.
Dimethyl-1-(4-methylbenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4b): Yellow solid, 82 mg, 80% yield. m.p. 152~153 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.69 (d, J=8.4 Hz, 2H), 7.52 (d, J=7.2 Hz, 1H), 7.19 (d, J=8.0 Hz, 2H), 7.13~7.04 (m, 2H), 6.84 (dd, J=7.2, 1.2 Hz, 1H), 6.27 (d, J=7.6 Hz, 1H), 5.96 (d, J=2.0 Hz, 1H), 5.45 (d, J=8.0 Hz, 1H), 5.32 (d, J=1.6 Hz, 1H), 3.82 (s, 3H), 3.67 (s, 3H), 2.30 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 192.9, 164.2, 163.9, 143.8, 138.6, 133.6, 129.5, 129.1, 129.0, 128.6, 128.5, 128.3, 126.7, 126.6, 124.8, 103.4, 101.6, 58.7, 53.3, 52.6, 43.7, 21.1; IR (KBr) ν: 2950, 1733, 1703, 1668, 1579, 1561, 1436, 1339, 1266, 1243, 1209, 1071, 1004, 843, 779, 700, 529 cm-1; HRMS (ESI-TOF) calcd for C24H22NO5S [M+H]+ 436.1213, found 436.1208.
Dimethyl-1-(4-methoxybenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (3c): Yellow solid, 21 mg, 21% yield. m.p. 151~152 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.97 (d, J=8.8 Hz, 2H), 7.40 (d, J=8.0 Hz, 1H), 7.21~7.08 (m, 3H), 6.95 (d, J=8.8 Hz, 2H), 6.59 (d, J=7.6 Hz, 1H), 6.16 (d, J=7.6 Hz, 1H), 5.47 (d, J=4.0 Hz, 1H), 4.66 (d, J=4.0 Hz, 1H), 3.82 (d, J=2.4 Hz, 6H), 3.62 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 190.7, 164.4, 163.3, 163.1, 139.4, 131.5, 131.4, 129.9, 128.7, 128.2, 128.0, 126.6, 125.7, 124.5, 113.8, 109.3, 58.8, 55.6, 53.2, 52.5, 42.9; IR (KBr) ν: 2954, 1740, 1700, 1660, 1598, 1550, 1421, 1327, 1308, 1250, 1206, 1179, 1144, 1703, 994, 852, 771, 692 cm-1; HRMS (ESI-TOF) calcd for C24H22- NO6S [M+H]+ 452.1162, found 452.1167.
Dimethyl-1-(4-methoxybenzoyl)-1,11b-dihydro[1,4]-thiazino[3,4-a]isoquinoline-3,4-dicarboxylate (4c): Yellow solid, 47 mg, 48% yield. m.p. 211~212 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.77 (d, J=8.8 Hz, 2H), 7.51 (d, J=7.6 Hz, 1H), 7.13~7.04 (m, 2H), 6.89 (d, J=8.8 Hz, 2H), 6.84 (d, J=7.2 Hz, 1H), 6.26 (d, J=7.6 Hz, 1H), 5.93 (d, J=2.0 Hz, 1H), 5.44 (d, J=8.0 Hz, 1H), 5.30 (s, 1H), 3.80 (d, J=15.6 Hz, 6H), 3.67 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 191.8, 164.2, 163.9, 163.1, 130.8, 138.5, 129.6, 129.1, 129.0, 128.6, 128.3, 126.7, 126.6, 124.8, 113.7, 103.3, 101.9, 58.8, 55.6, 53.2, 52.6, 43.7; IR (KBr) ν: 2950, 1735, 1742, 1663, 1600, 1560, 1513, 1417, 1340, 1244, 1206, 1171, 1142, 996, 833, 779, 694 cm-1; HRMS (ESI-TOF) calcd for C24H22NO6S [M+H]+ 452.1162, found 452.1165.
Dimethyl-1-(4-fluorobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (3d): Yellow solid, 21 mg, 21% yield. m.p. 158~159 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.13 (dd, J=8.4, 5.6 Hz, 2H), 7.54 (d, J=7.6 Hz, 1H), 7.29 (t, J=8.8 Hz, 2H), 7.23~7.14 (m, 3H), 6.65 (d, J=7.6 Hz, 1H), 6.24 (d, J=7.2 Hz, 1H), 5.61 (d, J=3.2 Hz, 1H), 4.70 (d, J=3.2 Hz, 1H), 3.83 (s, 3H), 3.59 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 190.5, 164.4, 163.8, 163.1, 132.1, 132.0, 131.9, 131.7, 131.7, 129.8, 129.2, 128.2, 126.8, 125.1, 124.6, 115.7, 115.5, 110.6, 58.4, 53.3, 52.5, 42.4; 19F NMR (471 MHz, DMSO- d6) δ: -105.36; IR (KBr) ν: 2950, 1719, 1700, 1667, 1594, 1556, 1435, 1354, 1248, 1206, 1142, 1008, 929, 837, 771, 723, 610 cm-1; HRMS (ESI-TOF) calcd for C23H19FNO5S [M+H]+ 440.0962, found 440.0977.
Dimethyl-1-(4-fluorobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate(4d): Yellow solid, 76 mg, 75% yield. m.p. 150~151 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.84 (dd, J=9.2, 5.6 Hz, 2H), 7.50 (d, J=7.6 Hz, 1H), 7.18 (t, J=8.8 Hz, 2H), 7.11 (td, J=7.6, 1.2 Hz, 1H), 7.08 - 7.04 (m, 1H), 6.81~6.79 (m, 1H), 6.26 (d, J=8.0 Hz, 1H), 6.00 (d, J=2.0 Hz, 1H), 5.42 (d, J=8.0 Hz, 1H), 5.32 (d, J=1.6 Hz, 1H), 3.82 (s, 3H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 192.3, 164.2, 163.8, 163.6, 138.4, 133.0, 131.5, 131.4, 128.8, 128.7, 128.4, 126.8, 126.7, 124.7, 115.6, 115.4, 103.4, 102.8, 58.9, 53.3, 52.6, 45.1; 19F NMR (471 MHz, DMSO-d6) δ: -105.26; IR (KBr) ν: 2950, 1727, 1710, 1675, 1556, 1431, 1339, 1246, 1208, 1162, 1070, 1006, 952, 841, 775, 702, 608 cm-1; HRMS (ESI-TOF) calcd for C23H19FNO5S [M+H]+ 440.0962, found 440.0987.
Dimethyl-1-(4-chlorobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (3e): Yellow solid, 3 mg, 3% yield. m.p. 166~167 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.08 (d, J=8.8 Hz, 2H), 7.60 (d, J=7.6 Hz, 1H), 7.54 (d, J=8.4 Hz, 2H), 7.24~7.20 (m, 3H), 6.67 (d, J=7.6 Hz, 1H), 6.28 (d, J=7.2 Hz, 1H), 5.63 (d, J=2.8 Hz, 1H), 4.72 (d, J=2.8 Hz, 1H), 3.83 (s, 3H), 3.57 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 190.6, 164.3, 163.0, 141.1, 138.3, 133.6, 132.1, 130.9, 129.7, 129.4, 128.6, 128.1, 126.9, 124.8, 124.6, 111.1, 58.2, 53.3, 52.4, 42.1; IR (KBr) ν:2945, 1722, 1704, 1666, 1556, 1435, 1354, 1243, 1206, 1139, 1092, 1006, 929, 831, 779, 716, 542 cm-1; HRMS (ESI-TOF) calcd for C23H19ClNO5S [M+H]+ 456.0667, found 456.0694.
Dimethyl-1-(4-chlorobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4e): Yellow solid, 66 mg, 68% yield. m.p. 183~184 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.76 (d, J=8.4 Hz, 2H), 7.50 (d, J=7.2 Hz, 1H), 7.42 (d, J=8.4 Hz, 2H), 7.09 (dt, J=23.2, 7.2 Hz, 2H), 6.80 (d, J=7.2 Hz, 1H), 6.26 (d, J=7.6 Hz, 1H), 5.99 (s, 1H), 5.42 (d, J=7.6 Hz, 1H), 5.32 (s, 1H), 3.82 (s, 3H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 192.7, 164.2, 163.8, 138.4, 138.2, 134.8, 130.2, 129.5, 128.7, 128.6, 128.5, 128.4, 126.8, 126.7, 124.7, 103.4, 102.8, 58.8, 53.3, 52.6, 45.2; IR (KBr) ν: 2948, 1742, 1704, 1664, 1562, 1435, 1333, 1244, 1204, 1139, 1092, 1073, 998, 835, 779, 523 cm-1; HRMS (ESI-TOF) calcd for C23H19ClNO5S [M+H]+ 456.0667, found 456.0676.
Dimethyl-1-(4-bromobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (3f): Yellow solid, 11 mg, 12% yield. m.p. 173~174 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.00 (d, J=8.4 Hz, 2H), 7.68 (d, J=8.8 Hz, 2H), 7.61 (d, J=7.2 Hz, 1H), 7.25~7.20 (m, 3H), 6.67 (d, J=7.2 Hz, 1H), 6.28 (d, J=7.2 Hz, 1H), 5.63 (d, J=2.8 Hz, 1H), 4.72 (d, J=2.8 Hz, 1H), 3.83 (s, 3H), 3.57 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 190.8, 164.3, 163.0, 141.2, 133.9, 132.1, 131.6, 131.0, 129.7, 129.4, 128.1, 127.5, 126.9, 124.8, 124.6, 111.2, 58.1, 53.3, 52.4, 42.0; IR (KBr) ν: 2945, 1723, 1706, 1662, 1585, 1558, 1437, 1354, 1242, 1200, 1142, 1068, 1004, 929, 831, 777, 523 cm-1; HRMS (ESI-TOF) calcd for C23H19BrNO5S [M+H]+ 500.0162, found 500.0148.
Dimethyl-1-(4-bromobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4f): Yellow solid, 60 mg, 67% yield. m.p. 188~189 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.68 (d, J=8.4 Hz, 2H), 7.57 (d, J=8.8 Hz, 2H), 7.49 (d, J=7.2 Hz, 1H), 7.14~7.04 (m, 2H), 6.82~6.79 (m, 1H), 6.26 (d, J=8.0 Hz, 1H), 5.99 (d, J=2.0 Hz, 1H), 5.42 (d, J=7.6 Hz, 1H), 5.32 (d, J=1.6 Hz, 1H), 3.82 (s, 3H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 190.5, 164.4, 163.1, 140.6, 132.1, 132.0, 131.9, 131.7, 129.8, 129.2, 128.2, 126.8, 125.1, 124.6, 115.7, 115.5, 110.6, 58.4, 53.3, 52.5, 42.40; IR (KBr) ν: 2952, 1742, 1706, 1662, 1641, 1560, 1433, 1335, 1242, 1202, 1142, 1073, 998, 821, 779, 702, 581 cm-1; HRMS (ESI-TOF) calcd for C23H19BrNO5S [M+H]+ 500.0162, found 500.0175.
Dimethyl-1-(4-nitrobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4g): Yellow solid, 58 mg, 61% yield. m.p. 182~183 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.14 (d, J=8.8 Hz, 2H), 7.96 (d, J=8.8 Hz, 2H), 7.51 (d, J=7.6 Hz, 1H), 7.12 (td, J=7.6, 1.2 Hz, 1H), 7.03 (t, J=7.2 Hz, 1H), 6.76~6.74 (m, 1H), 6.27 (d, J=8.0 Hz, 1H), 6.10 (d, J=2.0 Hz, 1H), 5.42 (d, J=8.0 Hz, 1H), 5.37 (d, J=1.6 Hz, 1H), 3.83 (s, 3H), 3.69 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 192.9, 164.1, 163.7, 149.6, 140.8, 138.3, 129.8, 129.4, 128.6, 128.5, 128.5, 126.9, 126.7, 124.7, 123.4 103.6, 103.2, 58.9, 53.3, 52.7, 46.6; IR (KBr) ν: 2954, 1710, 1641, 1560, 1514, 1433, 1356, 1262, 1202, 1131, 1068, 993, 825, 766, 739, 698 cm-1; HRMS (ESI-TOF) calcd for C23H19N2O7S [M+H]+ 467.0907, found 467.0918.
Dimethyl-1-(3-fluorobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4h): Yellow solid, 67 mg, 66% yield. m.p. 181~182 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.60 (d, J=6.8 Hz, 1H), 7.55~7.50 (m, 2H), 7.44~7.36 (m, 2H), 7.13 (t, J=6.8 Hz, 1H), 7.05 (t, J=7.2 Hz, 1H), 6.80 (d, J=7.2 Hz, 1H), 6.27 (d, J=8.0 Hz, 1H), 6.01 (d, J=2.0 Hz, 1H), 5.43 (d, J=8.0 Hz, 1H), 5.34 (d, J=0.8 Hz, 1H), 3.83 (s, 3H), 3.69 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 192.6, 164.1, 163.8, 138.4, 138.3, 130.7, 129.5, 128.6, 128.4, 126.7, 126.7, 124.7, 124.6, 120.3, 120.1, 115.1, 114.9, 103.4, 102.8, 58.9, 53.3, 52.6, 45.3; 19F NMR (471 MHz, DMSO-d6) δ: -112.313; IR (KBr) ν: 2952, 1742, 1698, 1673, 1639, 1562, 1437, 1242, 1210, 1142, 1079, 1000, 929, 829, 779, 687 cm-1; HRMS (ESI-TOF) calcd for C23H19FNO5S [M+H]+ 440.0962, found 440.0975.
Dimethyl-1-(3-chlorobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4i): Yellow solid, 68 mg, 70% yield. m.p. 179~180 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.74 (s, 1H), 7.68 (d, J=8.0 Hz, 1H), 7.58 (d, J=8.0 Hz, 1H), 7.51 (d, J=7.6 Hz, 1H), 7.38 (t, J=8.0 Hz, 1H), 7.14 (t, J=7.6 Hz, 1H), 7.05 (t, J=7.6 Hz, 1H), 6.78 (d, J=7.6 Hz, 1H), 6.25 (d, J=8.0 Hz, 1H), 6.02 (d, J=2.0 Hz, 1H), 5.41 (d, J=8.0 Hz, 1H), 5.32 (s, 1H), 3.82 (s, 3H), 3.69 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 192.7, 164.1, 163.7, 138.3, 137.9, 133.4, 132.9, 130.3, 129.4, 128.6, 128.6, 128.5, 128.2, 126.9, 126.7, 126.7, 124.7, 103.4, 103.3, 58.9, 53.3, 52.6, 45.8; IR (KBr) ν: 2952, 1742, 1696, 1668, 1641, 1565, 1435, 1333, 1204, 1142, 1079, 1002, 956, 845, 779, 689 cm-1; HRMS (ESI- TOF) calcd for C23H19ClNO5S [M+H]+ 456.0667, found 456.0683.
Dimethyl-1-(3-bromobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (3j): Yellow solid, 4 mg, 4% yield. m.p. 150~151 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.28 (t, J=1.6 Hz, 1H), 8.02 (d, J=8.0 Hz, 1H), 7.68 (dd, J=8.0, 0.8 Hz, 1H), 7.68 (d, J=7.6 Hz, 1H), 7.43 (t, J=8.0 Hz, 1H), 7.25~7.20 (m, 3H), 6.69 (d, J=7.6 Hz, 1H), 6.31 (d, J=7.6 Hz, 1H), 5.66 (d, J=2.8 Hz, 1H), 4.72 (d, J=2.4 Hz, 1H), 3.83 (s, 3H), 3.57 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 190.1, 164.3, 163.0, 141.4, 136.8, 135.8, 132.2, 131.5, 130.7, 129.7, 129.6, 128.1, 127.8, 126.9, 124.6, 124.5, 121.9, 111.5, 58.1, 53.3, 52.4, 42.0; IR (KBr) ν: 2946, 1714, 1675, 1558, 1433, 1339, 1264, 1206, 1135, 1066, 927, 820, 775, 720, 664 cm-1; HRMS (ESI-TOF) calcd for C23H19BrNO5S [M+ H]+ 500.0162, found 500.0172.
Dimethyl-1-(3-bromobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4j): Yellow solid, 4 mg, 72% yield. m.p. 174~175 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.86 (s, 1H), 7.71 (t, J=7.6 Hz, 2H), 7.51 (d, J=7.2 Hz, 1H), 7.30 (t, J=7.6 Hz, 1H), 7.14 (t, J=7.2 Hz, 1H), 7.05 (t, J=7.6 Hz, 1H), 6.78 (d, J=7.2 Hz, 1H), 6.25 (d, J=8.0 Hz, 1H), 6.01 (s, 1H), 5.41 (d, J=7.6 Hz, 1H), 5.31 (s, 1H), 3.82 (s, 3H), 3.69 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 192.6, 164.1, 163.7, 138.2, 138.1, 135.8, 131.1, 130.5, 129.4, 128.6, 128.5, 128.5, 127.2, 126.7, 126.7, 124.7, 121.8, 103.4, 58.9, 53.2, 52.6, 45.8; IR (KBr) ν: 2950, 1739, 1695, 1668, 1639, 1562, 1437, 1333, 1202, 1139, 1075, 1000, 954, 845, 781, 685 cm-1; HRMS (ESI-TOF) calcd for C23H19BrNO5S [M+H]+ 500.0162, found 500.0162.
Dimethyl-1-(2-chlorobenzoyl)-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate(4k): Yellow so- lid, 61 mg, 62% yield. m.p. 124~125 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.46 (d, J=7.2 Hz, 1H), 7.41~7.39 (m, 2H), 7.22 (dd, J=4.8, 2.4 Hz, 2H), 7.10~7.01 (m, 2H), 6.92 (dd, J=7.2, 1.2 Hz, 1H), 6.30 (d, J=8.0 Hz, 1H), 5.68 (d, J=2.4 Hz, 1H), 5.54 (d, J=8.0 Hz, 1H), 5.31 (d, J=1.6 Hz, 1H), 3.84 (s, 3H), 3.71 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 196.1, 164.1, 163.8, 138.6, 137.9, 132.5, 130.3, 130.2, 129.5, 129.1, 128.5, 128.5, 128.4, 127.1, 126.8, 126.8, 124.9, 103.3, 102.5, 58.7, 53.4, 52.7, 49.4; IR (KBr) ν: 2954, 1722, 1700, 1645, 1556, 1433, 1258, 1210, 1145, 1070, 998, 895, 843, 775,743, 704 cm-1; HRMS (ESI-TOF) calcd for C23H19ClNO5S [M+H]+ 456.0667, found 456.0678.
Dimethyl-1-(2-naphthoyl)-1,11b-dihydro[1,4]thiazino-[3,4-a]isoquinoline-3,4-dicarboxylate (3l): Yellow solid, 7 mg, 7% yield. m.p. 197~198 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.80 (s, 1H), 8.06 (d, J=8.0 Hz, 1H), 8.02~7.95 (m, 3H), 7.69~7.58 (m, 3H), 7.17 (s, 3H), 6.69 (d, J=7.6 Hz, 1H), 6.27 (d, J=7.2 Hz, 1H), 5.76 (s, 1H), 4.78 (s, 1H), 3.85 (s, 3H), 3.57 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 191.7, 164.4, 163.1, 140.5, 135.0, 132.3, 132.0, 131.9, 131.1, 129.8, 129.6, 129.2, 129.0, 128.1, 127.7, 127.0, 126.8, 125.1, 124.6, 124.2, 110.4, 58.5, 53.3, 52.5, 42.6; IR (KBr) ν: 2952, 1729, 1698, 1662, 1556, 1435, 1245, 1210, 1139, 1068, 1004, 927, 864, 777, 752, 689 cm-1; HRMS (ESI-TOF) calcd for C27H22NO5S [M+H]+ 472.1213, found 472.1225.
Dimethyl-1-(2-naphthoyl)-1,11b-dihydro[1,4]thiazino-[3,4-a]isoquinoline-3,4-dicarboxylate (4l): Yellow solid, 30 mg, 32% yield. m.p. 207~208 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.44 (s, 1H), 7.93~7.90 (m, 2H), 7.87 (d, J=8.8 Hz, 1H), 7.78 (dd, J=8.4, 1.6 Hz, 1H), 7.66~7.57 (m, 3H), 7.11 (td, J=7.6, 0.8 Hz, 1H), 6.94 (t, J=7.6 Hz, 1H), 6.73 (d, J=6.8 Hz, 1H), 6.29 (d, J=7.6 Hz, 1H), 6.15 (d, J=2.0 Hz, 1H), 5.43 (d, J=8.0 Hz, 1H), 5.39 (d, J=1.6 Hz, 1H), 3.84 (s, 3H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 193.4, 164.2, 163.9, 138.5, 134.8, 133.3, 131.8, 130.9, 129.6, 129.5, 128.9, 128.9, 128.7, 128.3, 128.2, 127.6, 127.0, 126.8, 126.7, 124.7, 123.6, 103.4, 102.7, 59.0, 53.3, 52.6, 44.9; IR (KBr) ν: 2948, 1735, 1697, 1664, 1554, 1437, 1370, 1243, 1204, 1131, 1072, 1004, 929, 812, 781, 697 cm-1; HRMS (ESI-TOF) calcd for C27H22N- O5S [M+H]+ 472.1213, found 472.1224.
Dimethyl-1-(thiophene-2-carbonyl)-1,11b-dihydro[1,4]-thiazino[3,4-a]isoquinoline-3,4-dicarboxylate (3m): Yellow solid, 48 mg, 46% yield. m.p. 160~161 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 7.96 (d, J=12.5 Hz, 2H), 7.23 (d, J=7.0 Hz, 2H), 7.13~7.11 (m, 3H), 7.04 (d, J=5.0 Hz, 2H), 6.52 (d, J=7.0 Hz, 1H), 6.07 (d, J=7.0 Hz, 1H), 5.30 (s, 1H), 4.64 (s, 1H), 3.82 (s, 3H), 3.67 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 185.5, 164.3, 163.0, 142.6, 138.2, 136.4, 135.0, 130.9, 129.8, 128.8, 128.3, 127.9, 126.5, 126.2, 124.5, 108.0, 59.1, 53.2, 52.7, 45.4; IR (KBr) ν: 2948, 1737, 1698, 1645, 1562, 1412, 1244, 1206, 1139, 1072, 1000, 845, 779, 739, 687 cm-1; HRMS (ESI-TOF) calcd for C21H18NO5S2 [M+H]+ 428.0621, found 428.0637.
Dimethyl-1-(thiophene-2-carbonyl)-1,11b-dihydro-[1,4]thiazino[3,4-a]isoquinoline-3,4-dicarboxylate (4m): Yellow solid, 52 mg, 50% yield. m.p. 174~175 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 7.91 (d, J=4.0 Hz, 1H), 7.70 (s, 1H), 7.54 (d, J=7.5 Hz, 1H), 7.14 (t, J=7.0 Hz, 1H), 7.08 (t, J=7.0 Hz, 1H), 7.04 (s, 1H), 6.84 (d, J=7.0 Hz, 1H), 6.24 (d, J=7.5 Hz, 1H), 5.79 (s, 1H), 5.42 (d, J=7.5 Hz, 1H), 5.29 (s, 1H), 3.83 (s, 3H), 3.70 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 186.8, 164.2, 163.8, 143.6, 138.5, 136.0, 134.4, 129.5, 128.8, 128.6, 128.6, 128.5, 127.0, 126.7, 124.8, 103.3, 103.1, 59.0, 53.3, 52.7, 45.7; IR (KBr) ν: 2945, 1735, 1695, 1641, 1556, 1410, 1354, 1239, 1204, 1141, 1070, 1000, 833, 779, 737, 689 cm-1; HRMS (ESI-TOF) calcd for C21H18NO5S2 [M+H]+ 428.0621, found 428.0635.
Dimethyl-1-acetyl-1,11b-dihydro[1,4]thiazino[3,4-a]iso- quinoline-3,4-dicarboxylate (4n): Yellow solid, 98 mg, 75% yield. m.p. 178~179 ℃; 1H NMR (400 MHz, DMSO- d6) δ: 7.44~7.42 (m, 1H), 7.22 (t, J=4.0 Hz, 2H), 7.05~7.03 (m, 1H), 6.22 (d, J=8.0 Hz, 1H), 5.54(d, J=7.6 Hz, 1H), 5.16 (d, J=2.0 Hz, 1H), 4.96 (d, J=2.0 Hz, 1H), 3.80 (s, 3H), 3.71 (s, 3H), 1.87 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 201.9, 164.2, 163.7, 138.1, 129.4, 128.8, 128.5, 128.5, 127.0, 126.8, 124.9, 103.6, 103.1, 58.2, 53.2, 52.6, 51.5, 30.4; IR (KBr) ν: 2950, 1729, 1706, 1639, 1558, 1433, 1243, 1212, 1143, 1072, 1010, 841, 783, 708 cm-1; HRMS (ESI-TOF) calcd for C18H18NO5S [M+H]+ 360.0900, found 360.0915.
Dimethyl-1-benzoyl-8-bromo-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate(4p): Yellow so- lid, 64 mg, 51% yield. m.p. 194~195 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 7.73 (d, J=8.0 Hz, 2H), 7.52 (d, J=7.5 Hz, 2H), 7.35 (t, J=7.5 Hz, 2H), 7.30 (d, J=8.0 Hz, 1H), 7.02 (t, J=7.5 Hz, 1H), 6.43 (d, J=8.0 Hz, 1H), 5.98 (s, 1H), 5.55 (d, J=8.0 Hz, 1H), 5.33 (s, 1H), 3.83 (s, 3H), 3.70 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 193.7, 164.0, 163.4, 137.2, 136.2, 133.3, 132.0, 131.4, 130.9, 129.1, 128.3, 127.7, 126.4, 119.0, 105.5, 100.7, 58.5, 53.2, 52.7, 46.1; IR (KBr) ν: 2948, 1731, 1706, 1554, 1435, 1262, 1208, 1133, 1072, 1002, 958, 847, 762, 704, 648 cm-1; HRMS (ESI-TOF) calcd for C23H19BrNO5S [M+H]+ 500.0162, found 500.0155.
Dimethyl-1-benzoyl-10-bromo-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4q): Yellow solid, 29 mg, 23% yield. m.p. 195~196 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.80 (d, J=7.2 Hz, 3H), 7.55 (t, J=7.6 Hz, 1H), 7.39 (t, J=7.6 Hz, 2H), 7.21 (dd, J=8.0, 1.6 Hz, 1H), 6.75 (d, J=8.0 Hz, 1H), 6.31 (d, J=8.0 Hz, 1H), 6.06 (d, J=2.0 Hz, 1H), 5.42 (d, J=8.0 Hz, 1H), 5.34 (s, 1H), 3.82 (s, 3H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 193.6, 164.1, 163.7, 138.0, 136.1, 133.4, 131.1, 129.5, 129.3, 128.9, 128.5, 128.5, 126.5, 118.9, 102.2, 58.0, 53.3, 52.7, 45.1; IR (KBr) ν: 2952, 1737, 1702, 1668, 1575, 1558, 1431, 1245, 1210, 1141, 1072, 997, 829, 762, 700, 650 cm-1; HRMS (ESI-TOF) calcd for C23H19BrNO5S [M+H]+ 500.0162, found 500.0210.
Dimethyl-1-benzoyl-9-methoxy-1,11b-dihydro[1,4]thia-zino[3,4-a]isoquinoline-3,4-dicarboxylate (4s): Yellow solid, 59 mg, 52% yield. m.p. 180~181 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.78 (d, J=7.2 Hz, 2H), 7.54 (t, J=7.2 Hz, 1H), 7.45 (d, J=8.8 Hz, 1H), 7.39 (t, J=8.0 Hz, 2H), 6.68 (dd, J=8.4, 2.8 Hz, 1H), 6.41 (d, J=2.4 Hz, 1H), 6.28 (d, J=8.0 Hz, 1H), 5.95 (d, J=2.4 Hz, 1H), 5.42 (d, J=8.0 Hz, 1H), 5.26 (d, J=2.0 Hz, 1H), 3.83 (s, 3H), 3.65 (d, J=14.4 Hz, 6H); 13C NMR (100 MHz, DMSO-d6) δ: 193.5, 164.2, 163.9, 159.0, 138.5, 136.1, 133.2, 130.8, 128.9, 128.5, 128.4, 127.8, 121.0, 112.4, 109.6, 103.4, 101.9, 58.5, 55.0, 53.2, 52.6, 44.0; IR (KBr) ν: 2949, 1735, 1701, 1666, 1554, 1500, 1440, 1350, 1242, 1207, 1033, 1001, 852, 761, 719, 684 cm-1; HRMS (ESI-TOF) calcd for C24H22NO6S [M+H]+ 452.1162, found 452.1203.
Diethyl-1-benzoyl-1,11b-dihydro[1,4]thiazino[3,4-a]iso-quinoline-3,4-dicarboxylate (4t): Yellow solid, 49 mg, 43% yield. m.p. 150~151 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.76 (d, J=7.2 Hz, 2H), 7.52 (t, J=6.4 Hz, 2H), 7.36 (t, J=8.0 Hz, 2H), 7.11 (td, J=7.2, 1.2 Hz, 1H), 7.06~7.02 (m, 1H), 6.08 (dd, J=7.6, 0.8 Hz, 1H), 6.26 (d, J=8.0 Hz, 1H), 5.97 (d, J=2.0 Hz, 1H), 5.43 (d, J=7.6 Hz, 1H), 5.29 (d, J=1.6 Hz, 1H), 4.36~4.22 (m, 2H), 4.17~4.11 (m, 2H), 1.29 (t, J=7.2 Hz, 3H), 1.19 (t, J=6.8 Hz, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 193.78, 163.8, 163.3, 138.3, 136.1, 133.3, 129.6, 128.9, 128.6, 128.5, 128.4, 128.4, 126.8, 126.7, 124.7, 103.3, 103.1, 62.3, 61.4, 58.7, 45.0, 14.1, 13.7; IR (KBr) ν: 2979, 1720, 1695, 1675, 1554, 1448, 1350, 1243, 1202, 1141, 1075, 994, 954, 847, 766, 689 cm-1; HRMS (ESI-TOF) calcd for C25H24NO5S [M+H]+ 450.1370, found 450.1385.