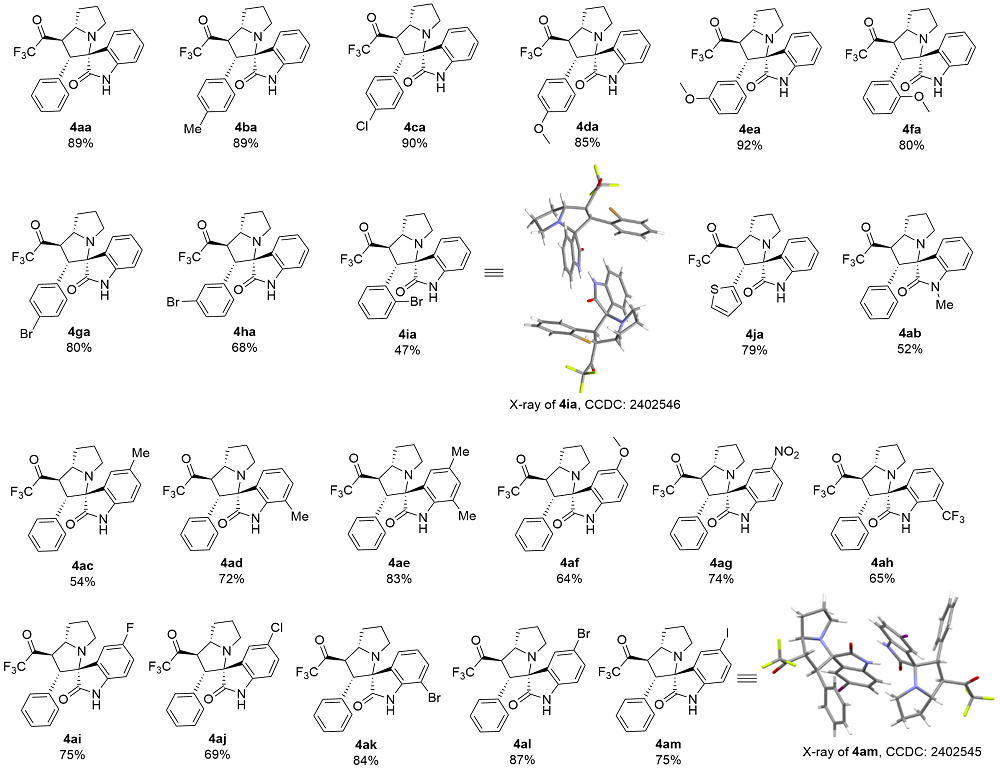
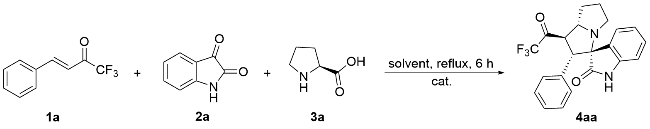
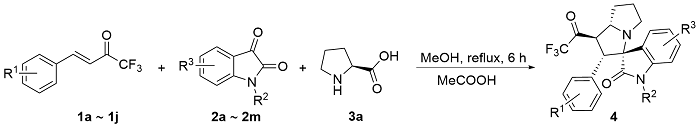
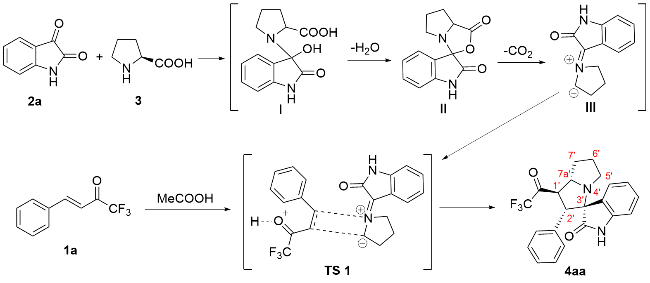
以2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4aa)为例. 将三氟甲基酮2a (0.2 mmol, 40 mg)、靛红(0.32 mmol, 47 mg)、L-脯氨酸(0.4 mmol, 46 mg)加入25 mL反应管中, 加入4 mL甲醇搅拌5 min, 然后加入冰乙酸(0.5 mmol, 3 μL), 回流6 h至薄层色谱(TLC)监测三氟甲基酮消失, 将反应液通过硅藻土过滤, 乙酸乙酯洗涤滤饼, 浓缩后快速柱色谱分离[V(PE):V(EA)=5:1], 得到目标产物2'-苯 基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚 啉-3,3'-吡咯里嗪]-2-酮(4aa), 黄色固体, 35.6 mg, 产率89%. m.p. 87.1 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.78 (s, 1H), 7.49 (d, J=7.2 Hz, 1H), 7.15 (t, J=7.0 Hz, 1H), 7.10 (d, J=7.2 Hz, 1H), 7.04 (d, J=7.2 Hz, 1H), 6.97 (t, J=7.4 Hz, 2H), 6.87 (d, J=7.2 Hz, 2H), 6.51 (d, J=7.4 Hz, 1H), 4.78 (dd, J=12.0, 8.0 Hz, 1H), 4.40 (d, J=12.0 Hz, 1H), 3.93 (q, J=8.0 Hz, 1H), 2.71 (t, J=8.4 Hz, 1H), 2.56 (td, J=9.8, 6.4 Hz, 1H), 2.13 (h, J=5.0, 4.4 Hz, 3H), 1.87 (tt, J=12.8, 6.2 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.5 (q, J=36.0 Hz), 179.2, 140.8, 132.8, 129.6, 129.2, 128.1, 128.0, 127.7, 124.3, 123.2, 115.5 (q, J=291.7 Hz), 109.8, 77.6, 69.1, 60.3, 51.4, 48.1, 32.2, 28.8; 19F NMR (376 MHz, Chloroform-d) δ: -77.14; IR (KBr) ν: 3198.7 (NH stretching vibration), 2966.8, 2870.8, 1756.5 (C=O), 1707.4 (C=O), 1620.3 (NH deformation vibration), 1472.0, 1258.8, 1202.7, 1149.5, 1061.2, 1014.0, 946.4, 844.7, 752.6, 741.0 cm-1; HRMS (ESI) calcd for C22H20F3N2O2 (M+H)+ 401.1472, found 401.1471.
2'-(对甲苯基)-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ba): 白色固体, 36.8 mg, 产率89%. m.p. 122.3 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.40 (s, 1H), 7.40 (d, J=7.0 Hz, 1H), 7.07 (t, J=7.0 Hz, 1H), 7.01 (t, J=7.0 Hz, 1H), 6.71 (d, J=8.0 Hz, 2H), 6.67 (d, J=8.2 Hz, 2H), 6.43 (d, J=7.4 Hz, 1H), 4.67 (dd, J=12.0, 8.2 Hz, 1H), 4.28 (d, J=12.0 Hz, 1H), 3.84 (q, J=8.0 Hz, 1H), 2.62 (t, J=7.6 Hz, 1H), 2.51~2.44 (m, 1H), 2.04 (s, 6H), 1.79 (td, J=13.6, 12.4, 6.0 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.6 (q, J=36.0 Hz), 179.1, 140.7, 137.5, 129.8, 129.8, 129.1, 128.8, 127.6, 124.2, 123.1, 115.5 (q, J=291.7 Hz), 109.7, 77.5, 69.2, 60.1, 51.5, 48.0, 32.2, 28.8, 21.0; 19F NMR (376 MHz, Chloroform-d) δ: -77.16; IR (KBr) ν: 3201.7 (NH stretching vibration), 2963.1, 2924.1, 2869.4, 1756.0 (C=O), 1707.4 (C=O), 1621.0 (NH deformation vibration), 1517.0, 1471.9, 1259.0, 1202.8, 1149.8, 1015.0, 844.8, 820.9, 747.5, 717.3 cm-1; HRMS (ESI) calcd for C23H22F3N2O2 (M+H)+ 415.1628, found 415.1629.
2'-(4-氯苯基)-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ca): 粉色固体, 39.0 mg, 产率90%. m.p. 104.5 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.54 (s, 1H), 7.49 (d, J=7.0 Hz, 1H), 7.15 (dt, J=26.0, 6.8 Hz, 2H), 7.00 (d, J=8.8 Hz, 2H), 6.81 (d, J=8.8 Hz, 2H), 6.56 (d, J=7.6 Hz, 1H), 4.72 (dd, J=12.0, 8.2 Hz, 1H), 4.35 (d, J=12.0 Hz, 1H), 3.95 (p, J=8.2, 7.4 Hz, 1H), 2.80~2.69 (m, 1H), 2.57 (td, J=9.4, 6.0 Hz, 1H), 2.15 (dt, J=15.2, 7.8 Hz, 3H), 1.96~1.83 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.3 (q, J=36.2 Hz), 178.9, 140.5, 133.9, 131.5, 129.3, 129.3, 129.1, 128.3, 124.2, 123.3,115.5 (q, J=291.7 Hz), 109.9, 77.4, 69.0, 59.6, 51.4, 48.0, 32.2, 28.9; 19F NMR (376 MHz, Chloroform-d) δ: -77.09; IR (KBr) ν: 3198.7 (NH stretching vibration), 2966.6, 2870.4, 1756.7 (C=O), 1709.2 (C=O), 1621.5 (NH deformation vibration), 1494.7, 1471.8, 1261.3, 1202.8, 1150.4, 1039.1, 1014.3, 927.3, 844.1, 747.7, 716.7 cm-1; HRMS (ESI) calcd for C22H19ClF3N2O2 (M+H)+ 435.1083, found 435.1086.
2'-(4-甲氧基苯基)-1'-(2,2,2-三氟乙酰基)- 1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4da): 黄色固体, 36.6 mg, 产率85%. m.p. 106.6 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.47 (s, 1H), 7.49 (d, J=7.2 Hz, 1H), 7.17 (t, J=7.0 Hz, 1H), 7.11 (t, J=7.0 Hz, 1H), 6.79 (d, J=8.4 Hz, 2H), 6.57~6.53 (m, 1H), 6.50 (d, J=8.8 Hz, 2H), 4.72 (dd, J=12.0, 8.2 Hz, 1H), 4.34 (d, J=12.0 Hz, 1H), 3.92 (q, J=8.2 Hz, 1H), 3.57 (s, 3H), 2.71 (t, J=7.6 Hz, 1H), 2.54 (td, J=9.4, 6.0 Hz, 1H), 2.12 (p, J=6.6, 5.8 Hz, 3H), 1.88 (tt, J=12.8, 6.0 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.6 (q, J=36.0 Hz), 179.2, 159.1, 140.7, 129.9, 129.1, 128.7, 124.7, 124.2, 123.1, 115.5 (q, J=291.7 Hz), 113.4, 109.7, 109.7, 77.6, 69.1, 59.8, 54.9, 51.7, 48.0, 32.2, 28.8; 19F NMR (376 MHz, Chloroform-d) δ: -77.16; IR (KBr) ν: 3198.6 (NH stretching vibration), 2964.7, 2871.9, 1756.1 (C=O), 1707.3 (C=O), 1620.5 (NH deformation vibration), 1515.7, 1471.6, 1305.9, 1251.8, 1202.8, 1185.2, 1150.1, 1035.5, 1017.0, 947.7, 926.8, 833.7, 773.0, 749.6, 703.5 cm-1; HRMS (ESI) calcd for C23H22F3N2O3 (M+H)+ 431.1578, found 431.1580.
2'-(3-甲氧基苯基)-1'-(2,2,2-三氟乙酰基)- 1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ea): 白色固体, 39.6 mg, 产率92%. m.p. 82.6 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.50~8.13 (m, 1H), 7.52 (d, J=7.2 Hz, 1H), 7.15 (dt, J=22.9, 7.5 Hz, 2H), 6.91 (t, J=7.9 Hz, 1H), 6.61 (d, J=8.2 Hz, 1H), 6.56 (d, J=7.5 Hz, 1H), 6.50 (d, J=7.6 Hz, 1H), 6.29 (s, 1H), 4.75 (dd, J=12.0, 8.2 Hz, 1H), 4.38 (d, J=12.0 Hz, 1H), 3.92 (q, J=8.2 Hz, 1H), 3.45 (s, 3H), 2.74 (t, J=7.6 Hz, 1H), 2.59 (td, J=9.2, 6.2 Hz, 1H), 2.14 (q, J=9.0, 7.2 Hz, 3H), 1.88 (dt, J=21.0, 10.8 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.5 (q, J=35.9 Hz), 178.8, 159.0, 140.8, 134.4, 129.8, 129.1, 129.1, 124.2, 123.1, 119.5, 115.5 (q, J=291.7 Hz), 114.3, 112.6, 109.7, 77.3, 69.2, 60.2, 54.8, 51.3, 48.1, 32.2, 28.9; 19F NMR (376 MHz, Chloroform-d) δ: -76.98; IR (KBr) ν: 3223.6 (NH stretching vibration), 2926.5, 2869.9, 2358.8, 2339.0, 1756.6 (C=O), 1709.2 (C=O), 1620.9 (NH deformation vibration), 1603.1, 1489.8, 1471.7, 1327.4, 1259.3, 1208.0, 1150.5, 1041.3, 1021.7, 843.8, 753.0, 708.1 cm-1; HRMS (ESI) calcd for C23H22F3N2O3 (M+ H)+ 431.1578, found 431.1580.
2'-(2-甲氧基苯基)-1'-(2,2,2-三氟乙酰基)- 1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4fa): 黄色固体, 34.4 mg, 产率80%. m.p. 102.4 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.30 (d, J=24.0 Hz, 1H), 7.55 (d, J=6.8 Hz, 1H), 7.30 (d, J=7.6 Hz, 1H), 7.06 (dq, J=23.0, 7.2 Hz, 3H), 6.65 (t, J=7.6 Hz, 1H), 6.53 (dd, J=13.0, 8.0 Hz, 2H), 5.17 (d, J=12.4 Hz, 1H), 4.76 (dd, J=12.2, 8.2 Hz, 1H), 3.94 (q, J=8.0 Hz, 1H), 3.27 (s, 3H), 2.72 (t, J=7.8 Hz, 1H), 2.61~2.54 (m, 1H), 2.13 (q, J=6.8, 6.0 Hz, 3H), 1.96~1.84 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.6 (q, J=35.8 Hz), 179.5, 157.6, 140.3, 129.9, 128.8, 128.5, 127.6, 125.6, 122.3, 121.6, 120.2, 115.5 (q, J=291.7 Hz), 110.4, 109.1, 77.3, 69.1, 54.9, 52.1, 51.4, 48.0, 32.2, 28.9; 19F NMR (376 MHz, Chloroform-d) δ: -77.26; IR (KBr) ν: 3262.7 (NH stretching vibration), 2965.3, 2872.6, 1756.3 (C=O), 1712.1 (C=O), 1621.7 (NH deformation vibration), 1604.2, 1495.4, 1471.3, 1392.2, 1254.2, 1208.2, 1150.4, 1056.7, 1031.1, 1041.6, 845.1, 751.8, 704.8 cm-1; HRMS (ESI) calcd for C23H22- F3N2O3 (M+H)+ 431.1578, found 431.1580.
2'-(4-溴苯基)-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ga): 黄色固体, 38.2 mg, 产率80%. m.p. 114.4 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.54 (s, 1H), 7.48 (d, J=7.2 Hz, 1H), 7.19~7.09 (m, 4H), 6.75 (d, J=8.4 Hz, 2H), 6.56 (d, J=7.6 Hz, 1H), 4.71 (dd, J=12.0, 8.2 Hz, 1H), 4.34 (d, J=12.0 Hz, 1H), 3.94 (q, J=8.2 Hz, 1H)., 2.75 (t, J=7.8 Hz, 1H), 2.57 (td, J=9.4, 6.2 Hz, 1H), 2.19~2.09 (m, 3H), 1.89 (td, J=9.4, 4.6 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.2 (q, J=36.2 Hz), 178.9, 140.5, 132.0, 131.3, 129.4, 129.3, 129.3, 124.2, 123.3, 122.2, 115.5 (q, J=291.6 Hz), 109.9, 77.3, 69.0, 59.6, 51.4, 48.0, 32.2, 28.9; 19F NMR (376 MHz, Chloroform-d) δ: -77.02; IR (KBr) ν: 3213.6 (NH stretching vibration), 2965.5, 2873.5, 1755.8 (C=O), 1709.3 (C=O), 1620.8 (NH deformation vibration), 1491.4, 1471.7, 1409.5, 1329.7, 1202.6, 1150.1, 1078.0, 1010.6, 843.0, 750.1, 715.2 cm-1; HRMS (ESI) calcd for C22H19- BrF3N2O2 (M+H)+ 479.0577, found 479.0577.
2'-(3-溴苯基)-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ha): 粉色固体, 32.5 mg, 产率68%. m.p. 98.5 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.28 (s, 1H), 7.51 (d, J=7.2 Hz, 1H), 7.21 (t, J=7.0 Hz, 2H), 7.15 (t, J=7.0 Hz, 1H), 6.95 (s, 1H), 6.86 (dt, J=14.8, 7.8 Hz, 2H), 6.61 (d, J=7.4 Hz, 1H), 4.71 (dd, J=12.0, 8.2 Hz, 1H), 4.34 (d, J=12.1 Hz, 1H), 3.92 (q, J=8.4 Hz, 1H), 2.74 (t, J=7.6 Hz, 1H), 2.59 (td, J=9.4, 6.2 Hz, 1H), 2.15 (tt, J=12.2, 5.8 Hz, 3H), 1.96~1.84 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.2 (q, J=36.1 Hz), 178.8, 140.5, 135.3, 131.1, 130.9, 129.7, 129.4, 129.2, 126.1, 124.2, 123.4, 122.2, 115.5 (q, J=291.6 Hz), 109.9, 77.3, 69.0, 59.5, 51.3, 48.1, 32.2, 28.9; 19F NMR (376 MHz, Chloroform-d) δ: -77.11; IR (KBr) ν: 3195.7 (NH stretching vibration), 2969.2, 2872.4, 2361.0, 2340.9, 1757.4 (C=O), 1709.4 (C=O), 1621.7 (NH deformation vibration), 1568.7, 1472.2, 1430.1, 1333.5, 1260.3, 1202.9, 1151.0, 1078.3, 1016.2, 844.3, 745.5, 708.3 cm-1; HRMS (ESI) calcd for C22H19BrF3N2O2 (M+H)+ 479.0577, found 479.0577.
2'-(2-溴苯基)-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ia): 白色固体, 22.5 mg, 产率47%. m.p. 115.4 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.17 (s, 1H), 7.65 (d, J=7.2 Hz, 1H), 7.55 (d, J=8.0 Hz, 1H), 7.30 (d, J=8.0 Hz, 1H), 7.16 (t, J=7.2 Hz, 1H), 7.06 (dt, J=11.2, 7.6 Hz, 2H), 6.94 (t, J=7.6 Hz, 1H), 6.58 (d, J=7.6 Hz, 1H), 5.26 (d, J=12.0 Hz, 1H), 4.66 (dd, J=12.0, 8.2 Hz, 1H), 3.98 (q, J=7.8 Hz, 1H), 2.74 (t, J=8.0 Hz, 1H), 2.60 (td, J=9.2, 6.2 Hz, 1H), 2.21~2.10 (m, 3H), 1.94 (dt, J=15.6, 7.4 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.0 (q, J=36.1 Hz), 179.5, 140.1, 133.3, 132.6, 129.4, 129.3, 128.8, 128.2, 127.4, 126.7, 126.3, 122.7, 115.4 (q, J=291.6 Hz), 109.3, 77.3, 68.7, 56.4, 53.7, 47.7, 32.1, 29.0; 19F NMR (376 MHz, Chloroform-d) δ: -77.23; IR (KBr) ν: 3415.8 (NH stretching vibration), 2964.1, 2869.8, 1756.5 (C=O), 1705.0 (C=O), 1621.2 (NH deformation vibration), 1472.1, 1437.7, 1259.5, 1203.3, 1149.8, 1028.1, 842.9, 746.9, 718.9 cm-1; HRMS (ESI) calcd for C22H19BrF3N2O2 (M+H)+ 479.0577, found 479.0577.
2'-(2-噻吩基)-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ja): 棕色固体, 32.1 mg, 产率79%. m.p. 64.2 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.43 (s, 1H), 7.49 (d, J=7.2 Hz, 1H), 7.24 (t, J=7.4 Hz, 1H), 7.14 (t, J=7.0 Hz, 1H), 6.90 (d, J=4.0 Hz, 1H), 6.68~6.65 (m, 2H), 6.61 (d, J=3.2 Hz, 1H), 4.69 (dd, J=12.0, 7.6 Hz, 1H), 4.63 (d, J=11.8 Hz, 1H), 3.88 (q, J=7.8 Hz, 1H), 2.75 (t, J=7.6 Hz, 1H), 2.58 (td, J=9.4, 6.0 Hz, 1H), 2.14 (p, J=7.0, 5.6 Hz, 3H), 1.94~1.83 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.1 (q, J=36.1 Hz), 179.1, 141.1, 135.8, 129.4, 126.5, 125.3, 124.7, 124.3, 123.3, 115.5 (q, J=291.6 Hz), 109.9, 77.1, 69.2, 55.3, 53.3, 48.3, 32.2, 28.8; 19F NMR (376 MHz, Chloroform-d) δ: -76.87; IR (KBr) ν: 3207.2 (NH stretching vibration), 2962.4, 2871.8, 1756.7 (C=O), 1710.1 (C=O), 1621.4 (NH deformation vibration), 1471.5, 1278.9, 1241.2, 1204.6, 1151.0, 1043.9, 846.0, 753.0, 703.2 cm-1; HRMS (ESI) calcd for C20H18F3N2O2S (M+H)+ 407.1036, found 407.1037.
1-甲基-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7', 7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ab): 粉色固体, 21.5 mg, 产率52%. m.p. 110.7 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 7.56 (d, J=6.4 Hz, 1H), 7.21 (t, J=7.0 Hz, 1H), 7.14 (t, J=7.4 Hz, 1H), 7.04 (dt, J=23.2, 7.0 Hz, 3H), 6.83 (d, J=7.2 Hz, 2H), 6.48 (d, J=7.6 Hz, 1H), 4.82 (dd, J=12.0, 8.2 Hz, 1H), 4.36 (d, J=12.0 Hz, 1H), 3.96 (q, J=8.0 Hz, 1H), 2.71 (s, 4H), 2.59 (td, J=9.6, 6.0 Hz, 1H), 2.20 (td, J=12.6, 12.0, 5.4 Hz, 3H), 1.94 (dt, J=16.6, 7.8 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.6 (q, J=36.0 Hz), 176.4, 143.3, 132.8, 129.2, 129.1, 127.9, 127.8, 127.6, 123.6, 123.1, 115.5 (q, J=291.7 Hz), 107.7, 77.6, 69.5, 60.9, 51.2, 48.1, 32.2, 29.0, 25.3; 19F NMR (376 MHz, Chloroform-d) δ: -77.35; IR (KBr) ν: 3192.6 (NH stretching vibration), 2964.2, 2865.0, 1754.8 (C=O), 1707.8 (C=O), 1613.4 (NH deformation vibration), 1492.9, 1470.1, 1373.2, 1351.1, 1204.8, 1150.1, 1122.4, 1086.3, 1021.2, 947.9, 752.8 cm-1; HRMS (ESI) calcd for C23H22F3N2O2 (M+H)+ 415.1628, found 415.1629.
5-甲基-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7', 7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ac): 黄色固体, 22.4 mg, 产率54%. m.p. 91.4 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 7.95 (s, 1H), 7.36 (s, 1H), 7.07 (t, J=7.2 Hz, 1H), 7.02~6.96 (m, 3H), 6.87 (d, J=7.4 Hz, 2H), 6.44 (d, J=7.8 Hz, 1H), 4.77 (dd, J=12.0, 8.2 Hz, 1H), 4.38 (d, J=12.0 Hz, 1H), 3.92 (q, J=8.0 Hz, 1H), 2.72 (t, J=7.6 Hz, 1H), 2.61~2.55 (m, 1H), 2.38 (s, 3H), 2.20~2.09 (m, 3H), 1.94~1.85 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.6 (q, J=36.0 Hz), 178.7, 138.1, 132.9, 132.7, 129.7, 129.5, 128.1, 127.9, 127.7, 124.8, 115.5 (q, J=291.7 Hz), 109.3, 77.5, 69.2, 60.2, 51.4, 48.1, 32.2, 28.8, 21.2; 19F NMR (376 MHz, Chloroform-d) δ: -77.16; IR (KBr) ν: 3183.2 (NH stretching vibration), 2967.7, 2923.5, 2869.2, 1756.7 (C=O), 1706.0 (C=O), 1627.5 (NH deformation vibration), 1493.9, 1455.0, 1265.4, 1204.2, 1150.1, 1061.5, 1018.2, 854.3, 812.2, 739.2 cm-1; HRMS (ESI) calcd for C23H22F3N2O2 (M+H)+ 415.1628, found 415.1629.
7-甲基-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7', 7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ad): 黄色固体, 31.1 mg, 产率72%. m.p. 86.7 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 9.03 (d, J=14.6 Hz, 1H), 7.38 (d, J=7.0 Hz, 1H), 7.09~7.01 (m, 3H), 6.93 (t, J=7.6 Hz, 2H), 6.82 (d, J=7.4 Hz, 2H), 4.75 (dd, J=12.0, 8.2 Hz, 1H), 4.38 (d, J=12.0 Hz, 1H), 3.96~3.88 (m, 1H), 2.72 (t, J=7.6 Hz, 1H), 2.52 (td, J=9.4, 6.2 Hz, 1H), 2.18~2.04 (m, 6H), 1.96~1.87 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.5 (q, J=35.9 Hz), 180.0, 180.0, 139.4, 132.8, 130.6, 129.3, 128.0, 127.9, 127.6, 123.1, 121.5, 119.0, 115.5 (q, J=291.7 Hz), 77.9, 69.2, 60.4, 51.4, 48.1, 32.2, 28.9, 15.7; 19F NMR (376 MHz, Chloroform-d) δ: -77.25; IR (KBr) ν: 3183.1 (NH stretching vibration), 2965.3, 2866.9, 1756.7 (C=O), 1701.7 (C=O), 1628.8 (NH deformation vibration), 1607.1, 1457.2, 1274.6, 1203.8, 1145.6, 1063.1, 1017.9, 839.0, 747.4 cm-1; HRMS (ESI) calcd for C23H22F3N2O2 (M+H)+ 415.1628, found 415.1629.
5,7-二甲基-2'-phenyl-1'-(2,2,2-三氟乙酰基)-1',2',5', 6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ae): 黄色固体, 35.5 mg, 产率83%. m.p. 155.6 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 9.07 (d, J=14.8 Hz, 1H), 7.21 (s, 1H), 7.03 (t, J=7.2 Hz, 1H), 6.92 (t, J=7.8 Hz, 2H), 6.83 (d, J=7.2 Hz, 3H), 4.74 (dd, J=12.0, 8.2 Hz, 1H), 4.37 (d, J=12.0 Hz, 1H), 3.91 (q, J=8.2 Hz, 1H), 2.72 (t, J=7.8 Hz, 1H), 2.51 (td, J=9.4, 6.4 Hz, 1H), 2.37 (s, 3H), 2.17~2.02 (m, 6H), 1.96~1.87 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.5 (q, J=35.9 Hz), 179.9, 137.0, 132.9, 132.6, 131.1, 129.3, 128.0 127.8, 127.6, 122.1, 118.7, 115.5 (q, J=291.7 Hz), 77.9, 69.3, 60.3, 51.5, 48.1, 32.2, 28.9, 21.1, 15.7; 19F NMR (376 MHz, Chloroform-d) δ: -77.21; IR (KBr) ν: 3181.3 (NH stret- ching vibration), 2926.1, 2867.6, 1757.2 (C=O), 1699.7 (C=O), 1628.1 (NH deformation vibration), 1481.9, 1455.2, 1266.9, 1204.1, 1148.8, 1061.7, 1013.9, 849.3, 739.8 cm-1; HRMS (ESI) calcd for C24H24F3N2O2 (M+ H)+ 429.1785, found 429.1784.
5-甲氧基-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6', 7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4af): 黄色固体, 27.5 mg, 产率64%. m.p. 86.3 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.39~8.22 (m, 1H), 7.15 (d, J=2.6 Hz, 1H), 7.06 (t, J=7.2 Hz, 1H), 6.98 (t, J=7.6 Hz, 2H), 6.87 (d, J=7.4 Hz, 2H), 6.72 (dd, J=8.4, 2.6 Hz, 1H), 6.48 (d, J=8.4 Hz, 1H), 4.77 (dd, J=12.0, 8.2 Hz, 1H), 4.37 (d, J=12.0 Hz, 1H), 3.91 (q, J=8.2 Hz, 1H), 3.84 (s, 3H), 2.72 (t, J=7.6 Hz, 1H), 2.56 (td, J=9.2, 6.0 Hz, 1H), 2.13 (t, J=7.0 Hz, 3H), 1.94~1.82 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.6 (q, J=36.0 Hz), 179.0, 156.4, 133.9, 132.8, 131.1, 128.1, 127.9, 127.6, 115.5 (q, J=291.7 Hz), 114.6, 110.3, 110.2, 77.9, 69.2, 60.4, 55.8, 51.4, 48.0, 32.1, 28.8; 19F NMR (376 MHz, Chloroform-d) δ: -77.24; IR (KBr) ν: 3162.2 (NH stretching vibration), 2964.1, 2868.1, 1755.2 (C=O), 1702.0 (C=O), 1607.8 (NH deformation vibration), 1490.0, 1457.5, 1270.4, 1203.9, 1150.0, 1032.1, 1016.9, 853.7, 808.9, 737.4 cm-1; HRMS (ESI) calcd for C23H22- F3N2O3 (M+H)+ 431.1578, found 431.1580.
5-硝基-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7', 7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ag): 黄色固体, 33.1 mg, 产率74%. m.p. 118.0 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.48 (d, J=2.4 Hz, 1H), 8.31 (s, 1H), 8.18 (dd, J=8.6, 2.3 Hz, 1H), 7.14 (t, J=7.4 Hz, 1H), 7.05 (t, J=7.6 Hz, 2H), 6.87 (d, J=7.4 Hz, 2H), 6.73 (d, J=8.6 Hz, 1H), 4.71 (dd, J=12.0, 8.4 Hz, 1H), 4.45 (d, J=12.0 Hz, 1H), 3.97 (td, J=8.6, 6.0 Hz, 1H), 2.77 (t, J=7.4 Hz, 1H), 2.58 (td, J=9.2, 6.4 Hz, 1H), 2.24~2.06 (m, 3H), 2.00~1.87 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 190.8 (q, J=36.3 Hz), 146.2, 144.1, 131.9, 131.3, 128.4, 127.6, 126.3, 120.3, 115.4 (q, J=291.7 Hz), 109.6, 77.3, 69.2, 60.7, 51.4, 48.0, 32.1, 28.9; 19F NMR (376 MHz, Chloroform-d) δ: -77.25; IR (KBr) ν: 3103.1 (NH stretching vibration), 2922.5, 2869.7, 1752.1 (C=O), 1722.1 (C=O), 1626.9 (NH deformation vibration), 1606.7, 1526.6, 1479.0, 1452.1, 1338.1, 1261.1, 1203.2, 1150.8, 856.9, 832.5, 761.5, 733.3 cm-1; HRMS (ESI) calcd for C22H19F3N3O4 (M+H)+ 446.1323, found 446.1323.
2'-苯基-1'-(2,2,2-三氟乙酰基)-7-(三氟甲基)-1',2',5', 6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ah): 黄色固体, 30.4 mg, 产率65%. m.p. 142.3 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.68 (d, J=23.0 Hz, 1H), 7.75 (d, J=7.4 Hz, 1H), 7.42 (d, J=8.0 Hz, 1H), 7.24 (t, J=8.0 Hz, 1H), 7.11 (t, J=7.4 Hz, 1H), 6.99 (t, J=7.6 Hz, 2H), 6.80 (d, J=7.4 Hz, 2H), 4.76 (dd, J=12.0, 8.2 Hz, 1H), 4.38 (d, J=12.0 Hz, 1H), 3.94 (q, J=7.4, 6.2 Hz, 1H), 2.75 (t, J=7.6 Hz, 1H), 2.60 (td, J=9.2, 6.2 Hz, 1H), 2.29~2.03 (m, 3H), 1.93 (p, J=9.6 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.4 (q, J=36.2 Hz), 178.9, 138.2, 132.1, 131.7, 128.2, 127.6, 127.3, 126.0 (q, J=4.1 Hz), 123.6 (q, J=272.9 Hz), 122.8, 115.5 (q, J=291.7 Hz), 112.1 (q, J=33.2 Hz), 76.7, 69.3, 60.7, 51.0, 48.1, 32.1, 28.9; 19F NMR (376 MHz, Chloroform-d) δ: -59.27, -77.33; IR (KBr) ν: 3207.2 (NH stretching vibration), 2968.2, 2870.5, 1756.8 (C=O), 1719.3 (C=O), 1622.4 (NH deformation vibration), 1456.9, 1342.2, 1195.9, 1154.2, 1127.0, 1017.0, 968.9, 846.0, 749.9 cm-1; HRMS (ESI) calcd for C23H19F6N2O2 (M+H)+ 469.13452, found 469.13489.
5-氟-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ai): 黄色固体, 31.3 mg, 产率75%. m.p. 82.7 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.38 (d, J=24.2 Hz, 1H), 7.29 (dd, J=7.8, 2.6 Hz, 1H), 7.09 (t, J=7.2 Hz, 1H), 7.01 (t, J=7.6 Hz, 2H), 6.93~6.83 (m, 3H), 6.50 (dd, J=8.4, 4.2 Hz, 1H), 4.74 (dd, J=12.0, 8.2 Hz, 1H), 4.36 (d, J=12.0 Hz, 1H), 3.97~3.86 (m, 1H), 2.73 (t, J=7.6 Hz, 1H), 2.55 (td, J=9.4, 6.2 Hz, 1H), 2.12 (dq, J=15.8, 8.0, 6.8 Hz, 3H), 1.97~1.84 (m, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.3 (q, J=36.1 Hz), 179.2, 160.8, 158.4, 136.4, 132.5, 131.8 (d, J=7.4 Hz), 128.2, 128.1, 127.6, 115.6 (d, J=23.8 Hz), 115.5 (q, J=291.7 Hz), 112.1 (d, J=24.6 Hz), 110.3 (d, J=7.8 Hz), 77.8, 69.2, 60.5, 51.4, 48.0, 32.1, 28.8; 19F NMR (376 MHz, Chloroform-d) δ: -77.21, 117.29; IR (KBr) ν: 3200.4 (NH stretching vibration), 2962.2, 2926.5, 2870.8, 1757.0 (C=O), 1708.7 (C=O), 1630.1 (NH deformation vibration), 1485.5, 1458.7, 1273.3, 1203.8, 1150.0, 1061.7, 1016.7, 856.6, 814.2, 739.7 cm-1; HRMS (ESI) calcd for C22H19F4N2O2 (M+ H)+ 419.1378, found 419.1379.
5-氯-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4aj): 黄色固体, 29.9 mg, 产率69%. m.p. 90.7 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.27~7.92 (m, 1H), 7.54 (d, J=2.2 Hz, 1H), 7.17 (dd, J=8.2, 2.2 Hz, 1H), 7.10 (t, J=7.2 Hz, 1H), 7.02 (t, J=7.4 Hz, 2H), 6.87 (d, J=7.4 Hz, 2H), 6.51 (d, J=8.2 Hz, 1H), 4.72 (dd, J=12.0, 8.2 Hz, 1H), 4.36 (d, J=12.0 Hz, 1H), 3.99~3.81 (m, 1H), 2.73 (t, J=7.8 Hz, 1H), 2.55 (q, J=9.0 Hz, 1H), 2.21~2.03 (m, 3H), 1.91 (dt, J=15.4, 8.8 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.2 (q, J=36.2 Hz), 178.7, 139.0, 132.4, 131.8, 129.2, 128.7, 128.2, 128.1, 127.6, 124.7, 115.5 (q, J=291.7 Hz), 110.6, 77.6, 69.2, 60.5, 51.4, 48.0, 32.1, 28.9; 19F NMR (376 MHz, Chloroform-d) δ: -77.05; IR (KBr) ν: 3201.3(NH stretching vibration), 2968.2, 2868.7, 1756.5 (C=O), 1709.8 (C=O), 1619.8 (NH deformation vibration), 1475.6, 1267.8, 1203.7, 1151.4, 1062.1, 1017.2, 849.0, 815.6 cm-1; HRMS (ESI) calcd for C22H19ClF3N2O2 (M+H)+ 435.1083, found 435.1086.
7-溴-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4ak): 白色固体, 40.2 mg, 产率84%. m.p. 158.5 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.29 (s, 1H), 7.50 (d, J=7.2 Hz, 1H), 7.34 (s, 1H), 7.12~6.97 (m, 4H), 6.87 (d, J=7.6 Hz, 2H), 4.75 (dd, J=12.0, 8.2 Hz, 1H), 4.38 (d, J=12.0 Hz, 1H), 3.96~3.87 (m, 1H), 2.73 (t, J=7.6 Hz, 1H), 2.56 (td, J=9.2, 6.2 Hz, 1H), 2.21~2.06 (m, 3H), 1.92 (qd, J=11.4, 10.8, 6.0 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.3 (q, J=36.1 Hz), 178.4, 140.0, 132.3, 131.9, 131.5, 128.2, 127.6, 124.3, 123.0, 115.5 (q, J=291.7 Hz), 102.8, 78.7, 69.3, 60.6, 51.3, 48.0, 32.2, 29.0; 19F NMR (376 MHz, Chloroform-d) δ: -77.12; IR (KBr) ν: 3191.1 (NH stretching vibration), 2968.3, 2868.2, 1756.9 (C=O), 1712.2 (C=O), 1618.4 (NH deformation vibration), 1472.6, 1452.0, 1271.3, 1204.5, 1150.9, 1016.2, 844.5, 766.1, 740.0 cm-1; HRMS (ESI) calcd for C22H19BrF3N2O2 (M+H)+ 479.0577, found 479.0577.
5-溴-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4al): 黄色固体, 41.6 mg, 产率87%. m.p. 103.0 ℃; 1H NMR (400 MHz, Chloroform-d) δ: 8.30 (s, 1H), 7.68 (d, J=1.8 Hz, 1H), 7.32 (dd, J=8.2, 2.0 Hz, 1H), 7.09 (t, J=7.2 Hz, 1H), 7.01 (t, J=7.4 Hz, 2H), 6.87 (d, J=7.6 Hz, 2H), 6.46 (d, J=8.2 Hz, 1H), 4.71 (dd, J=12.0, 8.2 Hz, 1H), 4.36 (d, J=12.0 Hz, 1H), 3.97~3.82 (m, 1H), 2.73 (t, J=7.6 Hz, 1H), 2.54 (td, J=9.2, 6.2 Hz, 1H), 2.21~2.04 (m, 3H), 1.90 (dt, J=14.8, 8.2 Hz, 1H); 13C NMR (101 MHz, Chloroform-d) δ: 191.2 (q, J=36.2 Hz), 178.7, 139.5, 132.4, 132.1, 128.2, 128.2, 127.6, 127.5, 115.9, 115.5 (q, J=291.7 Hz), 111.1, 77.6, 69.2, 60.5, 51.4, 48.0, 32.1, 28.8; 19F NMR (376 MHz, Chloroform-d) δ: -77.09; IR (KBr) ν: 3179.1 (NH stretching vibration), 2967.4, 2868.9, 1756.3 (C=O), 1709.6 (C=O), 1618.0 (NH deformation vibration), 1473.3, 1266.4, 1200.1, 1150.8, 1016.5, 847.9, 813.5, 739.8 cm-1; HRMS (ESI) calcd for C22H19BrF3N2O2 (M+H)+ 479.0577, found 479.0577.
5-碘-2'-苯基-1'-(2,2,2-三氟乙酰基)-1',2',5',6',7',7a'-六氢螺[吲哚啉-3,3'-吡咯里嗪]-2-酮(4am): 白色固体, 39.5 mg, 产率75%. m.p. 95.1~95.6 ℃; IR (KBr) ν: 3213.5 (NH stretching vibration), 2963.2, 2869.2, 1757.2 (C=O), 1710.1 (C=O), 1614.8 (NH deformation vibration), 1471.6, 1429.5, 1268.6, 1201.4, 1150.3, 1081.8, 1016.0, 947.9, 890.1, 811.4, 736.4 cm-1; HRMS (ESI) calcd for C22H19IF3N2O2 (M+H)+ 527.0438, found 527.0434.