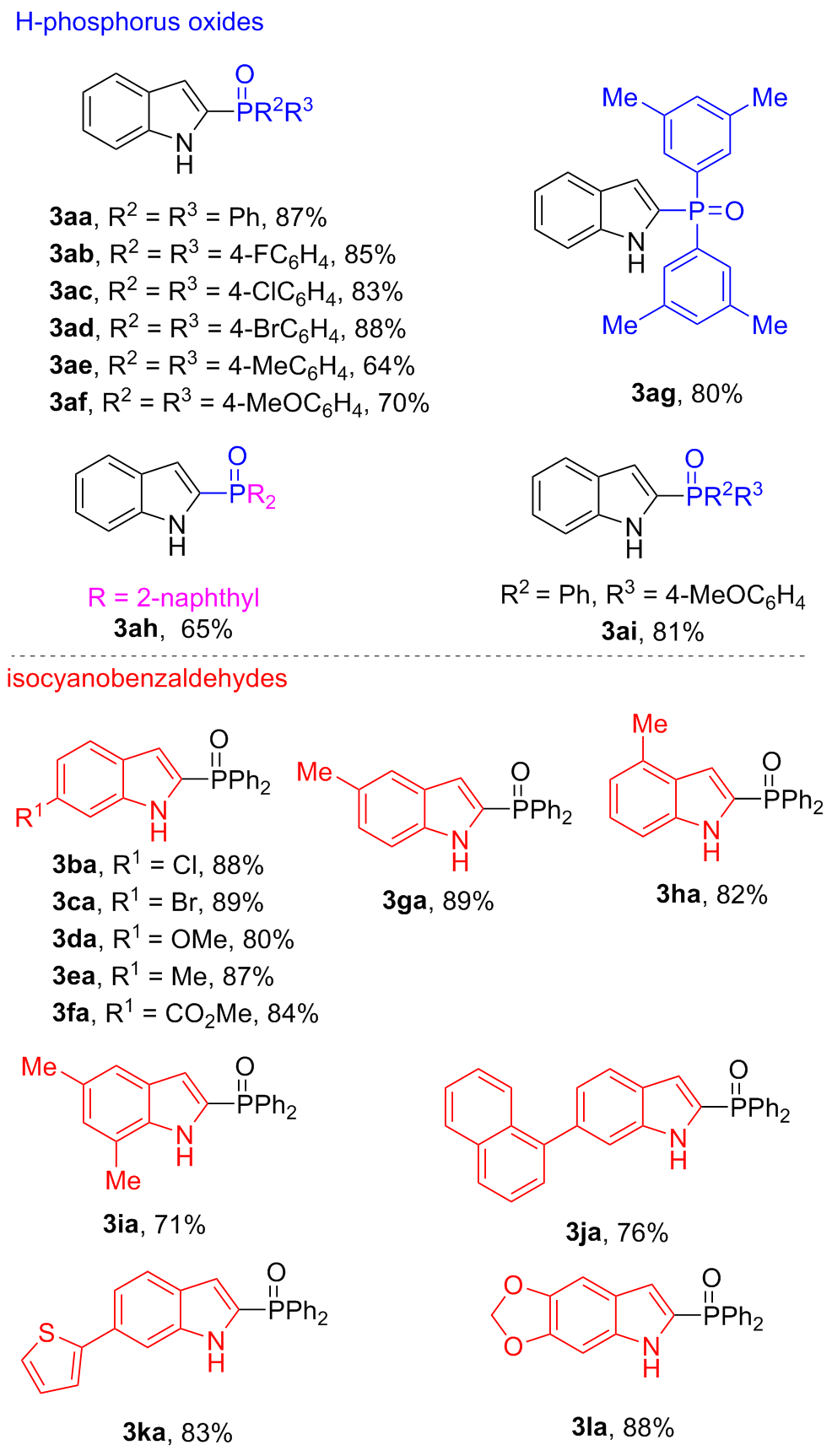
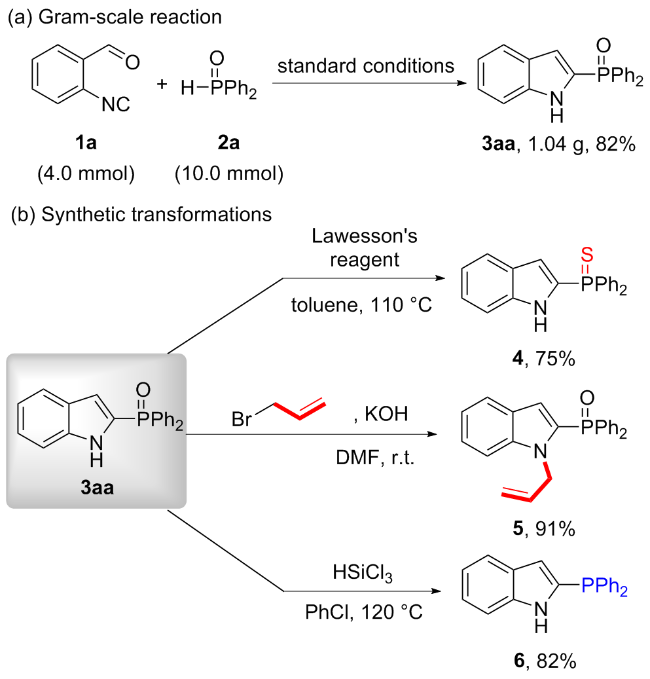
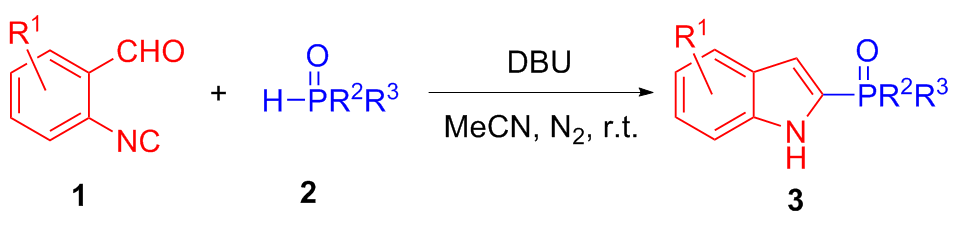In a flame-dried 10.0 mL Schlenk tube equipped with a magnetic stir bar was charged sequentially with 1a (0.2 mmol, 1.0 equiv.) and 2a (0.5 mmol, 2.5 equiv.), followed by the addition of anhydrous MeCN (1.0 mL). To the resulting mixture was added DBU (0.5 mmol, 2.5 equiv.) under nitrogen atmosphere. Then the mixture was stirred at room temperature for 12 h under nitrogen atmosphere until the reaction was completed, as monitored by TLC analysis. The residue was purified by silica gel chromatography with petroleum ether/ethyl acetate (V∶V=1∶1) to afford 2-phosphinoyl indoles 3aa (87% yield). Compounds 3ab~3la were prepared through the same procedure.
(1H-Indol-2-yl)diphenylphosphine oxide (3aa): White solid, 87% yield. m.p. 193~193.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.87 (s, 1H), 7.71~7.40 (m, 12H), 7.25 (t, J=6.7 Hz, 1H), 7.09 (t, J=6.9 Hz, 1H), 6.54 (s, 1H); 13C NMR (101 MHz,CDCl3) δ: 139.8~139.6 (m), 132.20 (d, J=2.6 Hz), 132.19 (d, J=110.0 Hz), 131.98 (d, J=10.5 Hz), 128.6 (d, J=12.6 Hz), 128.3 (d, J=124.5 Hz), 127.4 (d, J=12.6 Hz), 124.2, 121.5, 120.3, 113.2 (d, J=16.0 Hz), 112.9~112.8 (m); 31P NMR (162 MHz, CDCl3) δ: 22.38; HRMS (ESI) calcd for C20H16NOPNa [M+Na]+ 340.0862, found 340.0870.
Bis(4-fluorophenyl)(1H-indol-2-yl)phosphine oxide (3ab): White solid, 85% yield. m.p. 238.4~238.7 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.87 (s, 1H), 7.67~7.56 (m, 6H), 7.30~7.25 (m, 1H), 7.13~7.09 (m, 5H), 6.49 (s, 1H); 13C NMR (101 MHz, CDCl3) δ: 166.7 (d, JC-F=3.0 Hz), 164.1 (d, JC-F=3.3 Hz), 139.8 (d, J=9.8 Hz), 134.4 (dd, J=12.1, 8.9 Hz), 128.5~128.2 (m), 127.4~127.0 (m), 124.6, 121.6, 120.6, 116.1 (dd, J=21.4, 13.9 Hz), 113.6 (d, J=16.4 Hz), 112.8; 19F NMR (376 MHz, CDCl3) δ: 105.61; 31P NMR (162 MHz, CDCl3) δ: 21.06; HRMS (ESI) calcd for C20H14F2NOPNa [M+Na]+ 376.0673, found 376.0682.
Bis(4-chlorophenyl)(1H-indol-2-yl)phosphine oxide (3ac): White solid, 83% yield. m.p. 278.8~279.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.84 (s, 1H), 7.60~7.53 (m, 6H), 7.40 (d, J=6.7 Hz, 4H), 7.31~7.25 (m, 1H), 7.12 (t, J=7.4 Hz, 1H), 6.51 (s, 1H); 13C NMR (101 MHz, CDCl3) δ: 139.9 (d, J=9.7 Hz), 139.2 (d, J=3.4 Hz), 133.3 (d, J=11.5 Hz), 130.3 (d, J=112.2 Hz), 129.1 (d, J=13.3 Hz), 127.2 (d, J=12.8 Hz), 127.0 (d, J=127.4 Hz), 124.8, 121.7, 120.7, 113.8 (d, J=16.5 Hz), 112.8; 31P NMR (162 MHz, CDCl3) δ: 20.91; HRMS (ESI) calcd for C20H14Cl2- NOPNa [M+Na]+ 408.0082, found 408.0089.
Bis(4-bromophenyl)(1H-indol-2-yl)phosphine oxide (3ad): White solid, 88% yield. m.p.>288 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 11.97 (s, 1H), 7.80 (d, J=7.0 Hz, 4H), 7.64~7.59 (m, 5H), 7.44 (d, J=8.1 Hz, 1H), 7.24 (t, J=7.4 Hz, 1H), 7.07 (t, J=7.3 Hz, 1H), 6.68 (s, 1H); 13C NMR (101 MHz, DMSO-d6) δ: 139.3 (d, J=9.8 Hz), 133.3 (d, J=11.1 Hz), 132.0 (d, J=12.8 Hz), 131.0, 128.7, 127.0 (d, J=93.6 Hz), 126.7 (d, J=3.1 Hz), 124.2, 121.6, 120.1, 113.0 (d, J=15.8 Hz), 112.4; 31P NMR (162 MHz, DMSO- d6) δ: 16.89. HRMS (ESI) calcd for C20H14Br2- NOPNa [M+Na]+ 495.9072, found 495.9078.
(1H-Indol-2-yl)di-p-tolylphosphine oxide (3ae): White solid, 64% yield. m.p. 260.1~260.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.31 (s, 1H), 7.59~7.50 (m, 6H), 7.25~7.20 (m, 5H), 7.09 (t, J=7.2 Hz, 1H), 6.54~6.53 (m, 1H), 2.39 (s, 6H); 13C NMR (101 MHz, CDCl3) δ: 142.7 (d, J=2.8 Hz), 139.4 (d, J=9.4 Hz), 132.0 (d, J=10.9 Hz), 129.3 (d, J=13.0 Hz), 129.1 (d, J=113.1 Hz), 129.0 (d, J=124.2 Hz), 127.4 (d, J=12.4 Hz), 124.1, 121.5, 120.3, 112.9 (d, J=15.2 Hz), 112.7, 21.8; 31P NMR (162 MHz, CDCl3) δ: 22.51; HRMS (ESI) calcd for C22H20NOPNa [M+Na]+ 368.1175, found 368.1183.
(1H-Indol-2-yl)bis(4-methoxyphenyl)phosphine oxide (3af): White solid, 70% yield. m.p. 264.3~264.5 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.76 (s, 1H), 7.62~7.57 (m, 5H), 7.51 (d, J=8.1 Hz, 1H), 7.26~7.24 (m, 1H), 7.11 (t, J=7.3 Hz, 1H), 6.92 (d, J=7.2 Hz, 4H), 6.55 (s, 1H), 3.84 (s, 6H); 13C NMR (101 MHz, CDCl3) δ: 162.7 (d, J=2.6 Hz), 139.2 (d, J=9.0 Hz), 133.9 (d, J=11.9 Hz), 129.4 (d, J=124.4 Hz), 127.5 (d, J=12.2 Hz), 124.2, 123.1, 121.6, 120.4, 114.2 (d, J=13.1 Hz), 112.9 (d, J=15.3 Hz), 112.6, 55.5; 31P NMR (162 MHz, CDCl3) δ: 21.90; HRMS (ESI) calcd for C22H20NO3PNa [M+Na]+ 400.1073, found 400.1082.
Bis(3,5-dimethylphenyl)(1H-indol-2-yl)phosphine oxide (3ag): White solid, 80% yield. m.p. 283.0~283.7 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.08 (s, 1H), 7.62 (q, J=8.0 Hz, 2H), 7.36 (d, J=13.0 Hz, 4H), 7.29 (t, J=7.6 Hz, 1H), 7.18~7.12 (m, 3H), 6.62 (s, 1H), 2.31 (s, 12H); 13C NMR (101 MHz, CDCl3) δ: 139.3 (d, J=9.1 Hz), 138.2 (d, J=13.3 Hz), 134.0 (d, J=2.8 Hz), 132.8 (d, J=109.1 Hz), 129.5 (d, J=10.5 Hz), 128.2 (d, J=4.1 Hz), 127.6 (d, J=12.3 Hz), 124.0, 121.5, 120.3, 112.9, 112.7, 21.4; 31P NMR (162 MHz, CDCl3) δ: 22.78; HRMS (ESI) calcd for C24H24NOPNa [M+Na]+ 396.1488, found 396.1497.
(1H-Indol-2-yl)di(naphthalen-2-yl)phosphine oxide (3ah): White solid, 64% yield. m.p.>288 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 11.99 (s, 1H), 8.45 (d, J=14.2 Hz, 2H), 8.11~8.07 (m, 5H), 8.02 (d, J=8.1 Hz, 2H), 7.78~7.73 (m, 2H), 7.70~7.60 (m, 5H), 7.45 (d, J=8.2 Hz, 1H), 7.23 (t, J=7.4 Hz, 1H), 7.07 (t, J=7.5 Hz, 1H), 6.76 (s, 1H); 13C NMR (101 MHz, DMSO-d6) δ: 139.3 (d, J=9.6 Hz), 134.4 (d, J=2.3 Hz), 133.0 (d, J=9.8 Hz), 132.1 (d, J=13.4 Hz), 130.5, 130.0, 129.4, 128.8 (d, J=49.6 Hz), 128.7, 128.4, 127.5 (d, J=63.2 Hz), 126.7, 126.5 (d, J=11.0 Hz), 124.0, 121.6, 120.0, 112.7 (d, J=15.2 Hz), 112.4; 31P NMR (162 MHz, DMSO-d6) δ: 17.58; HRMS (ESI) calcd for C28H20- NOPNa [M+Na]+ 440.1175, found 440.1184.
(1H-Indol-2-yl)(4-methoxyphenyl)(phenyl)phosphine oxide (3ai): White solid, 81% yield. m.p. 190~190.2 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.28 (s, 1H), 7.63~7.58 (m, 2H), 7.54~7.42 (m, 5H), 7.33 (td, J=7.6, 2.9 Hz, 2H), 7.17 (t, J=7.3 Hz, 1H), 7.02 (t, J=7.3 Hz, 1H), 6.83 (dd, J=8.8, 2.1 Hz, 2H), 6.46~6.45 (m, 1H), 3.75 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 162.8 (d, J=3.0 Hz), 139.5 (d, J=9.4 Hz), 133.9 (d, J=11.9 Hz), 133.1, 132.1 (d, J=2.7 Hz), 132.0 (d, J=10.6 Hz), 128.8 (d, J=124.4 Hz), 128.5 (d, J=12.6 Hz), 127.4 (d, J=12.4 Hz), 124.2, 123.2 (d, J=116.6 Hz), 121.5, 120.3, 114.2 (d, J=13.7 Hz), 113.0 (d, J=15.8 Hz), 112.7, 55.5; 31P NMR (162 MHz, CDCl3) δ: 22.23; HRMS (ESI) calcd for C21H18NO2PNa [M+Na]+ 370.0967, found 370.0975.
(6-Chloro-1H-indol-2-yl)diphenylphosphine oxide (3ba): White solid, 88% yield. m.p. 284.7~284.8 ℃; 1H NMR (400 MHz, CDCl3) δ: 12.17 (s, 1H), 7.65 (dd, J=12.5, 7.7 Hz, 4H), 7.58~7.50 (m, 3H), 7.48~7.42 (m, 5H), 7.05 (d, J=8.4 Hz, 1H), 6.48 (s, 1H); 13C NMR (101 MHz, CDCl3) δ: 140.0 (d, J=9.8 Hz), 132.4 (d, J=3.0 Hz), 131.9 (d, J=10.6 Hz), 131.8 (d, J=111.1 Hz), 130.2, 129.8, 128.7 (d, J=12.7 Hz), 125.9 (d, J=12.6 Hz), 122.4, 121.3, 113.1 (d, J=16.0 Hz), 112.6; 31P NMR (162 MHz, CDCl3) δ: 22.51; HRMS (ESI) calcd for C20H15ClNOPNa [M+Na]+ 374.0472, found 374.0480.
(6-Bromo-1H-indol-2-yl)diphenylphosphine oxide (3ca): White solid, 89% yield. m.p. 283.4~283.6 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.94 (s, 1H), 7.71 (s, 1H), 7.65 (dd, J=12.6, 7.4 Hz, 4H), 7.55 (t, J=7.1 Hz, 2H), 7.45~7.42 (m, 5H), 7.19 (dd, J=8.5, 1.1 Hz, 1H), 6.49 (s, 1H); 13C NMR (101 MHz, CDCl3) δ: 140.2 (d, J=9.1 Hz), 132.5 (d, J=2.7 Hz), 132.0 (d, J=10.6 Hz), 131.8 (d, J=110.8 Hz), 129.8, 128.8 (d, J=13.1 Hz), 126.2 (d, J=12.5 Hz), 123.9, 122.8, 118.1, 115.6, 113.2 (d, J=15.8 Hz); 31P NMR (162 MHz, CDCl3) δ: 22.37; HRMS (ESI) m/z calcd for C20H15BrNOPNa [M+Na]+ 417.9967, found 417.9977.
(6-Methoxy-1H-indol-2-yl)diphenylphosphine oxide (3da): White solid, 80% yield. m.p. 267.8~268.0 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.47 (s, 1H), 7.68 (dd, J=12.5, 7.5 Hz, 4H), 7.53 (t, J=7.2 Hz, 2H), 7.44~7.42 (m, 5H), 6.99 (s, 1H), 6.77 (d, J=8.7 Hz, 1H), 6.46 (s, 1H), 3.80 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 158.2, 140.6 (d, J=9.5 Hz), 132.3 (d, J=110.3 Hz), 132.3~131.9 (m), 130.8 (d, J=11.5 Hz), 129.1 (d, J=12.1 Hz), 128.6 (d, J=12.6 Hz), 122.3, 121.8 (d, J=12.7 Hz), 113.7 (d, J=15.9 Hz), 112.0, 94.5, 55.6; 31P NMR (162 MHz, CDCl3) δ: 22.10; HRMS (ESI) m/z calcd for C21H18NO2PNa [M+Na]+ 370.0967, found 370.0977.
(6-Methyl-1H-indol-2-yl)diphenylphosphine oxide (3ea): White solid, 87% yield. m.p. 275.2~275.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.95 (s, 1H), 7.68 (dd, J=11.9, 7.7 Hz, 4H), 7.53~7.41 (m, 7H), 7.31 (s, 1H), 6.94 (d, J=7.9 Hz, 1H), 6.51 (s, 1H), 2.44 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 139.9 (d, J=10.1 Hz), 134.5, 132.8, 132.2~131.9 (m), 131.7, 128.6 (d, J=12.7 Hz), 127.4 (d, J=124.9 Hz), 125.4 (d, J=11.8 Hz), 122.6, 121.2, 113.3 (d, J=15.6 Hz), 112.3, 22.0; 31P NMR (162 MHz, CDCl3) δ: 22.25; HRMS (ESI) calcd for C21H18NOPNa [M+Na]+ 354.1018, found 354.1026.
2-(Diphenylphosphoryl)-1H-indol-6-yl acetate (3fa): White solid, 84% yield. m.p.>288 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 12.38 (s, 1H), 8.12 (s, 1H), 7.74~7.57 (m, 12H), 6.73 (s, 1H), 3.86 (s, 3H); 13C NMR (101 MHz, DMSO-d6) δ: 166.8, 138.3 (d, J=9.7 Hz), 133.2 (d, J=118.3 Hz), 132.67, 132.4 (d, J=2.5 Hz), 131.6, 131.3 (d, J=10.3 Hz), 130.0 (d, J=11.7 Hz), 128.8 (d, J=12.3 Hz), 124.8, 121.5, 120.2, 114.3, 112.1 (d, J=15.1 Hz), 52.0; 31P NMR (162 MHz, DMSO-d6) δ: 17.81; HRMS (ESI) calcd for C22H18NO3PNa [M+H]+ 376.1097, found 376.1103.
(5-Methyl-1H-indol-2-yl)diphenylphosphine oxide (3ga): White solid, 89% yield. m.p. 242.2~242.4 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.64 (s, 1H), 7.69~7.64 (m, 4H), 7.53~7.46 (m, 3H), 7.39 (td, J=7.6, 2.9 Hz, 4H), 7.34 (s, 1H), 7.09 (d, J=8.4 Hz, 1H), 6.45~6.44 (m, 1H), 2.41 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 138.1 (d, J=9.7 Hz), 132.2 (d, J=110.1 Hz), 132.4~131.8 (m), 129.5, 128.7, 128.5 (d, J=12.6 Hz), 127.6, 127.5 (d, J=2.1 Hz), 126.2, 120.7, 112.6 (d, J=9.1 Hz), 112.5, 21.9; 31P NMR (162 MHz, CDCl3) δ: 22.42; HRMS (ESI) calcd for C21H18NOPNa [M+Na]+ 354.1018, found 354.1026.
(4-Methyl-1H-indol-2-yl)diphenylphosphine oxide (3ha): White solid, 82% yield. m.p. 264.6~264.7 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.67 (s, 1H), 7.69 (dd, J=11.1, 7.6 Hz, 4H), 7.52~7.41 (m, 7H), 7.16 (t, J=7.2 Hz, 1H), 6.89 (d, J=6.3 Hz, 1H), 6.55 (s, 1H), 2.47 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 139.4 (d, J=9.4 Hz), 132.8, 131.7, 132.1 (dd, J=21.0, 6.5 Hz), 131.2, 128.6 (d, J=12.6 Hz), 127.6 (d, J=12.4 Hz), 127.5 (d, J=124.8 Hz), 124.4, 120.3, 111.6 (d, J=15.7 Hz), 110.3, 18.9; 31P NMR (162 MHz, CDCl3) δ: 22.39; HRMS (ESI) calcd for C21H18- NOPNa [M+Na]+ 354.1018, found 354.1028.
(5,7-Dimethyl-1H-indol-2-yl)diphenylphosphine oxide (3ia): White solid, 71% yield. m.p. 235.6~235.9 ℃; 1H NMR (400 MHz, CDCl3) δ: 10.57 (s, 1H), 7.68~7.63 (m, 4H), 7.53 (td, J=7.4, 1.3 Hz, 2H), 7.42 (td, J=7.7, 2.9 Hz, 4H), 7.20 (s, 1H), 6.91 (s, 1H), 6.47 (dd, J=3.7, 2.0 Hz, 1H), 2.52 (s, 3H), 2.39 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 137.6 (d, J=8.9 Hz), 132.3 (d, J=109.9 Hz), 132.2 (d, J=2.8 Hz), 132.0 (d, J=10.6 Hz), 130.1, 128.6 (d, J=12.6 Hz), 127.9 (d, J=97.3 Hz), 127.2 (d, J=14.8 Hz), 126.9, 121.8, 118.4, 113.4 (d, J=15.8 Hz), 21.5, 17.2; 31P NMR (162 MHz, CDCl3) δ: 22.25; HRMS (ESI) calcd for C22H20NOPNa [M+Na]+ 368.1175, found 368.1184.
(6-(Naphthalen-1-yl)-1H-indol-2-yl)diphenylphosphine oxide (3ja): White solid, 75% yield. m.p. 201~202 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 12.07 (s, 1H), 7.97 (dd, J=21.4, 8.0 Hz, 2H), 7.85 (d, J=8.2 Hz, 1H), 7.77~7.72 (m, 5H), 7.66~7.46 (m, 11H), 7.18 (d, J=8.0 Hz, 1H), 6.75 (s, 1H); 13C NMR (101 MHz, DMSO-d6) δ: 140.2, 139.3 (d, J=9.5 Hz), 135.8, 133.4, 133.1, 132.3 (d, J=2.0 Hz), 132.1, 131.3 (d, J=10.2 Hz), 131.1, 131.0 (d, J=121.3 Hz), 128.8 (d, J=12.2 Hz), 128.3, 127.4, 127.0, 126.2, 126.1, 125.9 (d, J=6.1 Hz), 125.6 (d, J=2.9 Hz), 122.5, 121.3, 113.2, 112.4 (d, J=15.1 Hz); 31P NMR (162 MHz, DMSO-d6) δ: 17.78; HRMS (ESI) calcd for C30H22NOPNa [M+Na]+ 466.1331, found 466.1336.
Diphenyl(6-(thiophen-2-yl)-1H-indol-2-yl)phosphine oxide (3ka): White solid, 83% yield. m.p. 275~275.2 ℃; 1H NMR (400 MHz, CDCl3) δ: 11.49 (s, 1H), 7.71 (s, 1H), 7.65 (dd, J=12.6, 7.5 Hz, 4H), 7.49~7.45 (m, 3H), 7.38~7.31 (m, 5H), 7.23 (d, J=3.1 Hz, 1H), 7.17 (d, J=5.0 Hz, 1H), 7.00 (t, J=3.8 Hz, 1H), 6.46 (s, 1H); 13C NMR (101 MHz, CDCl3) δ: 145.5, 139.8 (d, J=9.4 Hz), 132.4 (d, J=2.7 Hz), 132.0 (d, J=10.6 Hz), 131.2 (d, J=47.0 Hz), 128.7 (d, J=12.7 Hz), 128.1, 123.8 (d, J=149.3 Hz), 122.0, 119.7, 113.3 (d, J=15.6 Hz), 109.6; 31P NMR (162 MHz, CDCl3) δ: 22.14; HRMS (ESI) calcd for C24H18NOPSNa [M+Na]+ 422.0739, found 422.0746.
(5
H-[
1,
3]Dioxolo[5-f]indol-6-yl)diphenylphosphine oxide (
3la): White solid, 88% yield. m.p.>288 ℃;
1H NMR (400 MHz, DMSO-
d6)
δ: 11.76 (s, 1H), 7.70~7.55 (m, 10H), 7.05 (s, 1H), 6.89 (s, 1H), 6.47 (s, 1H), 5.98 (s, 2H);
13C NMR (101 MHz, DMSO-
d6)
δ: 146.4, 143.2, 134.9 (d,
J=9.7 Hz), 132.9 (d,
J=107.5 Hz), 132.1 (d,
J=2.0 Hz), 131.3 (d,
J=10.1 Hz), 128.7 (d,
J=12.1 Hz), 127.1 (d,
J=125.5 Hz), 120.8 (d,
J=12.3 Hz), 112.9 (d,
J=15.7 Hz), 100.7, 99.0, 92.1;
31P NMR (162 MHz, DMSO-
d6)
δ: 17.16; HRMS (ESI) calcd for C
21H
16NO
3PNa [M+Na]
+ 384.0760, found 384.0767.