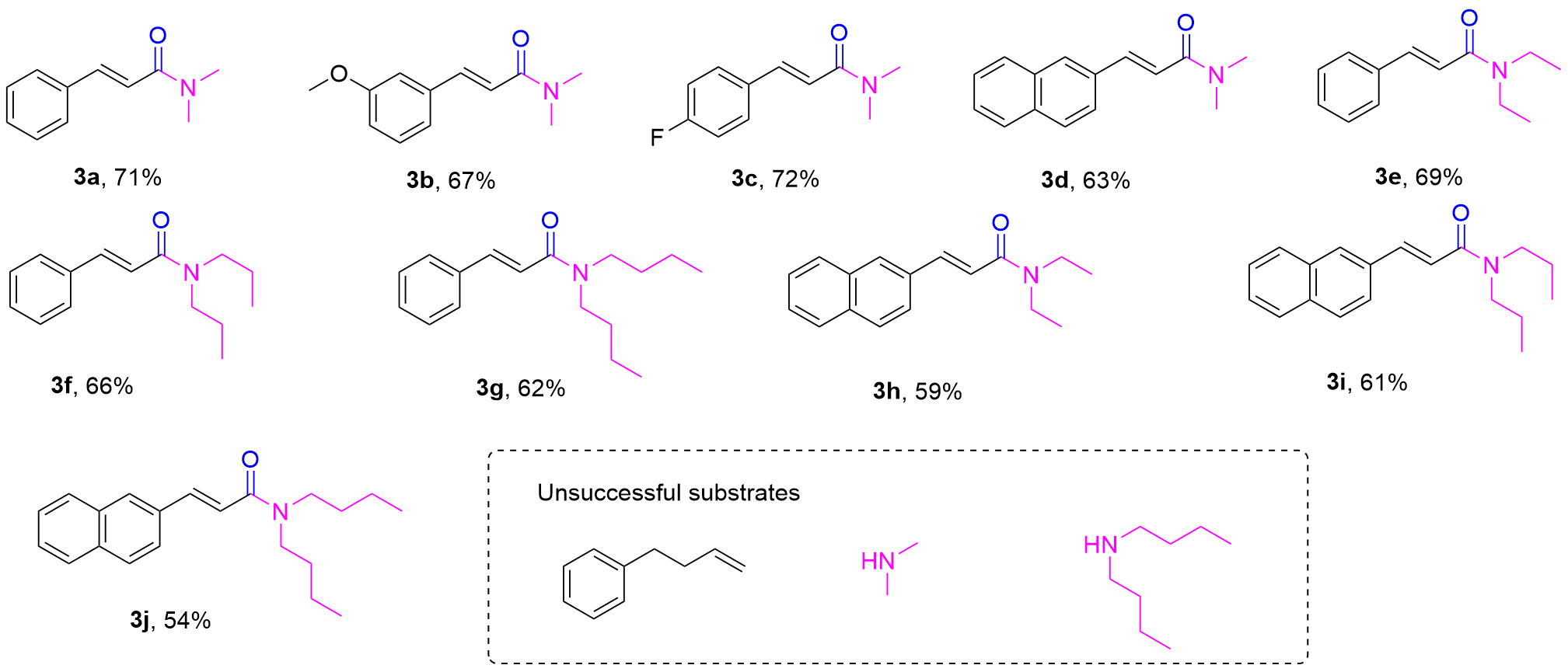
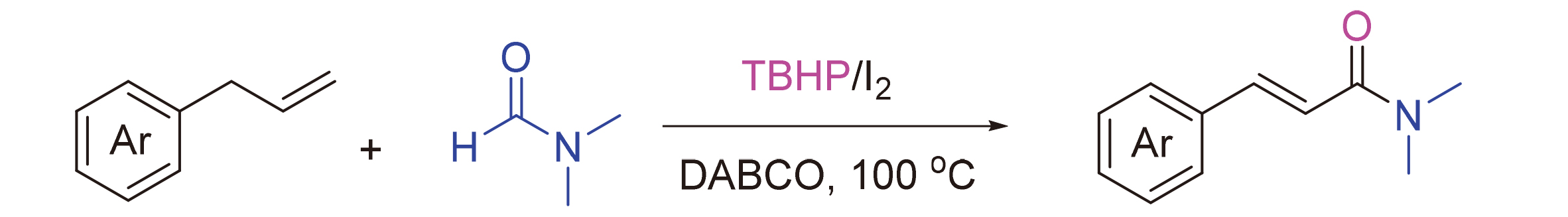
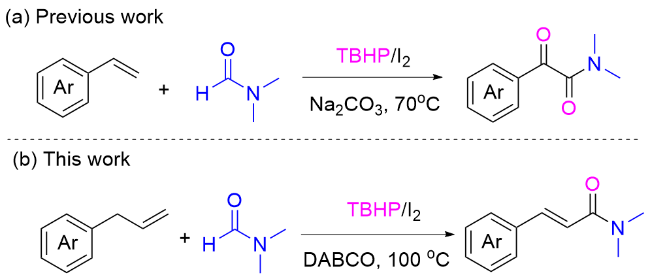
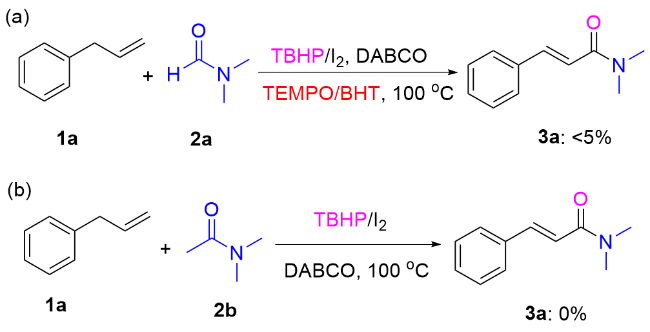
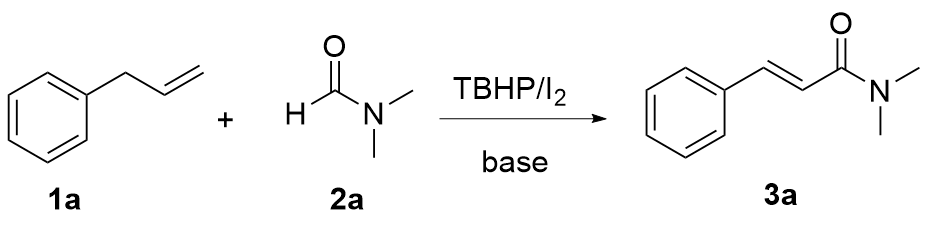Our investigation began with the examination of the oxidative coupling of allylbenzene (
1a) with DMF (
2a) in the presence of TBHP (
Table 1). A mixture of
1a (1 equiv.), TBHP (4 equiv.) and Na
2CO
3 (1 equiv.) in the presence of iodine (0.1 equiv.) and DMF (
2a) reacted at 70 ℃ for 24 h. Gratifyingly, the target unsaturated amide
3a was obtained in 48% yield (
Table 1, Entry 1). Then, a range of bases including CH
3COONa,
t-BuOK, 1,8-diazabicyclo- [5.4.0]undec-7-ene (DBU),
N,
N-dimethylpyridin-4-amine (DMAP), Et
3N and 1,4-diazabicyclo[2.2.2]octane (DABCO) were screened, among which DABCO was identified as one of the best additives (Entries 2~7). However, increasing the amount of DABCO was not helpful for increasing the yield (Entries 9, 10). Both base and I
2 are essential for this reaction. In the absence of base or I
2, no or only a trace amount of
3a could be observed (Entries 8, 11). In the further investigation, when the amount of I₂ was increased from 0.1 equiv. to 0.5 equiv., the yield of
3a decreased obviously (Entry 12). Subsequently, the reaction conditions were optimized by adjusting the amount of TBHP. The results indicated that the optimal amount of TBHP was 4 equiv., and further increasing it to 6 equiv. did not improve the yield of
3a (Entries 13~15). Next, the effect of temperature on the reaction was also investigated. To our delight, the reaction was found to be more effective when the temperature was increased to 100 ℃ and the reaction time was shortened to 6 h, affording
3a in 75% yield (Entries 16, 17). When the temperature was further increased to 120 ℃, no significant increase in yield was observed (Entries 18). Finally, to clarify the role of DMF, control experiments were performed. When allylbenzene (
1a) reacted with 2 equiv. of DMF in dimethyl sulfoxide (DMSO), PhCl or dioxane solvents in the presence of TBHP/I
2, the major product
3a was obtained in 66%, 59%, and 63% yields, respectively (Entries 19~21). It suggests that DMF serves not only as a reactant but also as a solvent in this transformation.