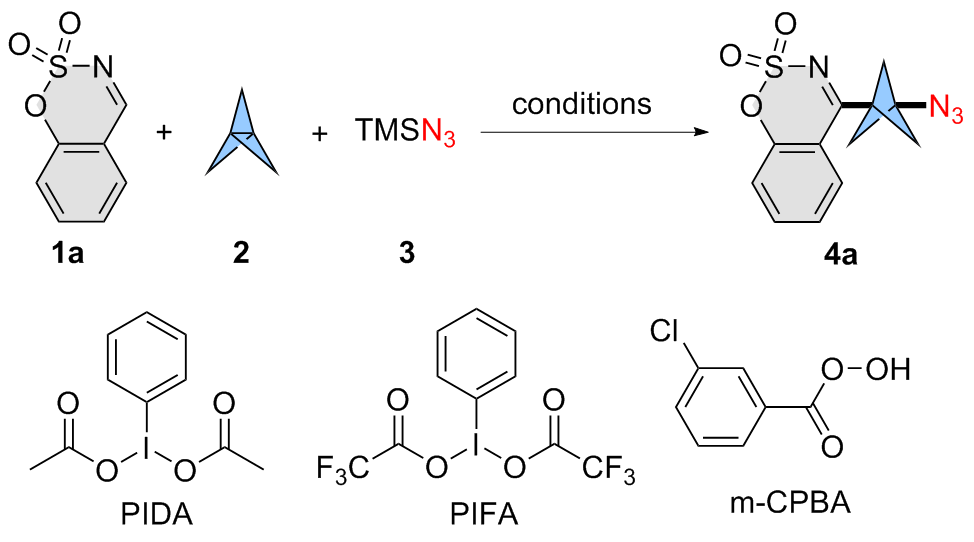
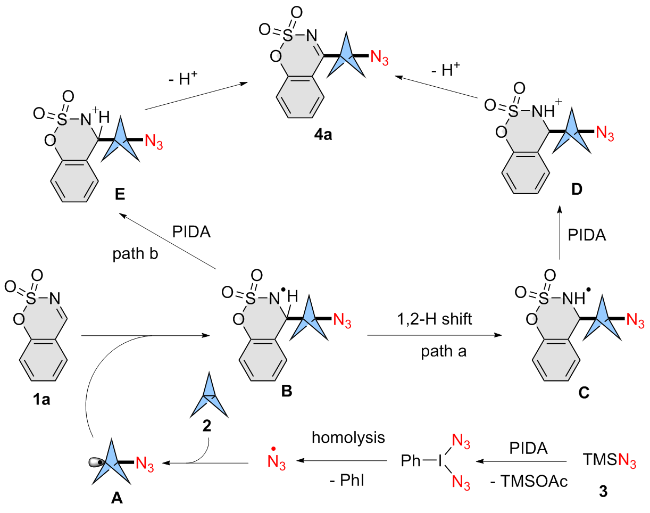
An oven-dried 25 mL Schlenk tube fitted with a magnetic stirring bar was charged with cyclic aldimines 1 (0.2 mmol, 1.0 equiv.), [1.1.1]propellane (2, 0.3 mmol, 1.5 equiv.) and PIDA (0.3 mmol, 1.5 equiv.) under N2 atmosphere. Then, the mixture was cooled down to 10 ℃ and TMSN3 (3) (0.3 mmol, 1.5 equiv.) in DMF (1 mL) was added dropwise under stirring. After stirring at 10 ℃ for 6 h, the mixture was poured into NaHCO3 aqueous solution, and extracted with EtOAc (5 mL×3). The collected organic layer was washed with brine, and dried with MgSO4. The solvent was removed in vacuo, and the obtained residue was further purified by silica gel column chromatography [200~300 mesh silica gel, V(petroleum ether, PE)∶V(ethyl acetate, EA)∶V(DCM)=5∶1∶1] to afford product 4.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)benzo[e][1,2,3]-oxathiazine 2,2-dioxide (4a): Yellow solid (42 mg, 72% yield). m.p. 105~106 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.92 (dd, J=8.0, 1.6 Hz, 1H), 7.73 (ddd, J=8.8, 7.5, 1.6 Hz, 1H), 7.39 (td, J=7.7, 1.2 Hz, 1H), 7.33 (dd, J=8.4, 1.1 Hz, 1H), 2.60 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 174.8, 153.7, 137.1, 128.4, 125.8, 119.5, 116.1, 56.2, 52.1, 39.1; HRMS (ESI-TOF) calcd for C12H10N4O3SNa [M+Na]+ 313.0366, found 313.0367.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-6-methylbenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4b): Yellow solid (42 mg, 70% yield). m.p. 146~147 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.63~7.57 (m, 1H), 7.45 (dd, J=8.5, 2.1 Hz, 1H), 7.14 (d, J=8.5 Hz, 1H), 2.52 (s, 6H), 2.37 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 174.8, 151.7, 137.9, 135.9, 128.2, 119.2, 115.9, 56.2, 52.1, 39.1, 21.1; HRMS (ESI- TOF) calcd for C13H13N4O3S [M+H]+ 305.0703, found 305.0700.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-6-(tert-butyl)-benzo[e][1,2,3]oxathiazine 2,2-dioxide (4c): Yellow solid (46 mg, 67% yield). m.p. 132~133 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.89 (d, J=2.4 Hz, 1H), 7.75 (dd, J=8.7, 2.4 Hz, 1H), 7.26~7.24 (m, 1H), 2.60 (s, 6H), 1.37 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 175.0, 151.5, 149.1, 134.8, 124.8, 118.9, 115.5, 56.3, 53.4, 52.1, 51.7, 39.1, 34.8, 31.2; HRMS (ESI-TOF) calcd for C16H18N4O3SNa [M+Na]+ 369.0992, found 369.0997.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-6-methoxybenzo-[e][1,2,3]oxathiazine 2,2-dioxide (4d): Yellow solid (47 mg, 73% yield). m.p. 137~138 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.33 (dd, J=2.1, 1.2 Hz, 1H), 7.27~7.24 (m, 2H), 3.87 (s, 3H), 2.59 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 174.5, 156.6, 147.3, 122.7, 120.3, 116.4, 112.5, 56.2, 56.1, 52.0, 39.1; HRMS (ESI-TOF) calcd for C13H12- N4O4SNa [M+Na]+ 343.0471, found 343.0471.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-6-(trifluorometh-oxy)benzo[e][1,2,3]oxathiazine 2,2-dioxide (4e): Yellow solid (52 mg, 70% yield). m.p. 135~136 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.79~7.72 (m, 1H), 7.61~7.57 (m, 1H), 7.39 (d, J=9.1 Hz, 1H), 2.60 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 173.8, 151.7, 145.5, 129.7, 121.3, 120.6, 120.3 (q, J=259.6 Hz), 116.5, 56.3, 52.0, 39.0; 19F NMR (471 MHz, CDCl3) δ: 58.31; HRMS (ESI-TOF) calcd for C13H9F3N4O4SNa [M+Na]+ 397.0189, found 397.0196.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-6-fluorobenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4f): Yellow solid (35 mg, 57% yield). m.p. 106~107 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.58 (dd, J=7.9, 3.0 Hz, 1H), 7.45 (ddd, J=9.1, 7.4, 3.0 Hz, 1H), 7.33 (dd, J=9.1, 4.4 Hz, 1H), 2.60 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 172.9 (d, J=2.5 Hz), 158.7 (d, J=254.1 Hz), 148.7 (d, J=2.5 Hz), 123.3 (d, J=23.4 Hz), 120.3 (d, J=8.8 Hz), 115.5 (d, J=8.8 Hz), 113.5 (d, J=23.4 Hz), 55.2, 50.1, 38.0; 19F NMR (471 MHz, CDCl3) δ: 112.59; HRMS (ESI-TOF) calcd for C12H9FN4O3- SNa [M+Na]+ 331.0272, found 331.0266.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-6-chlorobenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4g): Yellow solid (39 mg, 60% yield). m.p. 117~118 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.85 (d, J=2.4 Hz, 1H), 7.67 (dd, J=8.8, 2.5 Hz, 1H), 7.29 (d, J=8.9 Hz, 1H), 2.61 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 173.8, 152.1, 136.9, 131.3, 127.9, 121.0, 116.9, 56.3, 52.1, 39.0; HRMS (ESI-TOF) calcd for C12H9- BrN4O3SNa [M+Na]+ 346.9976, found 346.9970.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-6-bromobenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4h): White solid (46 mg, 62% yield). m.p. 118~119 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.00 (d, J=2.3 Hz, 1H), 7.81 (dd, J=8.8, 2.3 Hz, 1H), 7.23 (d, J=8.8 Hz, 1H), 2.61 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 173.7, 152.6, 139.8, 130.9, 121.2, 118.4, 117.3, 56.3, 52.1, 39.0; HRMS (ESI-TOF) calcd for C12H9- BrN4O3Na [M+Na]+ 390.9471, found 390.9475.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-7-methylbenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4i): Yellow solid (44 mg, 73% yield). m.p. 165~166 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.79 (d, J=8.1 Hz, 1H), 7.20~7.16 (m, 1H), 7.12 (s, 1H), 2.58 (s, 6H), 2.49 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 174.6, 153.8, 149.7, 128.2, 126.8, 119.6, 113.7, 56.2, 52.0, 39.0, 22.2; HRMS (ESI-TOF) calcd for C13H12- N4O3Na [M+Na]+ 327.0522, found 327.0527.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-7-methoxybenzo-[e][1,2,3]oxathiazine 2,2-dioxide (4j): Yellow solid (42 mg, 65% yield). m.p. 133~134 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.82 (d, J=8.9 Hz, 1H), 6.87 (dd, J=9.0, 2.5 Hz, 1H), 6.75 (d, J=2.5 Hz, 1H), 3.93 (s, 3H), 2.56 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 172.9, 165.5, 155.3, 129.0, 112.4, 108.5, 102.3, 55.4, 55.1, 51.0, 38.0; HRMS (ESI- TOF) calcd for C13H12N4O4SNa [M+Na]+ 343.0471, found 343.0480.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-7-(benzyloxy)ben- zo[e][1,2,3]oxathiazine 2,2-dioxide (4k): Yellow solid (52 mg, 66% yield). m.p. 155~156 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.81 (d, J=9.0 Hz, 1H), 7.41 (d, J=1.4 Hz, 6H), 6.92 (dd, J=9.0, 2.5 Hz, 1H), 6.82 (d, J=2.5 Hz, 1H), 5.17 (s, 2H), 2.56 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 173.9, 165.6, 156.2, 134.7, 123.0, 129.0, 128.9, 127.6, 113.9, 109.7, 104.3, 71.2, 56.1, 52.0, 39.0; HRMS (ESI-TOF) calcd for C14H12N4O5SNa [M+Na]+ 419.1471, found 419.1475.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-7-fluorobenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4l): Yellow solid (36 mg, 59% yield). m.p. 148~149 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.96 (dd, J=8.9, 5.7 Hz, 1H), 7.11 (ddd, J=8.9, 7.7, 2.5 Hz, 1H), 7.05 (dd, J=8.3, 2.5 Hz, 1H), 2.59 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 173.9, 166.9 (d, J=263.3 Hz), 155.7 (d, J=13.8 Hz), 130.8 (d, J=11.3 Hz), 113.9 (d, J=25.2 Hz), 112.9 (d, J=3.8 Hz), 107.5 (d, J=25.2 Hz), 56.2, 52.1, 39.1; 19F NMR (471 MHz, CDCl3) δ: 94.16; HRMS (ESI-TOF) calcd for C12H9FN4O3SNa [M+Na]+ 331.0272, found 331.0267.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-7-chlorobenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4m): Yellow solid (42 mg, 65% yield). m.p. 137~138 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.85 (d, J=8.5 Hz, 1H), 7.38~7.32 (m, 2H), 2.59 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 174.1, 154.2, 143.3, 129.3, 126.4, 119.9, 114.5, 56.2, 52.1, 39.1; HRMS (ESI-TOF) calcd for C12H9ClN4O3SNa [M+Na]+ 346.9976, found 346.9976.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-8-methylbenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4n): Yellow solid (46 mg, 75% yield). m.p. 125~126 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.75 (dd, J=8.0, 1.6 Hz, 1H), 7.60~7.53 (m, 1H), 7.30~7.26 (m, 1H), 2.59 (s, 6H), 2.40 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 175.2, 152.1, 138.5, 129.3, 125.9, 125.1, 115.9, 56.2, 52.0, 39.2, 15.1; HRMS (ESI- TOF) calcd for C13H12N4O3SNa [M+Na]+ 327.0522, found 327.0527.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-8-methoxybenzo-[e][1,2,3]oxathiazine 2,2-dioxide (4o): Yellow solid (49 mg, 77% yield). m.p. 133~134 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.46 (dd, J=7.6, 1.8 Hz, 1H), 7.32~7.26 (m, 2H), 3.95 (s, 3H), 2.58 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 175.1, 149.2, 143.4, 125.5, 119.1, 119.0, 119.0, 116.8, 56.6, 56.2, 52.0, 39.3; HRMS (ESI-TOF) calcd for C13H12N4O4SNa [M+Na]+ 343.0471, found 343.0467.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-8-ethoxybenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4p): Yellow solid (48 mg, 72% yield). m.p. 169~170 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.48~7.41 (m, 1H), 7.27 (d, J=6.9 Hz, 2H), 4.15 (q, J=7.0 Hz, 2H), 2.58 (s, 6H), 1.48 (t, J=7.0 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 175.2, 148.6, 143.5, 125.4, 120.2, 119.0, 116.9, 65.6, 56.2, 52.0, 39.3, 14.6; HRMS (ESI-TOF) calcd for C14H14N4O4SNa [M+Na]+ 357.0628, found 357.0629.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-8-bromobenzo[e]-[1,2,3]oxathiazine 2,2-dioxide (4q): Yellow solid (49 mg, 67% yield). m.p. 105~106 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.93 (dd, J=8.0, 1.4 Hz, 1H), 7.88 (dd, J=7.9, 1.5 Hz, 1H), 2.60 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 174.7, 150.8, 140.5, 127.3, 126.3, 117.5, 113.5, 56.3, 52.1, 39.1; HRMS (ESI-TOF) calcd for C12H9BrN4O3SNa [M+Na]+ 390.9471, found 390.9481.
Methyl 4-(3-azidobicyclo[1.1.1]pentan-1-yl)benzo[e]-[1,2,3]oxathiazine-8-carboxylate 2,2-dioxide (4r): Yellow solid (39 mg, 55% yield). m.p. 125~126 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.62 (s, 1H), 8.36 (dd, J=8.6, 2.0 Hz, 1H), 7.38 (d, J=8.7 Hz, 1H), 4.00 (s, 3H), 2.64 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 174.4, 164.5, 156.5, 137.7, 130.2, 127.9, 119.8, 115.6, 56.3, 53.1, 52.1, 39.1; HRMS (ESI-TOF) calcd for C14H12N4O5SNa [M+Na]+ 371.0421, found 371.0431.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-6,8-di-tert-butyl-benzo[e][1,2,3]oxathiazine 2,2-dioxide (4s): Yellow solid (39 mg, 49% yield). m.p. 154~155 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.75 (d, J=2.4 Hz, 1H), 7.73 (d, J=2.3 Hz, 1H), 2.58 (s, 6H), 1.46 (s, 9H), 1.36 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 175.9, 150.5, 148.1, 140.4, 132.2, 122.8, 116.4, 56.3, 52.0, 39.4, 35.3, 35.0, 31.3, 29.8; HRMS (ESI-TOF) calcd for C20H27N4O3S [M+H]+ 403.1798, found 403.1782.
4-(3-Azidobicyclo[1.1.1]pentan-1-yl)-5,7-dimethoxy-benzo[e][1,2,3]oxathiazine 2,2-dioxide (4t): Yellow solid (45 mg, 64% yield). m.p. 171~172 ℃; 1H NMR (500 MHz, CDCl3) δ: 6.40 (d, J=2.4 Hz, 1H), 6.32 (d, J=2.4 Hz, 1H), 3.95 (s, 3H), 3.91 (s, 3H), 2.42 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 175.4, 167.1, 160.1, 157.0, 103.1, 96.4, 95.8, 56.4, 55.8, 55.0, 51.7, 41.3; HRMS (ESI-TOF) calcd for C14H14N4O5SNa [M+Na]+ 373.0577, found 373.0572.
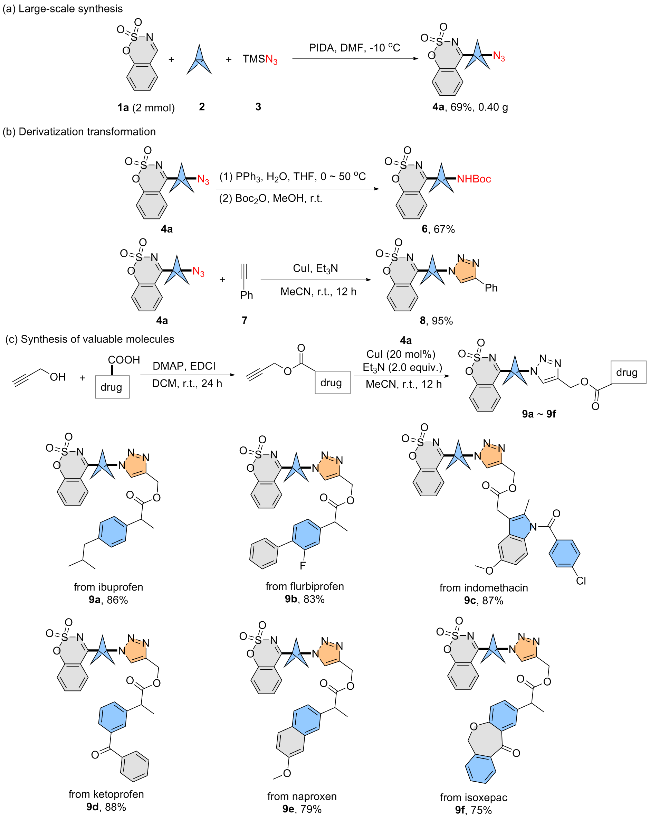
A mixture of compound 4a (0.2 mmol), phenylacetylene (7) or alkyne-containing drugs (0.3 mmol), CuI (20 mol%), Et3N (0.4 mmol, 2.0 equiv.), and MeCN (5 mL) in 25-mL Schlenk tube was stirred at room temperature for 12 h. After completion of the reaction, the reaction mixture was filtered and the solution was concentrated in vacuo. The obtained residue was further purified by silica gel column chromatography (200~300 mesh silica gel, V(PE)∶V(EA)=3∶1 for compound 8, V(PE)∶V(EA)=1∶1 for compounds 9).
4-(3-(4-Phenyl-1H-1,2,3-triazol-1-yl)bicyclo[1.1.1]-pentan-1-yl)benzo[e][1,2,3]oxathiazine 2,2-dioxide (8): Yellow solid (74 mg, 95% yield). m.p. 125~126 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.02 (dd, J=8.0, 1.6 Hz, 1H), 7.89~7.82 (m, 3H), 7.77 (ddd, J=8.7, 7.4, 1.5 Hz, 1H), 7.45 (t, J=7.6 Hz, 3H), 7.40~7.34 (m, 2H), 3.05 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 173.2, 152.8, 147.1, 136.4, 129.1, 127.9, 127.5, 127.3, 125.0, 124.9, 118.6, 117.1, 114.9, 56.0, 49.2, 39.0; HRMS (ESI-TOF) calcd for C12H10- FNO3SNa [M+Na]+ 415.0835, found 415.0843.
(1-(3-(2,2-Dioxidobenzo[e][1,2,3]oxathiazin-4-yl)bi-cyclo[1.1.1]pentan-1-yl)-1H-1,2,3-triazol-4-yl)methyl 2-(4- isobutylphenyl)propanoate (9a): Yellow solid (91 mg, 86% yield). m.p. 165~166 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 7.98 (dd, J=8.0, 1.6 Hz, 1H), 7.76 (ddd, J=8.7, 7.5, 1.5 Hz, 1H), 7.48 (s, 1H), 7.43 (td, J=7.7, 1.2 Hz, 1H), 7.35 (dd, J=8.4, 1.1 Hz, 1H), 7.19 (d, J=8.2 Hz, 2H), 7.09 (d, J=8.1 Hz, 2H), 5.27~5.20 (m, 2H), 3.73 (q, J=7.1 Hz, 1H), 2.95 (s, 6H), 2.44 (d, J=7.2 Hz, 2H), 1.84 (dt, J=13.5, 6.8 Hz, 1H), 1.49 (d, J=7.2 Hz, 3H), 0.89 (d, J=6.6 Hz, 6H); 13C NMR (126 MHz, CDCl3) δ: 174.6, 174.1, 153.8, 143.4, 140.8, 137.4, 137.3, 129.4, 128.3, 127.2, 126.0, 122.1, 119.6, 115.9, 57.8, 56.9, 50.1, 45.1, 45.0, 39.9, 30.2, 22.4, 18.4; HRMS (ESI-TOF) calcd for C28H30N4- O5SNa [M+Na]+ 557.1829, found 557.1829.
(1-(3-(2,2-Dioxidobenzo[e][1,2,3]oxathiazin-4-yl)bi-cyclo[1.1.1]pentan-1-yl)-1H-1,2,3-triazol-4-yl)methyl 2-(2- fluoro-[1'-biphenyl]-4-yl)propanoate (9b): Yellow solid (95 mg, 83% yield). m.p. 175~176 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.94 (dd, J=8.0, 1.6 Hz, 1H), 7.75 (ddd, J=8.6, 7.4, 1.5 Hz, 1H), 7.56 (s, 1H), 7.52 (dt, J=8.0, 1.5 Hz, 2H), 7.44~7.40 (m, 3H), 7.39~7.32 (m, 3H), 7.14 (dd, J=7.9, 1.8 Hz, 1H), 7.08 (dd, J=11.6, 1.8 Hz, 1H), 5.27 (s, 2H), 3.79 (q, J=7.2 Hz, 1H), 2.94 (s, 6H), 1.54 (d, J=7.1 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 174.1, 173.9, 159.6 (d, J=249.5 Hz), 153.7, 143.1, 141.4 (d, J=8.8 Hz), 137.4, 135.3, 130.8 (d, J=3.8 Hz), 128.9 (d, J=3.8 Hz), 128.6, 128.3, 127.9 (d, J=13.8 Hz), 127.8, 126.1, 123.7 (d, J=2.5 Hz), 122.2, 119.6, 115.9, 115.2 (d, J=23.4 Hz), 58.0, 56.9, 50.2, 44.8, 39.9, 18.2; 19F NMR (471 MHz, CDCl3) δ: 117.45; HRMS (ESI-TOF) calcd for C30H25F- N4O5SNa [M+Na]+ 595.1422, found 595.1415
(1-(3-(2,2-Dioxidobenzo[e][1,2,3]oxathiazin-4-yl)bi-cyclo[1.1.1]pentan-1-yl)-1H-1,2,3-triazol-4-yl)methyl 2-(1- (4-chlorobenzoyl)-5-methoxy-2-methyl-1H-indol-3-yl)ace-tate (9c): Yellow solid (119 mg, 87% yield). m.p. 154~155 ℃; 1H NMR (500 MHz, CDCl3) δ: 7.98 (dd, J=7.9, 1.5 Hz, 1H), 7.76 (ddd, J=8.6, 7.4, 1.5 Hz, 1H), 7.65 (d, J=8.5 Hz, 2H), 7.51~7.41 (m, 4H), 7.35 (dd, J=8.4, 1.0 Hz, 1H), 6.94~6.87 (m, 2H), 6.68 (dd, J=8.9, 2.5 Hz, 1H), 5.28 (s, 2H), 3.79 (s, 3H), 3.71 (s, 2H), 2.93 (s, 6H), 2.36 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 174.1, 170.7, 168.3, 156.0, 153.8, 139.4, 137.4, 136.1, 133.8, 131.2, 130.8, 130.5, 129.2, 128.3, 126.0, 119.6, 115.9, 115.0, 112.2, 111.7, 101.4, 58.1, 56.9, 55.8, 50.2, 39.9, 30.3, 13.4; HRMS (ESI-TOF) calcd for C34H28ClN5O7SNa [M+Na]+ 708.1296, found 708.1292.
(1-(3-(2,2-Dioxidobenzo[e][1,2,3]oxathiazin-4-yl)bi-cyclo[1.1.1]pentan-1-yl)-1H-1,2,3-triazol-4-yl)methyl 2-(3- benzoylphenyl)propanoate (9d): Yellow solid (102 mg, 88% yield). m.p. 166~167 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.95 (dd, J=8.0, 1.5 Hz, 1H), 7.68 (qd, J=6.7, 3.5 Hz, 4H), 7.58~7.49 (m, 3H), 7.39 (ddt, J=22.1, 15.3, 7.8 Hz, 5H), 7.26 (d, J=8.3 Hz, 1H), 5.22~5.12 (m, 2H), 3.77 (q, J=7.1 Hz, 1H), 2.88 (s, 6H), 1.47 (d, J=7.2 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 196.6, 174.4, 173.8, 153.7, 143.1, 140.6, 137.9, 137.3, 132.8, 131.7, 130.1, 129.2, 129.0, 128.6, 128.5, 128.5, 126.1, 122.3, 119.5, 115.9, 58.1, 56.9, 50.2, 45.2, 39.9, 18.3; HRMS (ESI-TOF) calcd for C31H26N4O6SNa [M+Na]+ 605.1465, found 605.1428.
(1-(3-(2,2-Dioxidobenzo[e][1,2,3]oxathiazin-4-yl)bi-cyclo[1.1.1]pentan-1-yl)-1H-1,2,3-triazol-4-yl)methyl 2-(7- methoxynaphthalen-2-yl)propanoate (9e): Yellow solid (88 mg, 79% yield). m.p. 171~172 ℃; 1H NMR (500 MHz, Chloroform-d) δ: 7.91 (dd, J=8.0, 1.5 Hz, 1H), 7.74 (s, 1H), 7.71~7.65 (m, 3H), 7.44~7.38 (m, 2H), 7.34 (dd, J=8.3, 1.0 Hz, 1H), 7.16~7.10 (m, 3H), 5.34~5.20 (m, 2H), 3.90 (d, J=7.2 Hz, 1H), 3.88 (s, 3H), 2.77 (s, 6H), 1.60 (d, J=7.1 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 174.4, 174.2, 157.9, 153.7, 143.5, 137.5, 135.2, 133.8, 129.3, 128.8, 128.4, 127.2, 126.4, 126.1, 126.1, 121.6, 119.6, 119.3, 115.9, 105.5, 57.9, 56.8, 55.4, 50.0, 45.3, 39.8, 18.0; HRMS (ESI-TOF) calcd for C29H27N4O6S [M+H]+ 559.1646, found 559.1629.
1-Ethyl-2-(3-(3,5-dioxo-2,4-di(prop-2-yn-1-yl)-2,3,4,5-tetrahydro-1,2,4-triazin-6-yl)bicyclo[1.1.1]pentan-1-yl)-acetate (9f): Yellow solid (92 mg, 75% yield). m.p. 162~163 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.09 (d, J=2.3 Hz, 1H), 7.97 (dd, J=7.9, 1.5 Hz, 1H), 7.86 (dd, J=7.6, 1.4 Hz, 1H), 7.77~7.69 (m, 2H), 7.54 (dd, J=7.5, 1.4 Hz, 1H), 7.47 (dd, J=7.5, 1.3 Hz, 1H), 7.41~7.33 (m, 4H), 7.02 (d, J=8.4 Hz, 1H), 5.28 (s, 2H), 5.18 (s, 2H), 3.67 (s, 2H), 2.97 (s, 6H); 13C NMR (126 MHz, CDCl3) δ: 189.9, 173.2, 170.3, 159.5, 152.7, 139.3, 136.4, 135.4, 134.5, 131.9, 131.4, 128.4, 128.3, 127.3, 126.9, 126.3, 125.0, 124.1, 120.2, 118.6, 114.9, 72.6, 57.0, 55.9, 49.2, 38.9, 38.9, 29.9; HRMS (ESI-TOF) calcd for C12H11FNO3S [M+H]+ 597.1438, found 597.1420.
Supporting Information Ineffective substrates, ESR experiments using 5,5-dimethyl-1-pyrroline
N-oxide (DM- PO), and the NMR spectra of compounds
4a~
4t,
6,
8,
9a~
9f. The Supporting Information is available free of charge via the Internet at
http://sioc-journal.cn.