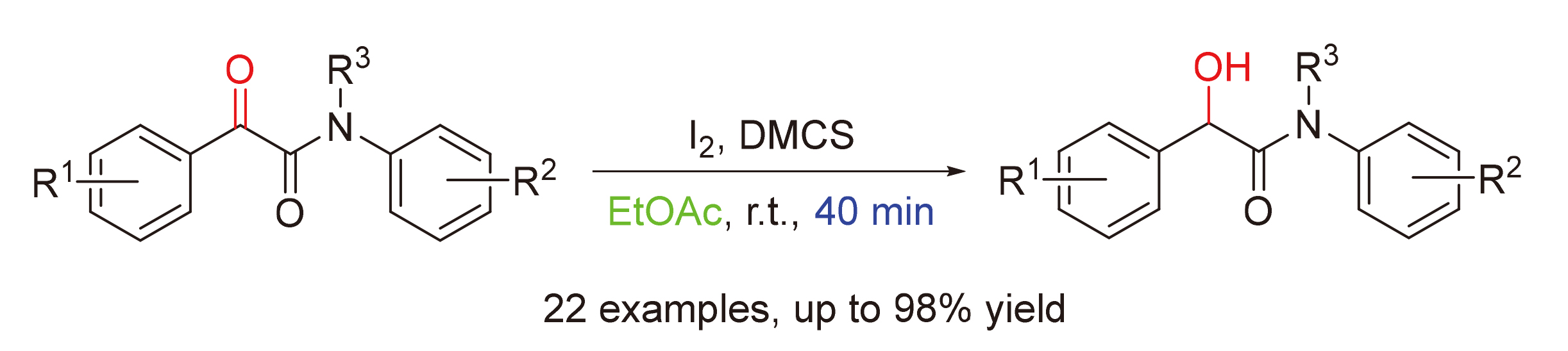
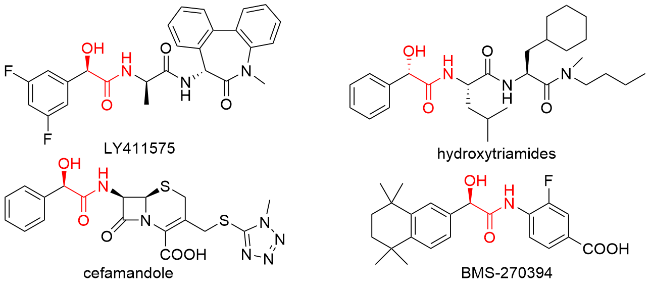
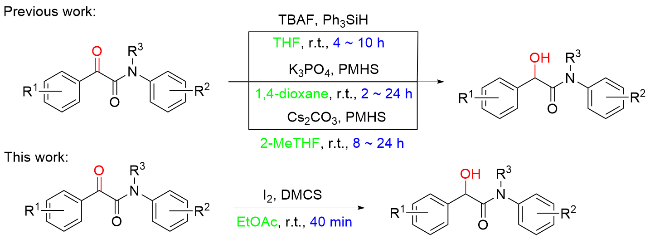
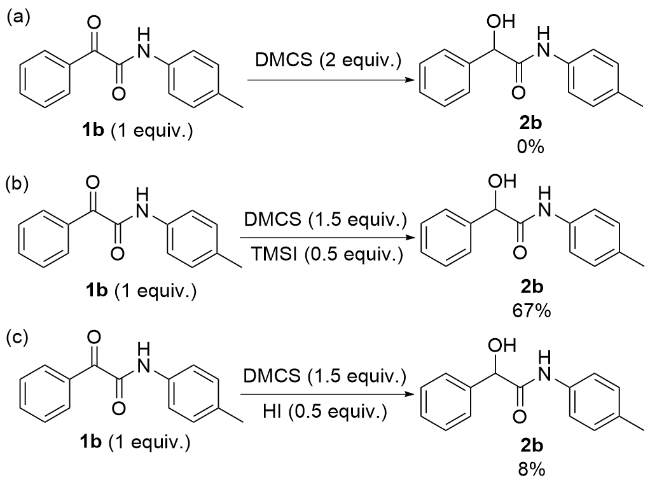
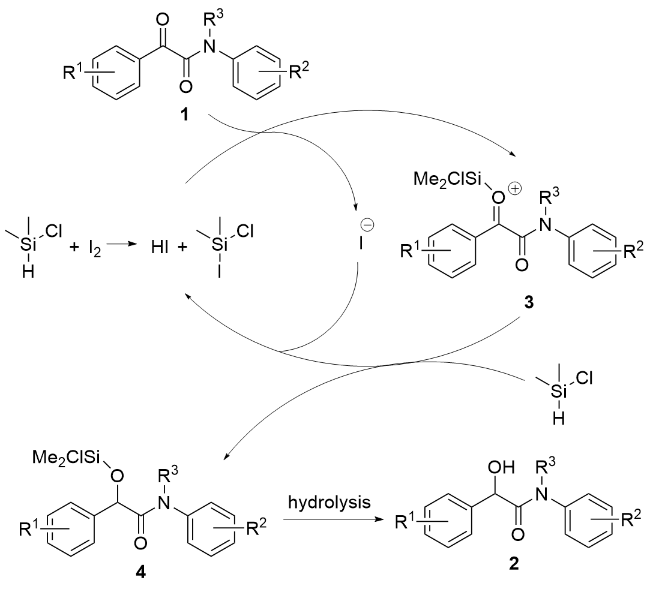
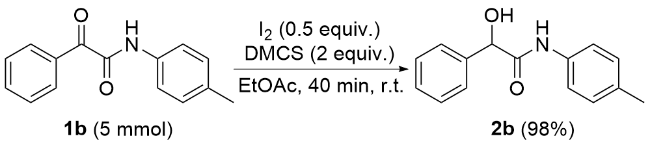1 结果与讨论
表1 反应条件优化aTable 1 Optimization of reaction conditions 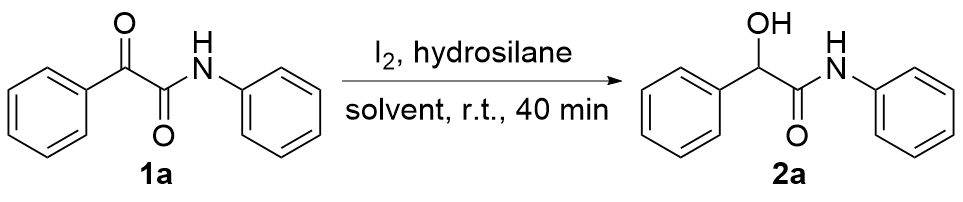 |
| Entry | Hydrosilane | Solvent | Yield b/% |
|---|---|---|---|
| 1 | Et3SiH | DCM | 71 |
| 2 | Et3SiH | EtOAc | 72 |
| 3 | Et3SiH | MeCN | Trace |
| 4 | Et3SiH | CHCl3 | 22 |
| 5 | Et3SiH | THF | 71 |
| 6 | Et3SiH | Toluene | 10 |
| 7 | Et3SiH | 1,4-Dioxane | 16 |
| 8 | Et3SiH | CH3NO2 | 58 |
| 9 | Et3SiH | C6H5NO2 | 20 |
| 10 | Et3SiH | DMF | 0 |
| 11 | Et3SiH | EtOH | 0 |
| 12 | Et3SiH | H2O | 0 |
| 13 | PMHS | EtOAc | 78 |
| 14 | TMDS | EtOAc | 81 |
| 15 | Ph3SiH | EtOAc | 17 |
| 16 | Ph2SiH2 | EtOAc | 69 |
| 17 | (EtO)3SiH | EtOAc | Trace |
| 18 | DMCS | EtOAc | 98 |
| 19 c | DMCS | EtOAc | 85 |
a Reaction conditions: α-keto amide 1a (0.5 mmol, 1.0 equiv.), I2 (0.5 equiv.), hydrosilane (2.0 equiv.), solvent (2 mL), stirred at room temperature for 40 min. b Isolated yields. c 0.2 equiv. of I2. |
表2 反应底物拓展aTable 2 Reaction substrate expansion 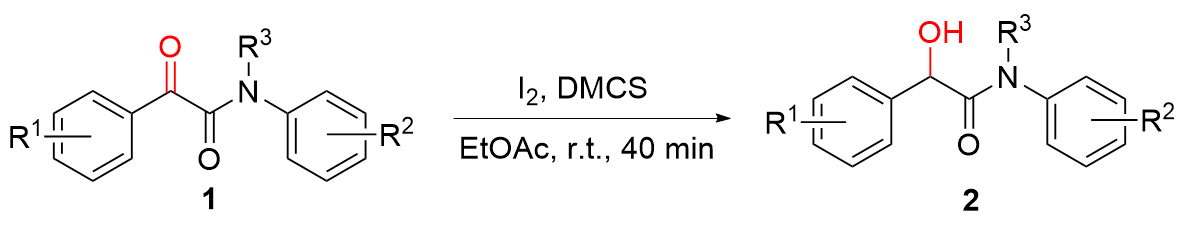 |
| Entry | Product | Yield b/% | Entry | Product | Yield b/% |
|---|---|---|---|---|---|
| 1 | 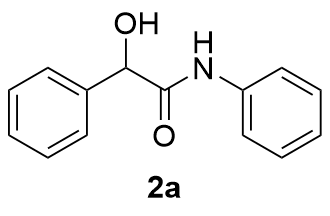 | 98 | 12 | 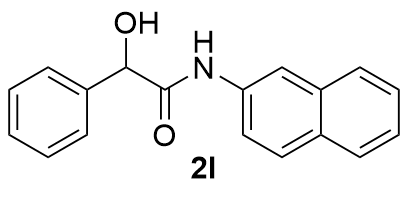 | 53 |
| 2 | 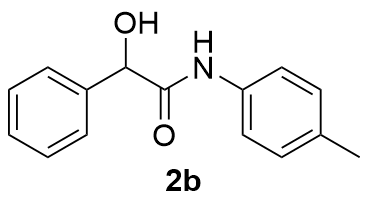 | 98 | 13 | 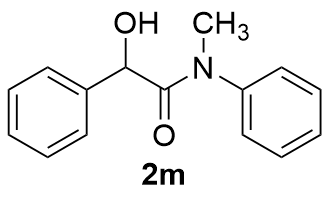 | 65 |
| 3 | 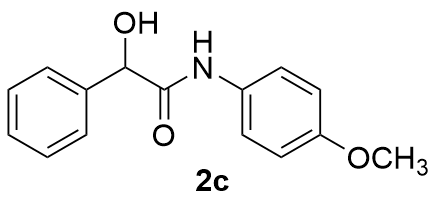 | 60 | 14 | 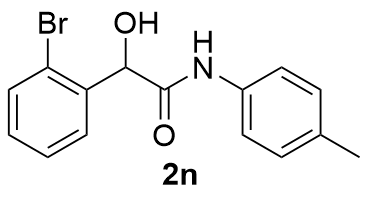 | 74 |
| 4 | 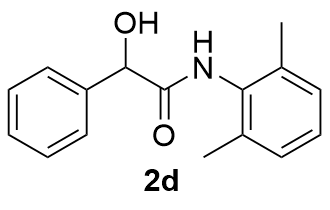 | 73 | 15 | 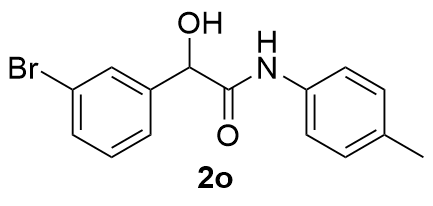 | 75 |
| 5 | 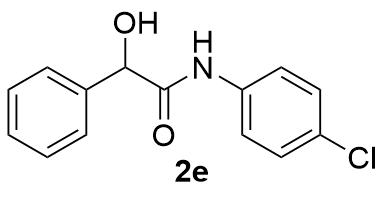 | 97 | 16 | 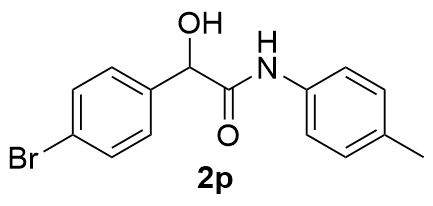 | 71 |
| 6 | 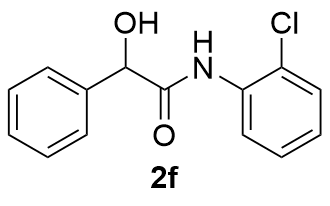 | 58 | 17 | 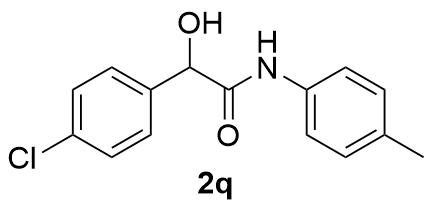 | 71 |
| 7 | 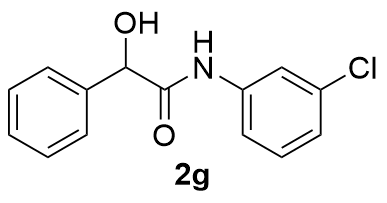 | 82 | 18 | 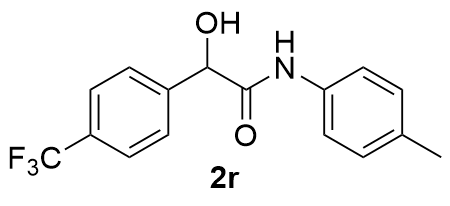 | 67 |
| 8 | 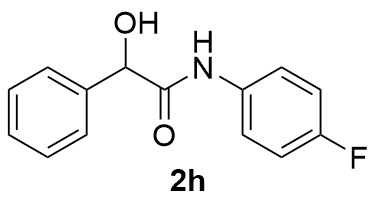 | 67 | 19 | 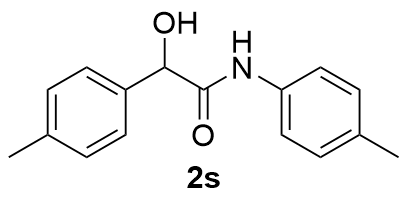 | 85 |
| 9 | 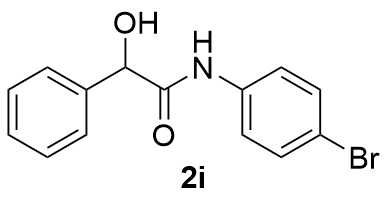 | 89 | 20 | 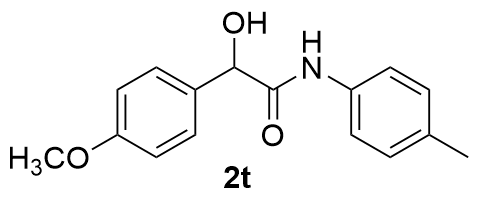 | 66 |
| 10 | 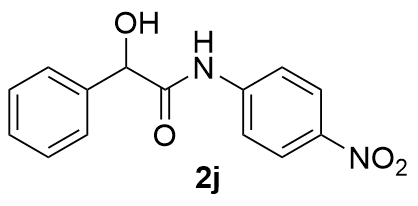 | 72 | 21 | 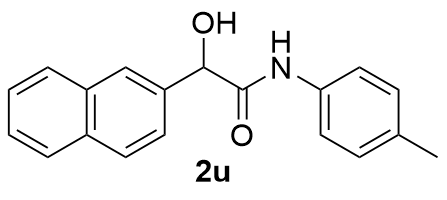 | 52 |
| 11 | 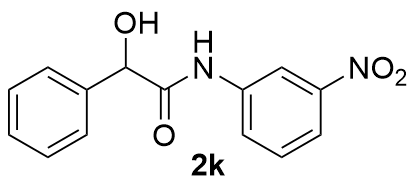 | 85 | 22 | 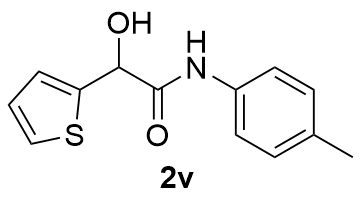 | 53 |
a Reaction conditions: α-keto amides 1 (0.5 mmol), I2 (0.25 mmol), DMCS (1.0 mmol), EtOAc (2 mL), stirred at room temperature for 40 min. b Isolated yields. |