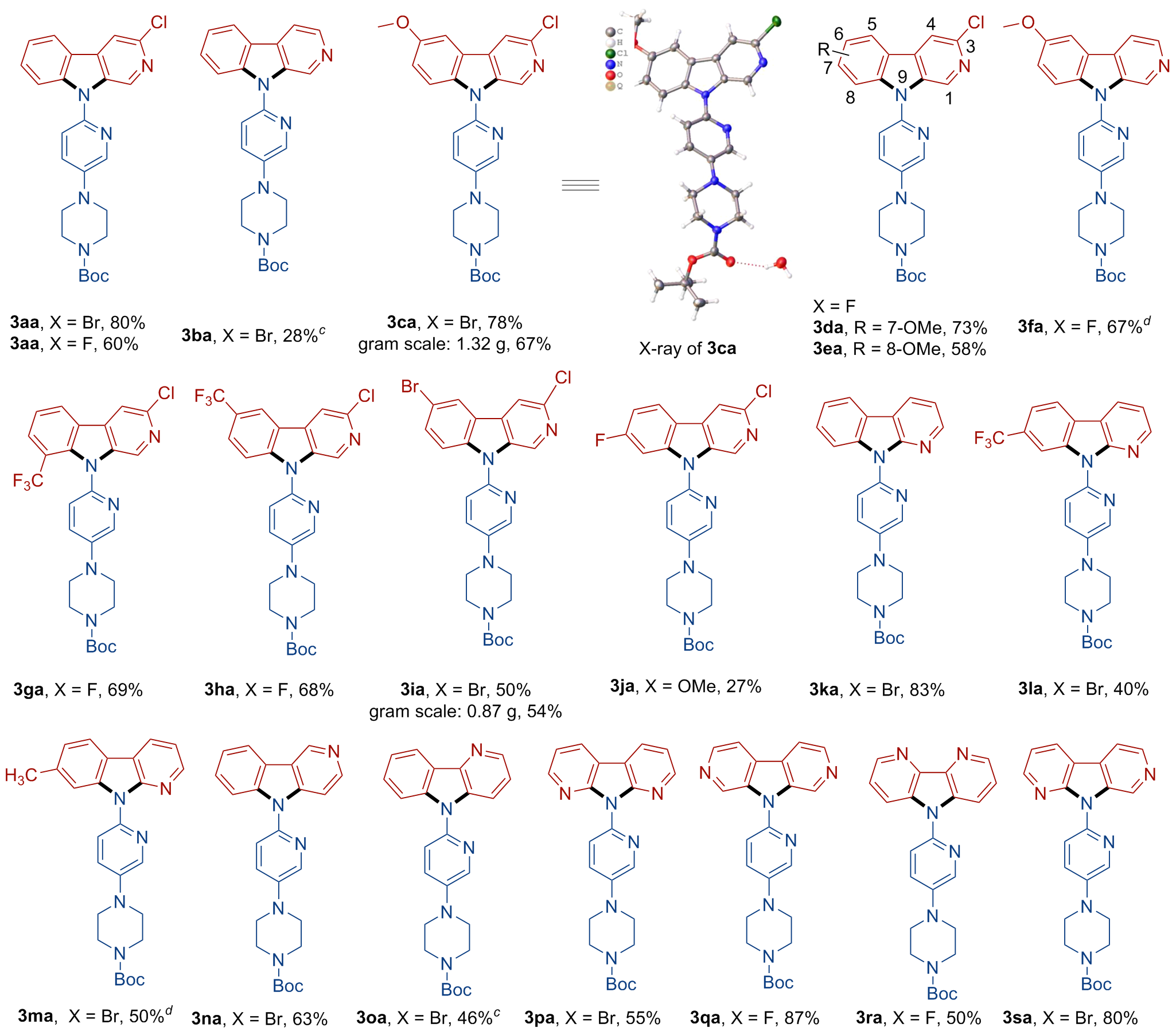
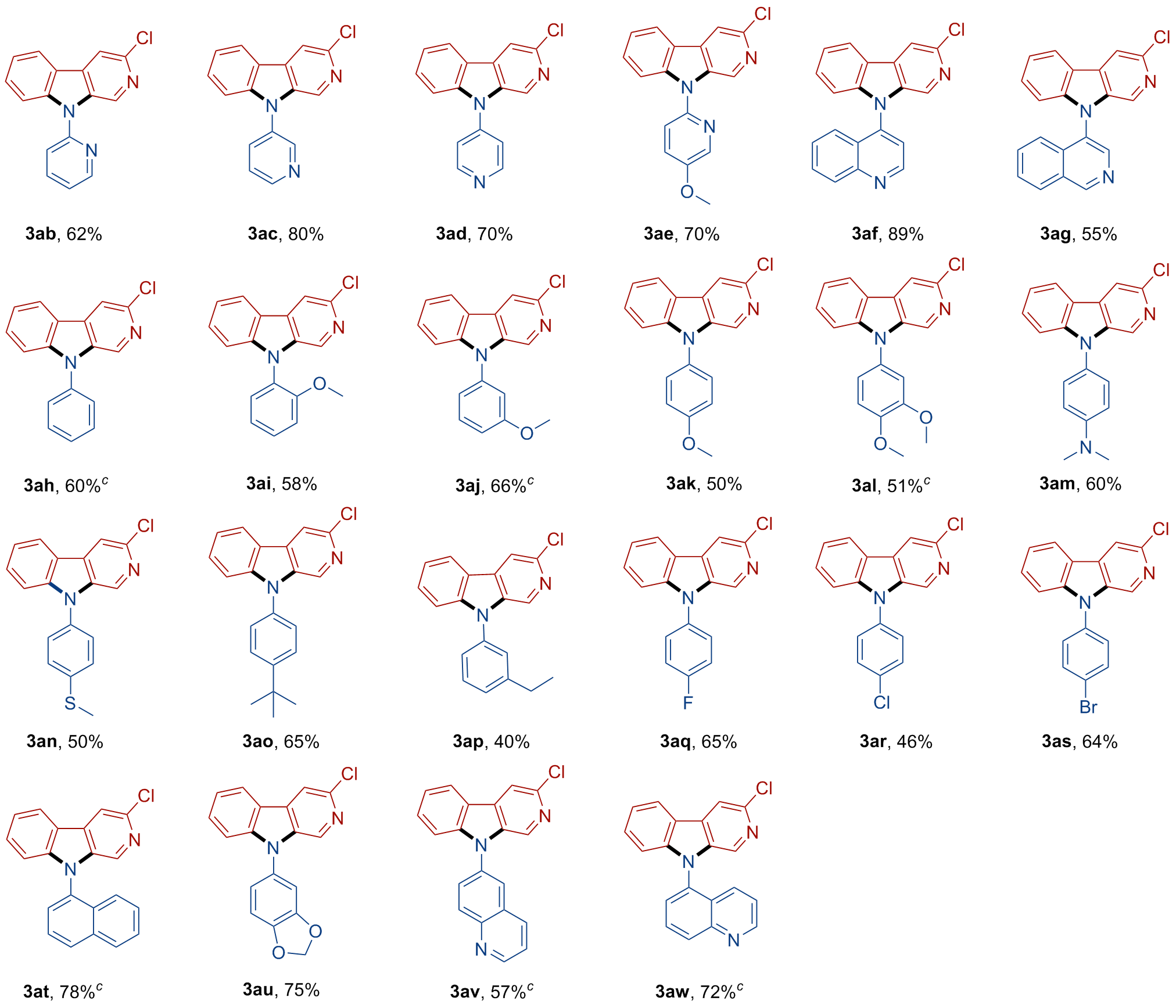
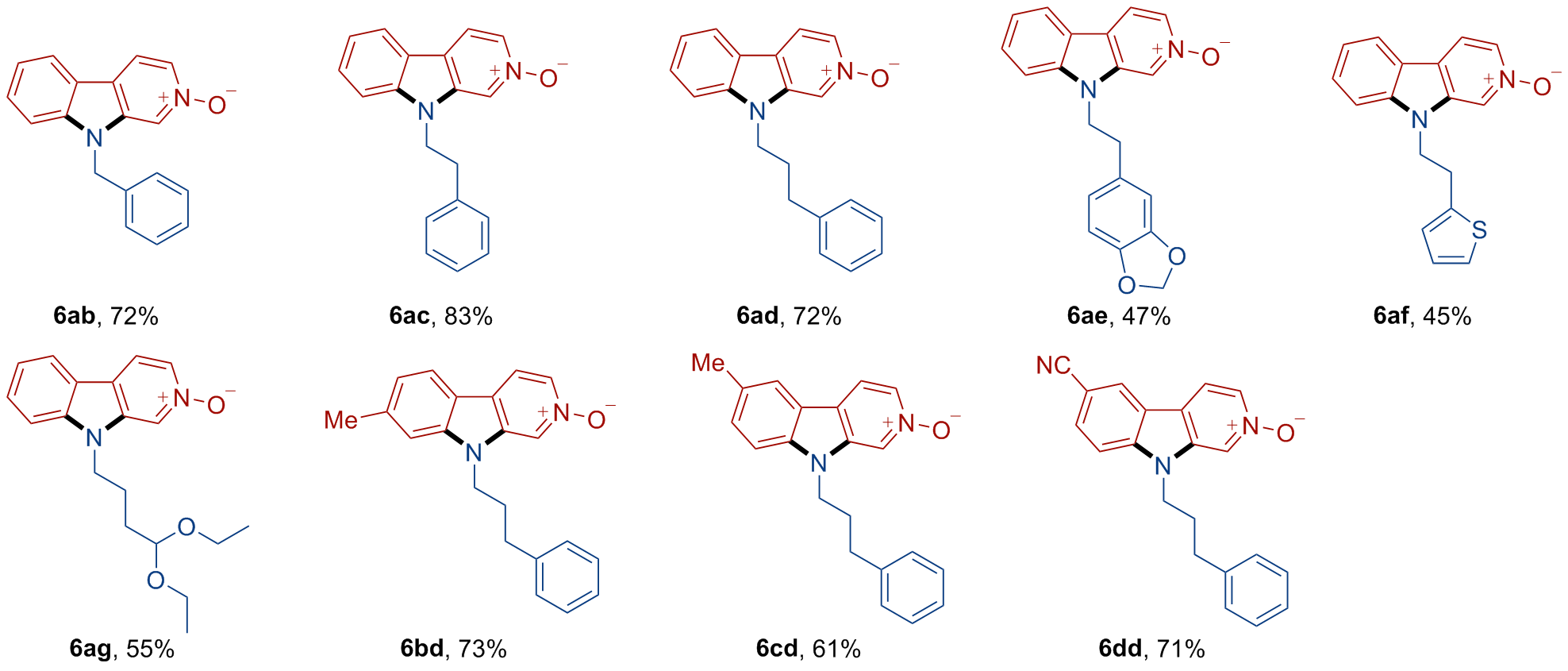
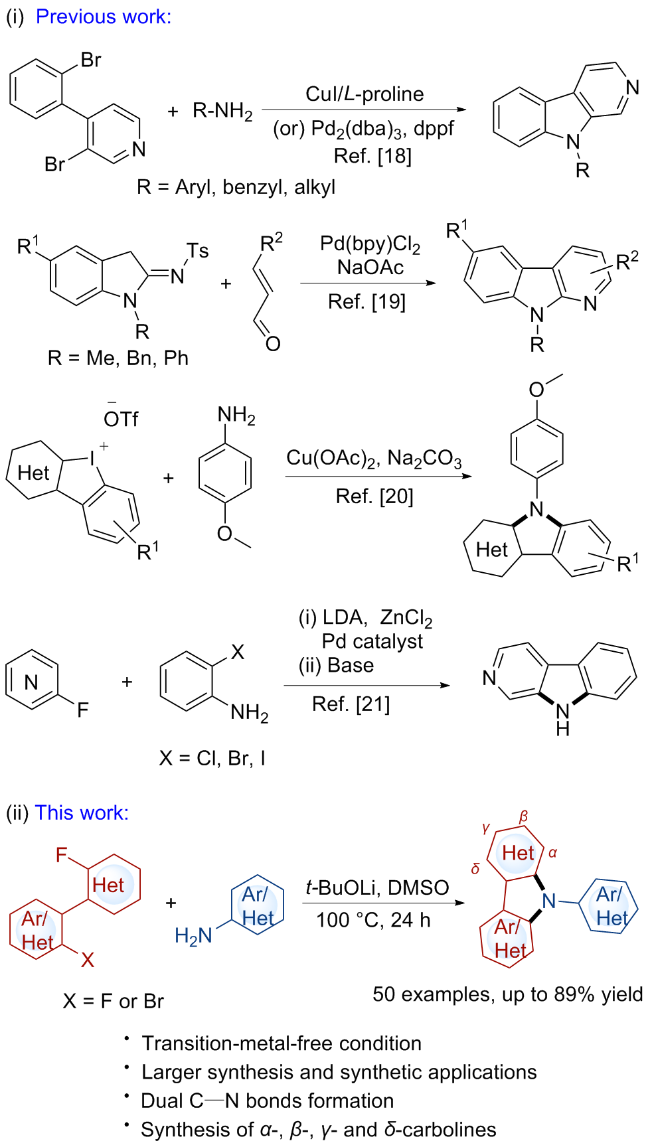
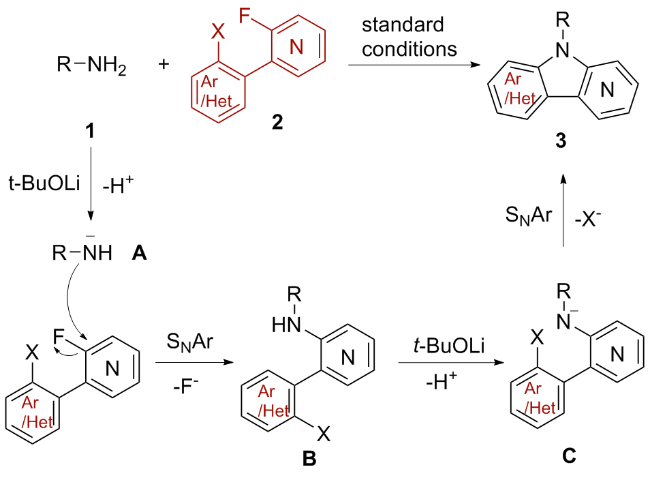
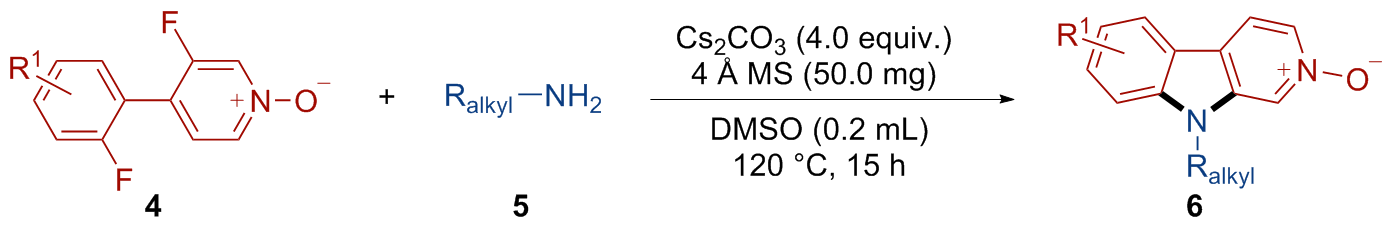A 10 mL oven-dried Schlenk tube equipped with a magnetic stirrer was charged with compound 1 (0.1 mmol, 1.0 equiv.), arylamine 2 (0.12 mmol, 1.2 equiv.), and t-BuOLi (0.6 mmol, 6.0 equiv.). Then, the tube was evacuated and backfilled with nitrogen three times. Subsequently, 0.5 mL of dry DMSO was added using a syringe under nitrogen. The tube was then sealed, and the mixture was stirred at 100 ℃ for 24 h. After that, the resulting mixture was quenched with H2O and extracted with EtOAc (10 mL×3). The combined organic phase was washed with brine (10 mL), dried over anhydrous Na2SO4 and concentrated in vacuo to give a residue. The residue was purified by pTLC to afford the desired product 3.
tert-Butyl 4-(6-(3-chloro-9H-pyrido[3,4-b]indol-9-yl)- pyridin-3-yl)piperazine-1-carboxylate (3aa): White solid, 37.0 mg, 80% yield. m.p. 72~74 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.87 (s, 1H), 8.36 (d, J=2.5 Hz, 1H), 8.12 (d, J=7.8 Hz, 1H), 7.98 (s, 1H), 7.73 (d, J=8.4 Hz, 1H), 7.58 (t, J=7.8 Hz, 1H), 7.50~7.44 (m, 2H), 7.35 (t, J=7.5 Hz, 1H), 3.67~3.65 (m, 4H), 3.30~3.28 (m, 4H), 1.51 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.74, 145.89, 142.46, 141.60, 141.49, 137.92, 135.54, 133.34, 132.88, 129.64, 125.57, 122.05, 121.49, 121.25, 119.23, 114.48, 111.73, 80.40, 48.69, 43.02, 28.56; 19F NMR (471 MHz, CDCl3) δ: 131.31 (s). HRMS (ESI) calcd for C25H27- ClN5O2 [M+H]+ 464.1848, found 464.1842.
tert-Butyl 4-(6-(9H-pyrido[3,4-b]indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ba): Yellow solid, 12.0 mg, 28% yield. m.p. 50~52 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.16 (s, 1H), 8.54 (d, J=5.2 Hz, 1H), 8.40 (d, J=2.9 Hz, 1H), 8.17 (d, J=7.8 Hz, 1H), 8.00 (d, J=5.0 Hz, 1H), 7.77 (d, J=8.4 Hz, 1H), 7.61~7.58 (m, 1H), 7.53 (d, J=8.7 Hz, 1H), 7.47 (dd, J=8.8, 3.0 Hz, 1H), 7.35 (t, J=7.3 Hz, 1H), 3.69 (t, J=5.0 Hz, 4H), 3.32 (t, J=5.0 Hz, 4H), 1.53 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.76, 145.80, 142.88, 140.77, 140.23, 137.97, 136.27, 134.13, 129.72, 128.89, 125.67, 122.13, 121.85, 121.22, 119.44, 114.60, 111.63, 80.39, 48.80, 43.11, 28.57. HRMS (ESI) calcd for C25H28N5O2 [M+H]+ 430.2238, found 430.2245.
tert-Butyl 4-(6-(3-chloro-6-methoxy-9H-pyrido[3,4-b]- indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ca): Yellow solid, 38.4 mg, 78% yield. m.p. 80~83 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.89 (s, 1H), 8.35 (d, J=2.0 Hz, 1H), 7.94 (s, 1H), 7.69 (d, J=9.1 Hz, 1H), 7.53 (d, J=2.5 Hz, 1H), 7.46 (d, J=4.1 Hz, 2H), 7.22 (dd, J=9.1, 2.5 Hz, 1H), 3.94 (s, 3H), 3.67 (t, J=5.0 Hz, 4H), 3.29 (t, J=5.0 Hz, 4H), 1.52 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.99, 154.63, 145.56, 142.62, 140.87, 137.73, 136.31, 135.62, 133.38, 132.47, 125.57, 121.55, 119.33, 118.67, 114.22, 112.74, 103.44, 80.27, 55.96, 48.63, 43.46, 28.44. HRMS (ESI) calcd for C26H29ClN5O3 [M+H]+ 494.1954, found 494.1960.
tert-Butyl 4-(6-(3-chloro-7-methoxy-9H-pyrido[3,4-b]- indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3da): Yellow solid, 36.0 mg, 73% yield. m.p. 78~80 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.69 (s, 1H), 8.31 (s, 1H), 7.91 (d, J=8.5 Hz, 1H), 7.80 (s, 1H), 7.41 (s, 2H), 7.12 (s, 1H), 6.90 (d, J=8.5 Hz, 1H), 3.84 (s, 3H), 3.63 (s, 4H), 3.26 (s, 4H), 1.49 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 161.72, 154.62, 145.73, 143.20, 142.19, 141.47, 137.70, 135.66, 132.98, 132.27, 125.40, 122.74, 119.10, 114.65, 113.46, 110.77, 95.03, 80.27, 55.68, 48.46, 43.08, 28.44. HRMS (ESI) calcd for C26H29ClN5O3 [M+H]+ 494.1954, found 494.1960.
tert-Butyl 4-(6-(3-chloro-8-methoxy-9H-pyrido[3,4-b]- indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ea): White solid. 28.6 mg, 58% yield. m.p. 180~182 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.58 (s, 1H), 8.27 (d, J=2.8 Hz, 1H), 7.94 (s, 1H), 7.72 (d, J=7.9 Hz, 1H), 7.37 (dd, J=8.7, 3.0 Hz, 1H), 7.28~7.25 (m, 2H), 7.05 (d, J=7.9 Hz, 1H), 3.74 (s, 3H), 3.68 (t, J=5.1 Hz, 4H), 3.32 (t, J=5.1 Hz, 4H), 1.52 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.63, 147.39, 146.07, 143.26, 141.14, 137.06, 136.18, 133.46, 132.56, 131.49, 124.11, 122.98, 122.23, 121.73, 114.30, 114.17, 110.83, 80.25, 55.86, 48.48, 43.16, 28.44. HRMS (ESI) calcd for C26H29ClN5O3 [M+H]+ 494.1954, found 494.1958.
tert-Butyl 4-(6-(6-methoxy-9H-pyrido[3,4-b]indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3fa): Yellow solid, 30.7 mg, 67% yield. m.p. 56~57 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.45 (d, J=5.2 Hz, 1H), 8.33 (d, J=2.9 Hz, 1H), 7.92~7.91 (m, 1H), 7.69 (d, J=9.0 Hz, 1H), 7.56 (d, J=2.5 Hz, 1H), 7.47 (d, J=8.7 Hz, 1H), 7.41 (dd, J=8.8, 3.0 Hz, 1H), 7.18 (dd, J=9.0, 2.5 Hz, 1H), 3.92 (s, 3H), 3.64 (t, J=5.0 Hz, 4H), 3.25 (t, J=5.0 Hz, 4H), 1.49 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.86, 154.63, 145.45, 142.97, 139.62, 137.74, 136.38, 135.45, 134.17, 129.32, 125.65, 122.45, 118.87, 118.42, 114.37, 112.59, 103.49, 80.24, 55.98, 48.69, 43.29, 28.43. HRMS (ESI) calcd for C26H30N5O3 [M+H]+ 460.2344, found 460.2349.
tert-Butyl 4-(6-(3-chloro-8-(trifluoromethyl)-9H-pyrido- [3,4-b]indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ga): Yellow solid, 36.6 mg, 69% yield. m.p. 160~162 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.27~8.30 (m, 2H), 8.19 (s, 1H), 7.96 (s, 1H), 7.86 (d, J=7.6 Hz, 1H), 7.34~7.40 (m, 3H), 3.65 (t, J=5.1 Hz, 4H), 3.33 (t, J=5.1 Hz, 4H), 1.49 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.65, 147.20, 142.08, 141.65, 138.62, 137.48, 133.29, 131.39, 128.18 (d, J=5.7 Hz), 126.02, 123.99, 123.79, 123.40 (d, J=272.0 Hz), 123.22, 120.38, 115.04 (q, J=33.5 Hz), 114.13, 80.34, 47.97, 43.23, 28.49; 19F NMR (471 MHz, CDCl3) δ: 57.10. HRMS (ESI) calcd for C26H26ClF3N5O2 [M+H]+ 532.1722, found 532.1730.
tert-Butyl 4-(6-(3-chloro-6-(trifluoromethyl)-9H-pyrido- [3,4-b]indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ha): Yellow solid, 36.1 mg, 68% yield. m.p. 111~112 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.86 (s, 1H), 8.37 (d, J=15.2 Hz, 2H), 8.01 (s, 1H), 7.79 (s, 2H), 7.47 (s, 2H), 3.67 (t, J=5.1 Hz, 4H), 3.32 (t, J=5.1 Hz, 4H), 1.51 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.72, 146.28, 142.94, 142.18, 141.42, 137.87, 136.05, 133.73, 132.23, 126.24 (q, J=3.3 Hz), 125.33, 124.82 (q, J=234.5 Hz), 123.61 (q, J=4.6 Hz), 120.80, 119.75 (q, J=4.0 Hz), 119.36, 114.74, 112.25, 80.46, 48.43, 43.59, 28.53. 19F NMR (471 MHz, CDCl3) δ: 60.78. HRMS (ESI) calcd for C26H26ClF3N5O2 [M+H]+ 532.1722, found 532.1732.
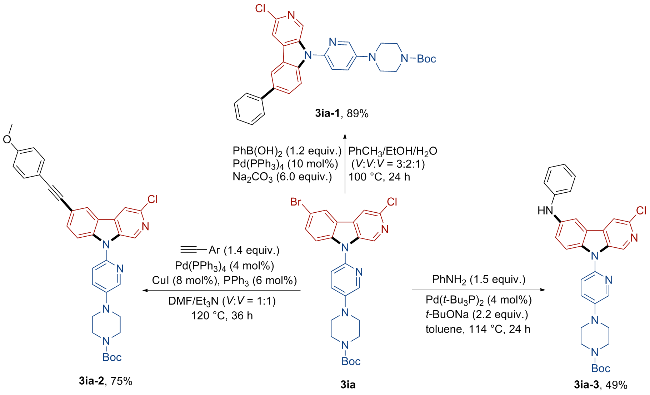
tert-Butyl 4-(6-(6-bromo-3-chloro-9H-pyrido[3,4-b]in- dol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ia): Bro- wn solid, 27.0 mg, 50% yield. m.p. 118~100 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.83 (s, 1H), 8.33 (s, 1H), 8.20 (d, J=1.5 Hz, 1H), 7.90 (s, 1H), 7.64~7.59 (m, 2H), 7.43 (s, 2H), 3.65 (t, J=5.0 Hz, 4H), 3.29 (t, J=5.0 Hz, 4H), 1.50 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.62, 145.95, 141.71, 141.59, 140.08, 137.74, 135.50, 133.45, 132.30, 131.50, 125.35, 124.58, 122.70, 119.04, 114.51, 114.07, 113.29, 80.34, 48.44, 43.01, 28.44. HRMS (ESI) calcd for C25H26BrClN5O2 [M+H]+ 542.0953, found 542.0963.
tert-Butyl 4-(6-(3-chloro-7-fluoro-9H-pyrido[3,4-b]in- dol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ja): Yellow solid. Yellow solid, 13.0 mg, 27% yield. m.p. 85~88 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.80 (s, 1H), 8.35 (s, 1H), 8.05 (dd, J=8.6, 5.3 Hz, 1H), 7.92 (s, 1H), 7.46 (d, J=1.5 Hz, 2H), 7.42 (dd, J=9.9, 2.1 Hz, 1H), 7.08 (td, J=8.8, 2.2 Hz, 1H), 3.66 (t, J=5.2 Hz, 4H), 3.30 (t, J=5.2 Hz, 4H), 1.51 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 164.00 (d, J=246.9 Hz), 154.62, 145.98, 142.42 (d, J=12.5 Hz), 141.81 (d, J=8.1 Hz), 137.77, 135.94, 132.86, 132.42, 125.38, 123.25 (d, J=10.7 Hz), 118.95, 117.48, 114.11, 110.11 (d, J=24.8 Hz), 98.79 (d, J=27.7 Hz), 80.32, 48.45, 43.26, 28.44; 19F NMR (471 MHz, CDCl3) δ: 108.79 (s). HRMS (ESI) calcd for C25H26ClFN5O2 [M+H]+ 482.1754, found 482.1761.
tert-Butyl 4-(6-(9H-pyrido[2,3-b]indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ka): White solid, 35.6 mg, 83% yield. m.p. 94~96 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.48 (d, J=4.4 Hz, 1H), 8.36~8.34 (m, 2H), 8.08 (d, J=7.7 Hz, 1H), 7.96 (d, J=8.3 Hz, 1H), 7.87 (d, J=8.7 Hz, 1H), 7.51~7.45 (m, 2H), 7.33 (t, J=7.5 Hz, 1H), 7.23 (dd, J=7.7, 4.8 Hz, 1H), 3.63 (t, J=5.1 Hz, 4H), 3.23 (t, J=5.1 Hz, 4H), 1.50 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.65, 151.32, 146.06, 145.52, 142.80, 139.36, 137.36, 128.30, 127.22, 125.68, 121.21, 121.07, 120.87, 120.65, 116.99, 116.46, 112.34, 80.16, 48.95, 43.65, 28.46. HRMS (ESI) calcd for C25H28N5O2 [M+H]+ 430.2238, found 430.2245.
tert-Butyl 4-(6-(7-(trifluoromethyl)-9H-pyrido[2,3-b]- indol-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3la): Yellow solid, 19.8 mg, 40% yield. m.p. 178~180 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.55 (dd, J=4.8, 1.4 Hz, 1H), 8.41 (dd, J=7.7, 1.4 Hz, 1H), 8.36 (d, J=3.0 Hz, 1H), 8.29 (s, 1H), 8.16 (d, J=8.1 Hz, 1H), 7.91 (d, J=8.8 Hz, 1H), 7.58 (d, J=8.1 Hz, 1H), 7.49 (dd, J=8.8, 3.1 Hz, 1H), 7.29 (dd, J=7.7, 4.8 Hz, 1H), 3.65 (t, J=5.0 Hz, 4H), 3.28 (t, J=5.0 Hz, 4H), 1.50 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.77, 152.05, 147.61, 145.90, 142.20, 138.67, 137.45, 129.28, 129.00 (q, J=32.1 Hz), 125.66, 124.76 (q, J=272.6 Hz), 123.78, 121.11, 120.92, 117.95 (q, J=3.9 Hz), 117.13, 116.04, 110.21 (q, J=4.4 Hz), 80.33, 48.90, 43.39, 28.55; 19F NMR (471 MHz, CDCl3) δ: 61.15. HRMS (ESI) calcd for [M+H]+ C26H27F3N5O2 498.2112, found 498.2118.
tert-Butyl 4-(6-(7-methyl-9H-pyrido[2,3-b]indol-9-yl)- pyridin-3-yl)piperazine-1-carboxylate (3ma): White solid, 22.1 mg, 50% yield. m.p. 159~162 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.43 (dd, J=4.9, 1.5 Hz, 1H), 8.36 (d, J=3.0 Hz, 1H), 8.29 (dd, J=7.6, 1.5 Hz, 1H), 7.95 (d, J=7.9 Hz, 1H), 7.84 (d, J=8.8 Hz, 1H), 7.74 (s, 1H), 7.47 (dd, J=8.8, 3.1 Hz, 1H), 7.20 (dd, J=7.6, 4.9 Hz, 1H), 7.15 (d, J=7.9 Hz, 1H), 3.64 (t, J=5.0 Hz, 4H), 3.25 (t,J=5.0 Hz, 4H), 2.52 (s, 3H), 1.50 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.66, 151.49, 145.48, 142.87, 139.76, 137.59, 137.41, 127.78, 125.72, 122.67, 120.99, 120.36, 118.73, 117.10, 116.37, 112.27, 80.16, 48.98, 43.18, 28.45, 22.37. HRMS (ESI) calcd for C26H30N5O2 [M+H]+ 444.2395, found 444.2399.
tert-Butyl 4-(6-(5H-pyrido[4,3-b]indol-5-yl)pyridin-3-yl)piperazine-1-carboxylate (3na): Brown solid, 27.0 mg, 63% yield. m.p. 71~73 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.35 (s, 1H), 8.52 (d, J=5.7 Hz, 1H), 8.35 (d, J=2.7 Hz, 1H), 8.17 (d, J=7.7 Hz, 1H), 7.68 (d, J=8.3 Hz, 1H), 7.55 (d, J=5.8 Hz, 1H), 7.50 - 7.42 (m, 3H), 7.37 (t, J=7.5 Hz, 1H), 3.65 (t, J=5.0 Hz, 4H), 3.28 (t, J=5.0 Hz, 4H), 1.50 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.62, 145.89, 145.25, 144.24, 142.66, 142.23, 140.00, 137.79, 127.16, 125.32, 122.08, 121.78, 120.46, 119.60, 111.11, 106.09, 80.30, 48.53, 43.33, 28.44. HRMS (ESI) calcd for C25H28N5O2 [M+H]+ 430.2238, found 430.2244.
tert-Butyl 4-(6-(5H-pyrido[3,2-b]indol-5-yl)pyridin-3-yl)piperazine-1-carboxylate (3oa): Yellow solid, 19.7 mg, 46% yield. m.p. 151~154 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.60 (dd, J=4.7, 1.2 Hz, 1H), 8.42 (d, J=7.8 Hz, 1H), 8.34 (d, J=2.8 Hz, 1H), 8.02 (dd, J=8.3, 1.2 Hz, 1H), 7.72 (d, J=8.3 Hz, 1H), 7.54~7.51 (m, 1H), 7.47 (d, J=8.7 Hz, 1H), 7.43 (dd, J=8.8, 3.0 Hz, 1H), 7.39~7.36 (m, 1H), 7.33 (dd, J=8.3, 4.7 Hz, 1H), 3.65 (t, J=5.1 Hz, 4H), 3.26 (t, J=5.1 Hz, 4H), 1.50 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.64, 145.51, 142.97, 142.92, 142.57, 140.51, 137.78, 133.55, 128.07, 125.59, 122.96, 121.29, 120.89, 120.35, 119.23, 118.25, 110.84, 80.27, 48.71, 43.31, 28.45. HRMS (ESI) calcd for C25H28N5O2 [M+H]+ 430.2238, found 430.2242.
tert-Butyl 4-(6-(9H-pyrrolo[2,3-b:5,4-b']dipyridin-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3pa): Yellow solid, 23.6 mg, 55% yield. m.p. 151~154 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.53 (d, J=4.8 Hz, 2H), 8.42 (d, J=2.8 Hz, 1H), 8.33 (d, J=7.6 Hz, 2H), 7.63 (d, J=8.7 Hz, 1H), 7.45 (dd, J=8.7, 2.9 Hz, 1H), 7.27~7.24 (m, 2H), 3.61 (t, J=5.1 Hz, 4H), 3.25 (t, J=5.1 Hz, 4H), 1.48 (s, 9H); 13C NMR (126 MHz, CD3OD) δ: 156.32, 152.53, 148.63, 148.07, 140.67, 138.25, 131.37, 126.58, 124.95, 118.72, 115.95, 81.49, 49.29, 44.32, 28.67. HRMS (ESI) calcd for C24H27N6O2 [M+H]+ 431.2190, found 431.2190.
tert-Butyl 4-(6-(9H-pyrrolo[2,3-c:5,4-c']dipyridin-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3qa): Yellow solid, 37.4 mg, 87% yield. m.p. 78~83 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.26 (s, 2H), 8.57 (d, J=4.8 Hz, 2H), 8.35 (d, J=2.9 Hz, 1H), 8.03 (d, J=5.0 Hz, 2H), 7.53 (d, J=8.7 Hz, 1H), 7.46 (dd, J=8.7, 2.9 Hz, 1H), 3.64 (t, J=5.0 Hz, 4H), 3.29 (t, J=5.0 Hz, 4H), 1.48 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.60, 145.98, 141.59, 140.35, 137.72, 136.24, 135.34, 127.72, 125.40, 118.83, 115.60, 80.28, 48.42, 43.11, 28.42. HRMS (ESI) calcd for C24H27N6O2 [M+H]+ 431.2190, found 431.2195.
tert-Butyl 4-(6-(9H-pyrrolo[2,3-b:5,4-c']dipyridin-9-yl)pyridin-3-yl)piperazine-1-carboxylate (3ra): Yellow solid, 21.5 mg, 50% yield. m.p. 78~80 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.72 (d, J=3.8 Hz, 2H), 8.32 (t, J=1.6 Hz, 1H), 8.08 (dd, J=8.4, 1.0 Hz, 2H), 7.45 (d, J=1.6 Hz, 2H), 7.40 (dd, J=8.4, 4.6 Hz, 2H), 3.65 (t, J=5.0 Hz, 4H), 3.27 (t, J=5.0 Hz, 4H), 1.49 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.61, 145.70, 144.34, 142.18, 140.89, 137.73, 134.23, 125.60, 121.79, 118.77, 118.70, 80.30, 48.60, 43.16, 28.43. HRMS (ESI) calcd for [M+H]+ C24H27N6O2 431.2190, found 431.2197.
tert-Butyl 4-(6-(5H-pyrrolo[3,2-b:4,5-b']dipyridin-5-yl)pyridin-3-yl)piperazine-1-carboxylate (3sa): Colourless liquid, 34.4 mg, 80% yield. 1H NMR (500 MHz, CDCl3) δ: 9.55 (s, 1H), 8.65 (dd, J=4.8, 1.6 Hz, 1H), 8.60~8.59 (m, 1H), 8.45 (dd, J=7.7, 1.6 Hz, 1H), 8.35 (d, J=3.0 Hz, 1H), 8.06 (d, J=8.9 Hz, 1H), 7.97 (d, J=5.1 Hz, 1H), 7.50 (dd, J=8.9, 3.1 Hz, 1H), 7.32 (dd, J=7.7, 4.8 Hz, 1H), 3.66 (t, J=5.0 Hz, 4H), 3.27 (t, J=5.0 Hz, 4H), 1.51 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 154.63, 151.32, 149.04, 145.59, 142.25, 140.88, 137.06, 136.18, 135.16, 130.25, 126.92, 125.54, 119.95, 117.10, 115.13, 114.65, 80.17, 48.83, 43.20, 28.43. HRMS (ESI) calcd for C24H27- N6O2 [M+H]+ 431.2190, found 431.2197.
3-Chloro-9-(pyridin-2-yl)-9H-pyrido[3,4-b]indole (3ab): White solid, 17.3 mg, 62% yield. m.p. 154~156 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.04 (s, 1H), 8.73 (dd, J=4.8, 1.1 Hz, 1H), 8.12 (d, J=7.8 Hz, 1H), 7.99~7.96 (m, 2 H), 7.88 (d, J=8.4 Hz, 1H), 7.64 (d, J=8.1 Hz, 1H), 7.60 (ddd, J=8.4, 7.1, 1.1 Hz, 1H), 7.39~7.35 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 150.84, 149.96, 141.99, 141.05, 139.00, 135.16, 133.84, 133.39, 129.79, 122.05, 122.03, 121.97, 121.69, 118.48, 114.47, 112.08. HRMS (ESI) calcd for C16H11ClN3 [M+H]+ 280.0637, found 280.0631.
3-Chloro-9-(pyridin-3-yl)-9H-pyrido[3,4-b]indole (3ac): White solid, 22.3 mg, 80% yield. m.p. 170~172 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.90 (s, 1H), 8.79 (d, J=4.4 Hz, 1H), 8.52 (s, 1H), 8.15 (d, J=7.9 Hz, 1H), 8.01 (s, 1H), 7.91 (d, J=8.1 Hz, 1H), 7.63~7.58 (m, 2H), 7.42~7.37 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 149.47, 148.22, 142.48, 141.83, 136.23, 134.36, 133.58, 132.84, 131.59, 130.00, 124.92, 122.38, 121.75, 121.08, 114.85, 110.38. HRMS (ESI) calcd for C16H11ClN3 [M+H]+ 280.0637, found 280.0630.
3-Chloro-9-(pyridin-4-yl)-9H-pyrido[3,4-b]indole (3ad): White solid, 19.5 mg, 70% yield. m.p. 200~201 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.90 (d, J=4.5 Hz, 2H), 8.69 (s, 1H), 8.14 (d, J=7.9 Hz, 1H), 7.99 (s, 1H), 7.63~7.56 (m, 4H), 7.40 (ddd, J=7.9, 6.0, 2.0 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 152.18, 144.43, 142.33, 141.27, 135.14, 133.36, 131.93, 130.08, 122.44, 122.22, 121.54, 120.35, 114.89, 110.76. HRMS (ESI) calcd for C16H11ClN3 [M+H]+ 280.0637, found 280.0632.
3-Chloro-9-(5-methoxypyridin-2-yl)-9H-pyrido[3,4-b]indole (3ae): White solid, 21.6 mg, 70% yield. m.p. 159~161 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.86 (s, 1H), 8.39 (d, J=3.0 Hz, 1H), 8.11 (d, J=7.9 Hz, 1H), 7.97 (s, 1H), 7.72 (d, J=8.4 Hz, 1H), 7.60~7.56 (m, 1H), 7.53 (d, J=8.7 Hz, 1H), 7.47 (dd, J=8.7, 3.0 Hz, 1H), 7.35 (t, J=7.5 Hz, 1H), 3.97 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 154.68, 143.45, 141.53, 136.80, 135.49, 133.26, 132.92, 129.68, 123.78, 122.04, 121.56, 121.25, 119.50, 114.47, 111.64, 56.20. HRMS (ESI) calcd for C17H13ClN3O [M+H]+ 310.0742, found 310.0737.
3-Chloro-9-(quinolin-4-yl)-9H-pyrido[3,4-b]indole (3af): White solid, 29.2 mg, 89% yield. m.p. 172~174 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.17 (d, J=4.4 Hz, 1H), 8.33 (d, J=8.5 Hz, 1H), 8.25 (s, 1H), 8.21 (d, J=7.9 Hz, 1H), 8.08 (s, 1H), 7.84 (ddd, J=8.3, 6.7, 1.3 Hz, 1H), 7.59 (d, J=4.5 Hz, 1H), 7.55~7.52 (m, 1H), 7.50~7.47 (m, 1H), 7.41 (dd, J=16.5, 8.4 Hz, 2H), 7.13 (d, J=8.3 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 151.09, 150.44, 143.04, 141.94, 141.60, 136.75, 132.93, 132.36, 130.89, 130.59, 129.95, 128.04, 125.09, 123.16, 122.36, 121.81, 121.19, 120.13, 114.91, 111.25. HRMS (ESI) calcd for C20H13ClN3 [M+H]+ 330.0793, found 330.0786.
3-Chloro-9-(isoquinolin-4-yl)-9H-pyrido[3,4-b]indole (3ag): Brown solid, 18.0 mg, 55% yield. m.p. 137~138 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.49 (s, 1H), 8.74 (s, 1H), 8.23~8.20 (m, 3H), 8.09 (s, 1H), 7.74 (t, J=7.3 Hz, 1H), 7.65~7.62 (m, 1H), 7.54~7.51 (m, 1H), 7.39 (t, J=7.4 Hz, 1H), 7.27~7.25 (m, 1H), 7.08 (d, J=8.3 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 153.99, 143.81, 143.27, 141.63, 137.43, 133.30, 132.61, 132.20, 132.08, 129.90, 129.70, 128.72, 128.57, 128.12, 122.32, 121.92, 121.49, 120.92, 114.88, 111.02. HRMS (ESI) calcd for C20H13ClN3 [M+H]+ 330.0793, found 330.0786.
3-Chloro-9-phenyl-9H-pyrido[4-b]indole (3ah): Bro- wn solid, 16.6 mg, 60% yield. m.p. 104~106 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.55 (s, 1H), 8.14 (d, J=7.9 Hz, 1H), 8.01 (s, 1H), 7.64 (t, J=7.8 Hz, 2H), 7.58~7.51 (m, 4H), 7.45 (d, J=8.4 Hz, 1H), 7.36 (d, J=7.8 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 142.70, 141.13, 136.54, 136.38, 132.35, 132.10, 130.37, 129.58, 128.42, 126.83, 122.15, 121.09, 120.76, 114.60, 110.84. HRMS (ESI) calcd for C17H12ClN2 [M+H]+ 279.0684, found 279.0679.
3-Chloro-9-(2-methoxyphenyl)-9H-pyrido[3,4-b]indole (3ai): Brown solid, 17.8 mg, 58% yield. m.p. 39~41 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.28 (d, J=1.0 Hz, 1H), 8.14 (d, J=7.8 Hz, 1H), 8.01 (d, J=0.9 Hz, 1H), 7.56~7.52 (m, 2H), 7.46 (dd, J=7.6, 1.6 Hz, 1H), 7.34~7.31 (m, 1H), 7.24~7.16 (m, 3H), 3.73 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 155.57, 143.07, 140.65, 136.50, 132.73, 132.18, 130.37, 129.40, 129.31, 124.52, 121.98, 121.45, 120.74, 114.51, 112.83, 111.17, 55.76. HRMS (ESI) calcd for C18H14ClN2O [M+H]+ 309.0790, found 309.0781.
3-Chloro-9-(3-methoxyphenyl)-9H-pyrido[3,4-b]indole (3aj): Brown solid, 20.3 mg, 66% yield. m.p. 136~137 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.59 (s, 1H), 8.14 (d, J=7.9 Hz, 1H), 8.00 (s, 1H), 7.59~7.48 (m, 3H), 7.35 (t, J=7.5 Hz, 1H), 7.14 (d, J=7.8 Hz, 1H), 7.08~7.05 (m, 2H), 3.87 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 161.13, 142.62, 141.14, 137.61, 136.30, 132.36, 132.25, 131.08, 129.59, 122.13, 121.10, 120.77, 118.88, 114.58, 114.07, 112.47, 110.99, 55.70. HRMS (ESI) calcd for C18H14ClN2O [M+H]+ 309.0790, found 309.0783.
3-Chloro-9-(4-methoxyphenyl)-9H-pyrido[3,4-b]indole (3ak): Brown solid, 15.4 mg, 50% yield. m.p. 103~105 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.50 (s, 1H), 8.16 (d, J=7.9 Hz, 1H), 8.03 (s, 1H), 7.58 (t, J=7.7 Hz, 1H), 7.46 (d, J=8.8 Hz, 2H), 7.41~7.34 (m, 2H), 7.16 (d, J=8.8 Hz, 2H), 3.95 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 159.56, 143.20, 140.90, 136.83, 132.08, 129.51, 129.00, 128.31, 122.11, 120.86, 120.54, 115.53, 114.55, 110.80, 55.78. HRMS (ESI) calcd for C18H14ClN2O [M+H]+ 309.0790, found 309.0782.
3-Chloro-9-(3,4-dimethoxyphenyl)-9H-pyrido[3,4-b]indole (3al): Yellow oil, 17.2 mg, 51% yield. 1H NMR (500 MHz, CDCl3) δ: 8.51 (s, 1H), 8.15 (dd, J=8.0, 1.0 Hz, 1H), 8.01 (d, J=1.0 Hz, 1H), 7.57 (ddd, J=8.4, 7.1, 1.2 Hz, 1H), 7.42~7.40 (m, 1H), 7.35 (ddd, J=8.0, 7.1, 0.9 Hz, 1H), 7.12~7.07 (m, 2H), 7.00 (d, J=2.1 Hz, 1H), 4.01 (s, 3H), 3.89 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 150.13, 149.09, 143.05, 140.85, 136.68, 132.04, 131.98, 129.46, 129.04, 122.04, 120.81, 120.44, 119.32, 114.47, 111.90, 110.74, 110.21, 56.22, 56.20. HRMS (ESI) calcd for C19H16ClN2O2 [M+H]+ 339.0895, found 339.0891.
4-(3-Chloro-9H-pyrido[3,4-b]indol-9-yl)-N,N-dimethyl-aniline (3am): Yellow solid, 19.2 mg, 60% yield. m.p. 42~44 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.49 (s, 1H), 8.13 (d, J=7.9 Hz, 1H), 8.00 (s, 1H), 7.54 (ddd, J=8.2, 6.9, 1.0 Hz, 1H), 7.39~7.30 (m, 4H), 6.89 (d, J=9.0 Hz, 2H), 3.08 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 150.25, 143.40, 140.45, 136.96, 132.17, 131.74, 129.20, 127.80, 124.61, 121.90, 120.43, 120.28, 114.33, 113.07, 110.87. HRMS (ESI) calcd for C19H17ClN3 [M+H]+ 322.1106, found 322.0736.
3-Chloro-9-(4-(methylthio)phenyl)-9H-pyrido[3,4-b]indole (3an): Brown solid, 16.2 mg, 50% yield. m.p. 123~126 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.52 (s, 1H), 8.14 (d, J=7.9 Hz, 1H), 8.00 (s, 1H), 7.57 (t, J=7.6 Hz, 1H), 7.47 (q, J=8.6 Hz, 4H), 7.42 (d, J=8.4 Hz, 1H), 7.35 (t, J=7.5 Hz, 1H), 2.59 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 142.76, 141.08, 139.43, 136.46, 133.22, 132.28, 131.98, 129.61, 127.82, 127.23, 122.15, 121.12, 120.73, 114.65, 110.80, 15.83. HRMS (ESI) calcd for C18H14ClN2S [M+H]+ 325.0561, found 325.0555.
9-(4-(tert-Butyl)phenyl)-3-chloro-9H-pyrido[3,4-b]indole (3ao): Yellow solid, 21.7 mg, 65% yield. m.p. 133~136 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.57 (s, 1H), 8.15 (d, J=7.9 Hz, 1H), 8.01 (s, 1H), 7.64 (d, J=8.6 Hz, 2H), 7.56 (ddd, J=8.3, 7.0, 1.3 Hz, 1H), 7.48~7.46 (dd, J=8.6, 2.4 Hz, 3H), 7.34 (ddd, J=8.0, 7.0, 1.1 Hz, 1H), 1.43 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 151.44, 142.75, 140.85, 136.37, 133.67, 132.15, 129.35, 127.14, 126.19, 121.98, 120.82, 120.58, 114.43, 110.90, 34.90, 31.40. HRMS (ESI) calcd for C21H20ClN2 [M+H]+ 335.1310, found 335.1324.
3-Chloro-9-(3-ethylphenyl)-9H-pyrido[3,4-b]indole (3ap): Brown solid, 12.2 mg, 40% yield. m.p. 114~115 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.57 (s, 1H), 8.15 (d, J=7.9 Hz, 1H), 8.01 (s, 1H), 7.59~7.53 (m, 2H), 7.47 (d, J=8.3 Hz, 1H), 7.38~7.33 (m, 4H), 2.78 (q, J=7.5 Hz, 2H), 1.32 (t, J=7.6 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 146.77, 142.63, 140.90, 136.41, 136.30, 132.19, 132.15, 130.09, 129.43, 127.91, 126.02, 123.86, 122.02, 120.89, 120.62, 114.46, 110.84, 28.78, 15.45. HRMS (ESI) calcd for C19H16ClN2 [M+H]+ 307.0997, found 307.1000.
3-Chloro-9-(4-fluorophenyl)-9H-pyrido[3,4-b]indole (3aq): Yellow solid, 19.2 mg, 65% yield. m.p. 183~185 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.48 (d, J=0.6 Hz, 1H), 8.15 (d, J=7.9 Hz, 1H), 8.01 (d, J=0.7 Hz, 1H), 7.58 (ddd, J=8.3, 7.1, 1.1 Hz, 1H), 7.54~7.51 (m, 2H), 7.39~7.32 (m, 4H); 13C NMR (126 MHz, CDCl3) δ: 162.21 (d, J=249.0 Hz), 142.89, 141.31, 136.57, 132.45(d, J=3.8 Hz), 132.36, 131.86, 129.73, 128.85 (d, J=8.7 Hz), 122.24, 121.24, 120.74, 117.47 (d, J=22.9 Hz), 114.69, 110.62. 19F NMR (471 MHz, CDCl3) δ: 112.07. HRMS (ESI) calcd for C17H11ClFN2 [M+H]+ 297.0590, found 297.0585.
3-Chloro-9-(4-chlorophenyl)-9H-pyrido[3,4-b]indole (3ar): Yellow solid, 14.3 mg, 46% yield. m.p. 178~180 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.52 (s, 1H), 8.14 (d, J=7.9 Hz, 1H), 8.00 (s, 1H), 7.60 (dd, J=22.0, 8.2 Hz, 3H), 7.50 (d, J=8.5 Hz, 2H), 7.41 (d, J=8.4 Hz, 1H), 7.37 (t, J=7.5 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 142.53, 141.45, 136.24, 135.06, 134.18, 131.85, 130.66, 129.76, 128.13, 122.26, 121.38, 120.87, 114.70, 110.62. HRMS (ESI) calcd for C17H11Cl2N2 [M+H]+ 313.0294, found 313.0289.
9-(4-Bromophenyl)-3-chloro-9H-pyrido[3,4-b]indole (3as): Brown solid, 22.7 mg, 64% yield. m.p. 180~183 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.55 (s, 1H), 8.16 (d, J=7.8 Hz, 1H), 8.02 (s, 1H), 7.80 (dd, J=6.7, 4.8 Hz, 2H), 7.62~7.59 (m, 1H), 7.47~7.43 (m, 3H), 7.39 (t, J=7.5 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 142.34, 141.38, 136.05, 135.50, 133.55, 132.45, 131.75, 129.68, 128.31, 122.17, 121.96, 121.31, 120.79, 114.61, 110.51. HRMS (ESI) calcd for C17H11BrClN2 [M+H]+ 356.9789, found 356.9785.
3-Chloro-9-(naphthalen-1-yl)-9H-pyrido[3,4-b]indole (3at): Brown solid, 25.5 mg, 78% yield. m.p. 156~158 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.22~8.19 (m, 2H), 8.11~8.09 (m, 2H), 8.04 (d, J=8.3 Hz, 1H), 7.71~7.67 (m, 1H), 7.64 (dd, J=7.2, 1.1 Hz, 1H), 7.59~7.55 (m, 1H), 7.49 (ddd, J=8.3, 7.0, 1.0 Hz, 1H), 7.36 (t, J=7.6 Hz, 2H), 7.21 (d, J=8.5 Hz, 1H), 7.06 (d, J=8.3 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 143.93, 141.07, 137.49, 134.95, 132.62, 132.45, 132.18, 130.49, 129.91, 129.58, 128.80, 127.55, 127.14, 126.60, 125.97, 122.80, 122.13, 120.98, 120.59, 114.68, 111.27. HRMS (ESI) calcd for C21H14ClN2 [M+H]+ 329.0841, found 329.0835.
9-(Benzo[
d][
1,
3]dioxol-5-yl)-3-chloro-9
H-pyrido[3,4-
b]indole (
3au): Brown solid, 24.1 mg, 75% yield. m.p. 112~114 ℃;
1H NMR (500 MHz, CDCl
3)
δ: 8.51 (s, 1H), 8.13 (d,
J=7.9 Hz, 1H), 8.00 (s, 1H), 7.57 (ddd,
J=8.4, 7.2, 1.1 Hz, 1H), 7.40 (d,
J=8.4 Hz, 1H), 7.36~7.33 (m, 1H), 7.03~7.97 (m, 3H), 6.13 (s, 2H);
13C NMR (126 MHz, CDCl
3)
δ: 148.97, 147.73, 143.00, 140.89, 136.63, 132.06, 131.98, 129.91, 129.50, 122.03, 120.90, 120.60, 120.45, 114.48, 110.74, 109.17, 107.99, 102.12. HRMS (ESI) calcd for C
18H
12ClN
2O
2 [M+H]
+ 323.0582, found 323.0591.
3-Chloro-9-(quinolin-6-yl)-9H-pyrido[3,4-b]indole (3av): Brown solid, 18.7 mg, 57% yield. m.p. 107~109 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.06 (d, J=3.4 Hz, 1H), 8.62 (s, 1H), 8.40 (d, J=8.9 Hz, 1H), 8.27 (d, J=8.2 Hz, 1H), 8.18 (d, J=7.9 Hz, 1H), 8.04 (d, J=2.9 Hz, 2H), 7.91 (d, J=8.9 Hz, 1H), 7.61 (t, J=7.7 Hz, 1H), 7.56 (dd, J=8.2, 4.2 Hz, 1H), 7.52 (d, J=8.2 Hz, 1H), 7.40 (t, J=7.5 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 151.47, 147.38, 142.63, 141.53, 136.34, 136.15, 134.59, 132.63, 132.12, 131.92, 129.81, 129.03, 128.11, 125.01, 122.38, 122.30, 121.49, 120.95, 114.74, 110.70. HRMS (ESI) calcd for C20H13ClN3 [M+H]+ 330.0798, found 330.0787.
3-Chloro-9-(quinolin-5-yl)-9H-pyrido[3,4-b]indole (3aw): White solid, 23.6 mg, 72% yield. m.p. 226~227 ℃; 1H NMR (500 MHz, CDCl3) δ: 9.01 (dd, J=4.0, 1.2 Hz, 1H), 8.37 (d, J=8.6 Hz, 1H), 8.21 (d, J=7.9 Hz, 1H), 8.17 (s, 1H), 8.07 (s, 1H), 7.96~7.93 (m, 1H), 7.73 (d, J=7.3 Hz, 1H), 7.56 (d, J=8.5 Hz, 1H), 7.51 (t, J=7.7 Hz, 1H), 7.37 (t, J=7.5 Hz, 1H), 7.31 (dd, J=8.5, 4.1 Hz, 1H), 7.03 (d, J=8.3 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ: 151.59, 149.38, 143.83, 141.47, 137.44, 132.58, 132.35, 132.15, 131.48, 131.31, 129.84, 129.54, 127.16, 125.90, 122.35, 122.29, 121.34, 120.70, 114.81, 111.00. HRMS (ESI) calcd for C20H13ClN3 [M+H]+ 330.0793, found 330.0786.
9-Benzyl-9H-pyrido[3,4-b]indole 2-oxide (6ab): Yellow solid, 22.7 mg, 83% yield. m.p. 75~77 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.47 (s, 1H), 8.16 (d, J=6.6 Hz, 1H), 8.07 (d, J=7.9 Hz, 1H), 7.89 (d, J=6.6 Hz, 1H), 7.56 (t, J=7.4 Hz, 1H), 7.47 (d, J=8.3 Hz, 1H), 7.35 (t, J=7.5 Hz, 1H), 7.29 (d, J=7.1 Hz, 3H), 7.15 (dd, J=8.3, 6.0 Hz, 2H), 5.44 (s, 2H); 13C NMR (126 MHz, CDCl3) δ: 142.65, 137.73, 135.26, 131.61, 129.16, 128.32, 128.27, 126.49, 122.85, 121.56, 121.29, 121.19, 121.07, 116.28, 109.79, 47.28. HRMS (ESI) calcd for C18H15N2O [M+H]+ 275.1179, found 275.1181.
9-Phenethyl-9H-pyrido[3,4-b]indole 2-oxide (6ac): Yel- low solid, 23.9 mg, 83% yield. m.p. 72~76 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.36 (s, 1H), 8.11 (dd, J=6.6, 1.1 Hz, 1H), 8.02 (d, J=7.8 Hz, 1H), 7.83 (d, J=6.7 Hz, 1H), 7.53 (t, J=7.7 Hz, 1H), 7.34~7.28 (m, 2H), 7.25~7.21 (m, 3H), 7.09~7.06 (m, 2H), 4.44 (t, J=7.3 Hz, 2H), 3.12 (t, J=7.3 Hz, 2H); 13C NMR (126 MHz, CDCl3) δ: 142.06, 137.55, 137.47, 131.21, 128.86, 128.67, 128.05, 127.17, 122.37, 121.22, 121.19, 120.96, 120.92, 116.16, 109.56, 45.63, 35.08. HRMS (ESI) calcd for C19H17N2O [M+H]+ 289.1336, found 289.1338.
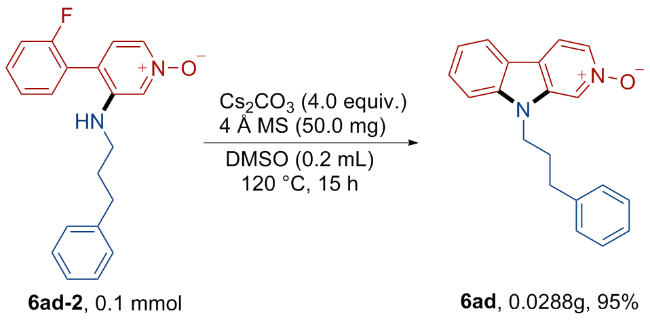
9-(3-Phenylpropyl)-9H-pyrido[3,4-b]indole 2-oxide (6ad): Yellow solid, 21.7 mg, 72% yield. m.p. 159~182 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.48 (s, 1H), 8.14 (d, J=6.5 Hz, 1H), 8.01 (d, J=7.8 Hz, 1H), 7.83 (d, J=6.5 Hz, 1H), 7.54 (t, J=7.6 Hz, 1H), 7.33~7.28 (m, 4H), 7.22 (t, J=7.3 Hz, 1H), 7.14 (d, J=7.3 Hz, 2H), 4.22 (t, J=7.4 Hz, 2H), 2.70 (t, J=7.6 Hz, 2H), 2.12~2.16 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 142.20, 140.14, 137.45, 131.20, 128.65, 128.21, 128.07, 126.43, 122.38, 121.32, 121.23, 120.97, 120.87, 116.23, 109.60, 43.14, 33.11, 29.89. HRMS (ESI) calcd for C20H19N2O [M+H]+ 303.1492, found 303.1495.
4-(2-Fluorophenyl)-3-((3-phenylpropyl)amino)pyridine 1-oxide (6ad-2): White solid, 50 mg, 77%. m.p. 110~114 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.72~7.71 (m, 1H), 7.68 (dd, J=6.3, 1.6 Hz, 1H), 7.45~7.40 (m, 1H), 7.30~7.14 (m, 6H), 7.10 (d, J=7.2 Hz, 2H), 6.93 (d, J=6.3 Hz, 1H), 3.78 (t, J=5.2 Hz, 1H), 3.06 (q, J=6.8 Hz, 2H), 2.63 (t, J=7.5 Hz, 2H), 1.88 (p, J=7.3 Hz, 2H); 13C NMR (126 MHz, CDCl3) δ: 159.59 (d, J=248.5 Hz), 143.95, 140.81, 131.18 (d, J=2.7 Hz), 131.07 (d, J=8.3 Hz), 128.60, 128.33, 127.96, 126.78, 126.25, 125.18 (d, J=3.4 Hz), 123.09, 122.48 (d, J=16.0 Hz), 120.16, 116.71 (d, J=21.7 Hz), 42.84, 33.06, 30.29; 19F NMR (471 MHz, CDCl3) δ: 113.14. HRMS (ESI) calcd for C20H20FN2O [M+H]+ 323.1555, found 323.1567.
9-(2-(Benzo[
d][
1,
3]dioxol-5-yl)ethyl)-9
H-pyrido[3,4-
b]indole 2-oxide (
6ae): Yellow solid, 15.6 mg, 47% yield. m.p. 205~208 ℃;
1H NMR (500 MHz, CDCl
3)
δ: 8.39 (s, 1H), 8.11 (d,
J=6.6 Hz, 1H), 7.99 (t,
J=13.9 Hz, 1H), 7.81 (t,
J=17.2 Hz, 1H), 7.53 (t,
J=7.7 Hz, 1H), 7.31 (dd,
J=17.9, 8.4 Hz, 1H), 6.64 (d,
J=7.8 Hz, 1H), 6.54 (s, 1H), 6.47 (d,
J=7.8 Hz, 1H), 5.89 (s, 1H), 4.38 (t,
J=7.2 Hz, 1H), 3.02 (t,
J=7.2 Hz, 1H);
13C NMR (126 MHz, CDCl
3)
δ: 147.97, 146.66, 142.05, 137.49, 131.26, 131.19, 128.03, 122.35, 121.70, 121.24, 121.13, 120.98, 120.92, 116.20, 109.54, 108.99, 108.59, 101.02, 45.78, 34.76. HRMS (ESI) calcd for C
20H
17N
2O
3 [M+H]
+ 333.1234, found 333.1236.
9-(2-(Thiophen-2-yl)ethyl)-9H-pyrido[3,4-b]indole 2-oxide (6af): Yellow oil, 13.2 mg, 45% yield. 1H NMR (500 MHz, CDCl3) δ: 8.42 (s, 1H), 8.11 (d, J=6.6 Hz, 1H), 8.02 (d, J=7.9 Hz, 1H), 7.84 (d, J=6.6 Hz, 1H), 7.54 (t, J=7.7 Hz, 1H), 7.36~7.30 (m, 2H), 7.12 (d, J=5.1 Hz, 1H), 6.85~6.83 (m, 1H), 6.66 (d, J=3.2 Hz, 1H), 4.48 (t, J=7.0 Hz, 2H), 3.34 (t, J=7.0 Hz, 2H); 13C NMR (126 MHz, CDCl3) δ: 142.11, 139.21, 137.58, 131.32, 128.22, 127.36, 126.24, 124.78, 122.43, 121.41, 121.32, 121.09, 121.03, 116.26, 109.56, 45.78, 41.02. HRMS (ESI) calcd for C17H15N2OS [M+H]+ 295.0900, found 295.0902.
9-(4,4-Diethoxybutyl)-9H-pyrido[3,4-b]indole 2-oxide (6ag): Yellow oil, 18.0 mg, 55% yield. 1H NMR (500 MHz, CDCl3) δ: 8.55 (s, 1H), 8.10 (d, J=6.5 Hz, 1H), 7.97 (d, J=7.8 Hz, 1H), 7.81 (d, J=6.6 Hz, 1H), 7.52 (t, J=7.5 Hz, 1H), 7.42 (d, J=8.3 Hz, 1H), 7.27 (t, J=7.6 Hz, 1H), 4.44 (t, J=5.4 Hz, 1H), 4.24 (t, J=7.3 Hz, 2H), 3.58 (dq, J=9.2, 7.0 Hz, 2H), 3.42 (dq, J=9.3, 7.0 Hz, 2H), 1.94~1.88 (m, 2H), 1.66~1.62 (m, 2H), 1.15 (t, J=7.1 Hz, 6H); 13C NMR (126 MHz, CDCl3) δ: 142.28, 137.48, 131.14, 128.08, 122.45, 121.35, 121.20, 120.92, 120.82, 116.21, 109.69, 102.41, 61.63, 43.55, 30.96, 23.96, 15.30. HRMS (ESI) calcd for C19H25N2O3 [M+H]+ 329.1860, found 329.1864.
7-Methyl-9-(3-phenylpropyl)-9H-pyrido[3,4-b]indole 2-oxide (6bd): Yellow solid, 23.0 mg, 73% yield. m.p. 175~179 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.43 (s, 1H), 8.09 (d, J=6.5 Hz, 1H), 7.84 (d, J=8.0 Hz, 1H), 7.73 (d, J=6.6 Hz, 1H), 7.28 (t, J=7.5 Hz, 2H), 7.20 (t, J=6.3 Hz, 1H), 7.13 (d, J=7.4 Hz, 2H), 7.10 (d, J=8.0 Hz, 1H), 7.03 (s, 1H), 4.14 (t, J=7.3 Hz, 2H), 2.67 (t, J=7.5 Hz, 2H), 2.52 (s, 3H), 2.18~2.12 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 142.67, 140.21, 138.77, 137.42, 130.96, 128.61, 128.27, 126.39, 122.49, 122.21, 121.52, 120.86, 118.64, 115.78, 109.67, 42.86, 33.01, 29.80, 22.36. HRMS (ESI) calcd for C21H21N2O [M+H]+ 317.1649, found 317.1652.
6-Methyl-9-(3-phenylpropyl)-9H-pyrido[3,4-b]indole 2-oxide (6cd): Yellow solid, 19.2 mg, 61% yield. m.p. 175~179 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.46 (s, 1H), 8.11 (d, J=6.6 Hz, 1H), 7.80 (s, 2H), 7.36 (d, J=8.4 Hz, 1H), 7.30 (t, J=7.5 Hz, 2H), 7.22 (t, J=6.4 Hz, 2H), 7.14 (d, J=7.5 Hz, 2H), 4.20 (t, J=7.3 Hz, 2H), 2.69 (t, J=7.6 Hz, 2H), 2.53 (s, 3H), 2.21~2.15 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 140.56, 140.21, 137.56, 130.91, 130.46, 129.54, 128.63, 128.22, 126.40, 122.30, 121.15, 121.09, 120.99, 116.10, 109.31, 43.16, 33.12, 29.94, 21.31. HRMS (ESI) calcd for C21H21N2O [M+H]+ 317.1649, found 317.1652.
6-Cyano-9-(3-phenylpropyl)-9H-pyrido[3,4-b]indole 2-oxide (6dd): White solid, 23.2 mg, 71% yield. m.p. 228~230 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.56 (s, 1H), 8.33 (s, 1H), 8.20 (d, J=6.7 Hz, 1H), 7.89 (d, J=6.7 Hz, 1H), 7.75 (dd, J=8.6, 1.3 Hz, 1H), 7.36 (d, J=8.6 Hz, 1H), 7.29 (t, J=7.4 Hz, 2H), 7.22 (d, J=7.3 Hz, 1H), 7.13 (d, J=7.2 Hz, 2H), 4.28 (t, J=7.4 Hz, 2H), 2.73 (dd, J=16.1, 8.7 Hz, 2H), 2.26~2.20 (m, 2H); 13C NMR (126 MHz, CDCl3) δ: 143.69, 139.67, 138.39, 132.78, 130.73, 128.75, 128.17, 126.63, 126.09, 123.10, 121.32, 119.70, 119.44, 116.82, 110.49, 104.08, 43.42, 32.94, 29.72. HRMS (ESI) calcd for C21H18N3O [M+H]+ 328.1445, found 328.1447.