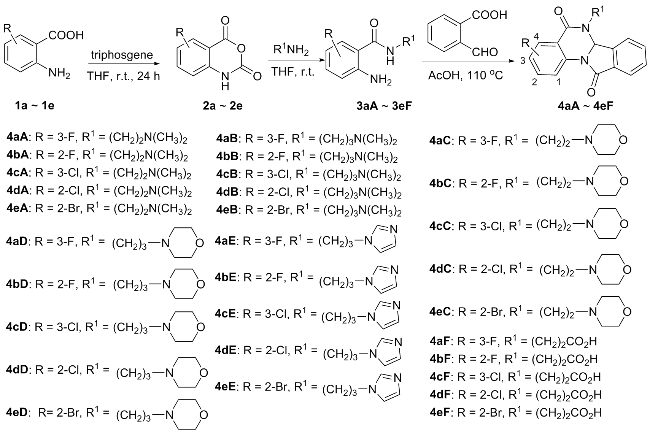
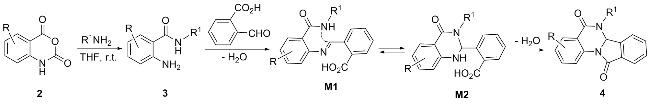
参考文献[
24]的方法并进行了适当改进, 具体实验操作如下: 将化合物
3溶解于15 mL乙酸, 随后加入等物质的量的2-醛基苯甲酸, 在氮气保护下将反应体系置于110 ℃的油浴锅中反应3~6 h. 待反应完后(TLC跟踪检测, 展开剂: 甲醇/乙酸乙酯,
V∶
V=1∶4), 减压除去部分有机溶剂, 粗产品经硅胶柱层析分离提纯(洗脱剂: 三乙胺/乙酸乙酯,
V∶
V=5∶1000), 得到目标化合物
4, 产物均为白色固体.
6-(2-二甲氨基乙基)-3-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4aA): 产率48%, m.p. 114~115 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.04~7.98 (m, 2H), 7.83 (d, J=7.6 Hz, 1H), 7.76~7.70 (m, 2H), 7.67~7.64 (m, 1H), 7.32~7.27 (m, 1H), 6.20 (s, 1H), 3.94~3.84 (m, 2H), 2.51~2.34 (m, 2H), 2.22 (s, 6H); 13C NMR (CDCl3, 125 MHz) δ: 164.8, 162.8, 160.8, 158.8, 137.9, 132.9, 132.7, 130.8, 125.4, 125.2, 122.3, 120.7, 120.6, 115.4, 115.2, 70.7, 56.8, 45.8, 41.6; HRMS (ESI) calcd for C19H19FN3O2 [M+H]+ 340.1461, found 340.1459.
6-(2-二甲氨基乙基)-2-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4bA): 产率38%, m.p. 110~111 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.11 (dd, J=8.7, 6.1 Hz, 1H), 8.03 (d, J=7.5 Hz, 1H), 7.87 (d, J=7.6 Hz, 1H), 7.82 (dd, J=9.7, 2.5 Hz, 1H), 7.75 (td, J=7.6, 1.2 Hz, 1H), 7.69 (t, J=7.4 Hz, 1H), 7.00 (td, J=8.5, 2.5 Hz, 1H), 6.25 (s, 1H), 4.05~3.81 (m, 2H), 2.58~2.43 (m, 2H), 2.24 (s, 6H); 13C NMR (125 MHz, CDCl3) δ: 166.7, 164.7, 163.2, 138.3, 138.0, 133.1, 132.5, 132.3, 131.5, 130.8, 125.4, 116.6, 112.9, 112.7, 107.5, 107.3, 70.9, 56.9, 45.8, 41.4; HRMS (ESI) calcd for C19H19FN3O2 [M+H]+ 340.1461, found 340.1459.
6-(2-二甲氨基乙基)-3-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4cA): 产率50%, m.p. 103~104 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.07~8.04 (m, 1H), 8.03~7.96 (m, 2H), 7.84 (d, J=7.6 Hz, 1H), 7.73 (d, J=7.5 Hz, 1H), 7.67 (d, J=7.5 Hz, 1H), 7.53~7.55 (m, 1H), 6.21 (s, 1H), 3.91 (t, J=7.5 Hz, 2H), 2.44 (t, J=7.5 Hz, 2H), 2.23 (s, 6H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.7, 137.9, 135.1, 133.4, 133.0, 132.6, 131.0, 128.8, 125.4, 125.2, 121.7, 70.7, 56.8, 45.8, 41.6; HRMS (ESI) calcd for C19H19ClN3O2 [M+H]+ 356.1166, found 356.1162.
6-(2-二甲氨基乙基)-2-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4dA): 产率32%, m.p. 112~114 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.13~8.11 (m, 1H), 8.06~8.02 (m, 2H), 7.87 (d, J=7.7 Hz, 1H), 7.76 (d, J=7.5Hz, 1H), 7.70 (d, J=7.5 Hz, 1H), 7.29 (d, J=2.1 Hz, 1H), 6.24 (s, 1H), 4.03~3.82 (m, 2H), 2.25 (s, 6H), 1.23~1.20 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 163.2, 139.7, 138.0, 137.5, 133.1, 132.5, 130.8, 130.3, 125.7, 125.4, 120.2, 118.7, 70.8, 58.3, 56.9, 45.8; HRMS (ESI) calcd for C19H19ClN3O2 [M+H]+ 356.1166, found 356.1159.
6-(2-二甲氨基乙基)-2-溴-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4eA): 产率43%, m.p. 136~137 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.20 (d, J=2.3 Hz, 1H), 7.99 (d, J=7.6 Hz, 1H), 7.95 (d, J=8.6 Hz, 1H), 7.83 (d, J=7.6 Hz, 1H), 7.72 (d, J=7.5 Hz, 1H), 7.67~7.58 (m, 2H), 6.20 (s, 1H), 3.90 (t, J=7.5 Hz, 2H), 2.46~2.34 (m, 2H), 2.22 (s, 6H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.5, 138.0, 136.2, 135.6, 133.0, 132.6, 131.8, 130.8, 125.4, 125.2, 121.9, 118.4, 70.6, 56.8, 45.9, 41.7; HRMS (ESI) calcd for C19H19BrN3O2 [M+H]+ 400.0661, found 400.0654.
6-(3-二甲氨基丙基)-3-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4aB): 产率60%, m.p. 69~70 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.00~7.90 (m, 2H), 7.78 (d, J=7.6 Hz, 1H), 7.72~7.63 (m, 1H), 7.60 (t, J=7.4 Hz, 1H), 7.48~7.42 (m, 1H), 7.25~7.19 (m, 1H), 6.16 (s, 1H), 3.86~3.66 (m, 2H), 2.28~2.13 (m, 2H), 2.11 (s, 6H), 1.62~1.53 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.6, 160.7, 158.8, 138.0, 135.1, 133.2, 132.8, 132.5, 130.8, 128.7, 125.4, 125.1, 122.2, 121.8, 121.6, 120.5, 120.3, 115.2, 115.1, 70.6, 56.8, 45.4, 41.6, 26.3; HRMS (ESI) calcd for C20H21FN3O2 [M+H]+ 354.1618, found 354.1610.
6-(3-二甲氨基丙基)-2-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4bB): 产率55%, m.p. 60~61 ℃; 1H NMR (500 MHz, CDCl3), δ: 8.15~7.96 (m, 2H), 7.82~7.84 (m, 2H), 7.75~7.63 (m, 2H), 7.02~6.95 (m, 1H), 6.26~6.22 (m, 1H), 3.90~3.75 (m, 2H), 2.41~2.31 (m, 2H), 2.23~2.20 (m, 6H), 1.79~1.66 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 166.6, 164.9, 163.3, 138.3, 138.1, 133.1, 132.4, 131.5, 130.8, 125.5, 116.7, 112.8, 112.7, 107.5, 107.3, 70.8, 56.5, 44.9, 41.2, 26.1; HRMS (ESI) calcd for C20H21FN3O2 [M+H]+ 354.1618, found 354.1610.
6-(3-二甲氨基丙基)-3-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4cB): 产率48%, m.p. 68~70 ℃; 1H NMR (500 MHz, CDCl3), δ: 8.07 (d, J=2.4 Hz, 1H), 8.03~8.00 (m, 2H), 7.82 (d, J=7.5 Hz, 1H), 7.71~7.73 (m, 1H), 7.66 (t, J=7.5 Hz, 1H), 7.54 (dd, J=2.5, 8.6 Hz, 1H), 6.21 (s, 1H), 3.89~3.77 (m, 2H), 2.26~2.10 (m, 2H), 2.15 (s, 6H), 1.74~1.59 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.8, 162.7, 138.1, 135.1, 133.3, 132.9, 132.6, 130.9, 130.7, 128.8, 125.4, 125.2, 121.9, 121.6, 70.6, 56.9, 45.4, 41.7, 26.4; HRMS (ESI) calcd for C20H21ClN3O2 [M+H]+ 370.1322, found 370.1316.
6-(3-二甲氨基丙基)-2-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4dB): 产率62%, m.p. 136~137 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.11~8.07 (m, 1H), 8.03~7.98 (m, 2H), 7.82 (d, J=7.5 Hz, 1H), 7.71 (d, J=7.4 Hz, 1H), 7.67~7.64 (m, 1H), 7.26~7.20 (m, 1H), 6.21 (s, 1H), 3.87~3.76 (m, 2H), 2.29~2.21 (m, 2H), 2.15 (s, 6H), 1.73~1.58 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 163.1, 139.5, 138.1, 137.4, 133.0, 132.5, 130.7, 130.3, 125.6, 125.3, 120.2, 118.9, 70.7, 56.9, 45.4, 41.5, 26.4; HRMS (ESI) calcd for C20H21ClN3O2 [M+H]+ 370.1322, found 370.1314.
6-(3-二甲氨基丙基)-2-溴-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4eB): 产率55%, m.p. 98~101 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.21 (d, J=2.2 Hz, 1H), 7.98~7.96 (m, 2H), 7.82 (d, J=7.6 Hz, 1H), 7.68~7.69 (m, 3H), 6.20 (s, 1H), 3.90~3.73 (m, 2H), 2.26~2.30 (m, 2H), 2.16 (s, 6H), 1.74~1.57 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.8, 162.5, 138.1, 136.2, 135.6, 133.0, 132.6, 131.8, 130.8, 125.4, 125.2, 122.1, 121.9, 118.4, 70.6, 56.8, 45.4, 41.6, 26.3; HRMS (ESI) calcd for C20H21- BrN3O2 [M+H]+ 414.0817, found 414.0813.
6-[2-(4-吗啡啉基)乙基]-3-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4aC): 产率68%, m.p. 128~129 ℃; 1H NMR (500 MHz, CDCl3) δ 8.40~8.32 (m, 2H), 8.15 (d, J=7.5 Hz, 1H), 8.12~8.09 (m, 1H), 8.08~7.99 (m, 2H), 7.68~7.61 (m, 1H), 6.54 (s, 1H), 4.27 (dt, J=7.8, 5.9 Hz, 2H), 3.96~3.90 (m, 4H), 2.78~2.74 (m, 4H), 2.72~2.59 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.8, 160.8, 158.9, 138.0, 132.9, 130.8, 125.3, 122.2, 120.7, 120.5, 115.3, 115.1, 70.6, 66.8, 56.1, 53.7, 40.8; HRMS (ESI) calcd for C21H21FN3O3 [M+H]+ 382.1567, found 382.1562.
6-[2-(4-吗啡啉基)乙基]-2-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4bC): 产率65%, m.p. 114~115 ℃; 1H NMR (500 MHz, CDCl3), δ: 8.09 (dd, J=8.7, 2.4 Hz, 1H), 8.01 (d, J=7.3 Hz, 1H), 7.84~7.80 (m, 2H), 7.73~7.65 (m, 2H), 6.98~6.70 (m, 1H), 6.23 (s, 1H), 3.93~3.78 (m, 2H), 3.65~3.64 (m, 4H), 2.35~2.20 (m, 4H), 1.76~1.64 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 166.6, 164.7, 163.3, 138.4, 132.9, 132.6, 131.4, 130.8, 125.3, 116.7, 112.9, 112.7, 107.5, 107.3, 70.8, 66.8, 56.1, 53.7, 40.6; HRMS (ESI) calcd for C21H21FN3O3 [M+H]+ 382.1567, found 382.1565.
6-[2-(4-吗啡啉基)乙基]-3-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4cC): 产率70%, m.p. 166~168 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.09~8.08 (m, 3H), 7.83 (d, J=7.3 Hz, 1H), 7.73~7.65 (m, 2H), 7.54~7.50 (m, 1H), 6.20 (s, 1H), 3.93~3.78 (m, 2H), 3.64~3.63 (m, 4H), 2.34~3.33 (m, 4H), 1.68~1.70 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.6, 162.8, 138.0, 135.2, 133.4, 132.9, 132.7, 130.9, 130.8, 128.8, 125.3, 121.8, 121.6, 70.6, 66.9, 56.3, 54.0, 40.8; HRMS (ESI) calcd for C21H21ClN3O3 [M+H]+ 398.1271, found 398.1264.
6-[2-(4-吗啡啉基)乙基]-2-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4dC): 产率66%, m.p. 145~146 ℃; 1H NMR (500 MHz, CDCl3), δ: 8.17~8.06 (m, 1H), 8.03 (d, J=8.3 Hz, 2H), 7.84 (d, J=7.5 Hz, 1H), 7.74 (br, 2H), 7.29~7.26 (m, 1H), 6.22 (s, 1H), 3.94~3.79 (m, 2H), 3.66~3.65 (m, 4H), 2.41~2.29 (m, 4H), 1.76~1.63 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.6, 163.3, 139.7, 138.1, 137.5, 133.0, 132.6, 130.8, 130.3, 125.7, 125.4, 125.3, 120.1, 118.8, 70.7, 66.9, 56.3, 54.0, 40.7; HRMS (ESI) calcd for C21H21ClN3O3 [M+H]+ 398.1271, found 398.1271.
6-[2-(4-吗啡啉基)乙基]-2-溴-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4eC): 产率63%, m.p. 167~168 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.21 (d, J=2.1 Hz, 1H), 8.02~7.95 (m, 2H), 7.83 (d, J=7.4 Hz, 1H), 7.69~7.60 (m, 3H), 6.20 (s, 1H), 3.93~3.77 (m, 2H), 3.65 (t, J=4.5 Hz, 4H), 2.39~2.28 (m, 4H), 1.75~1.61 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.6, 138.1, 136.2, 135.6, 132.8, 132.7, 131.8, 130.8, 125.3, 122.0, 121.8, 118.4, 77.3, 77.0, 76.8, 70.6, 66.9, 56.3, 54.0, 40.8; HRMS (ESI) calcd for C21H21BrN3O3 [M+H]+ 442.0766, found 442.0760.
6-[3-(4-吗啡啉基)丙基]-3-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4aD): 产率53%, m.p. 179~180 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.08~7.96 (m, 2H), 7.82 (d, J=7.3 Hz, 1H), 7.76 (dt, J=8.5, 2.9 Hz, 1H), 7.72~7.63 (m, 2H), 7.34~7.27 (m, 1H), 6.21 (s, 1H), 3.98~3.75 (m, 2H), 3.70~3.61 (m, 4H), 2.46~2.28 (m, 6H), 1.78~1.61 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.8, 160.8, 158.9, 138.0, 132.7, 130.8, 125.3, 122.2, 120.7, 120.5, 115.3, 115.1, 70.6, 66.8, 56.1, 53.7, 41.7, 25.0; HRMS (ESI) calcd for C22H23FN3O3 [M+H]+ 396.1723, found 396.1718.
6-[3-(4-吗啡啉基)丙基]-2-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4bD): 产率55%, m.p. 122~123 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.09~8.01 (m, 1H), 8.01 (dd, J=2.9, 7.4 Hz, 1H), 7.84~7.79 (m, 2H), 7.74~7.65 (m, 2H), 6.98~6.88 (m, 1H), 6.22 (s, 1H), 3.97~3.86 (m, 2H), 3.59 (br, 4H), 2.14~2.23 (m, 6H), 1.81~1.58 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 166.6, 164.7, 163.3, 138.3, 138.1, 132.9, 132.6, 131.4, 130.8, 125.3, 116.7, 112.9, 112.7, 107.5, 107.3, 70.8, 66.8, 56.1, 53.7, 41.5, 25.1; HRMS (ESI) calcd for C22H23FN3O3 [M+H]+ 396.1723, found 396.1720.
6-[3-(4-吗啡啉基)丙基]-3-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4cD): 产率50%, m.p. 68~69 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.06~7.99 (m, 3H), 7.81 (d, J=7.4 Hz, 1H), 7.74~7.66 (m, 2H), 7.58~7.51 (m, 1H), 6.20 (s, 1H), 3.92~3.88 (m, 2H), 3.62~3.56 (m, 4H), 2.43~2.39 (m, 6H), 2.32~2.26 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.7, 138.1, 135.1, 133.4, 132.8, 132.7, 130.9, 130.8, 128.8, 125.4, 125.3, 121.8, 121.6, 70.6, 66.9, 56.2, 53.8, 41.7, 25.1; HRMS (ESI) calcd for C22H23ClN3O3 [M+H]+ 412.1428, found 412.1423.
6-[3-(4-吗啡啉基)丙基]-2-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4dD): 产率58%, m.p. 126~127 ℃; 1H NMR (500 MHz, CDCl3), δ: 8.12~8.08 (m, 1H), 8.04~8.00 (m, 2H), 7.82 (d, J=7.6 Hz, 1H), 7.71~7.68 (m, 2H), 7.33~7.26 (m, 1H), 6.21 (s, 1H), 3.97~3.86 (m, 2H), 3.59 (br, 4H), 2.42 (br, 6H), 2.40~2.29 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.6, 163.2, 139.6, 138.1, 137.4, 132.9, 132.6, 130.8, 130.3, 125.7, 125.3, 120.2, 118.8, 70.7, 66.9, 56.2, 53.8, 41.6, 25.2; HRMS (ESI) calcd for C22H23ClN3O3 [M+H]+ 412.1428, found 412.1426.
6-[3-(4-吗啡啉基)丙基]-2-溴-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4eD): 产率60%, m.p. 114~115 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.21 (d, J=1.1 Hz, 1H), 8.02~7.95 (m, 2H), 7.83 (d, J=7.4 Hz, 1H), 7.73~7.64 (m, 3H), 6.20 (s, 1H), 3.95~3.74 (m, 2H), 3.68~3.60 (m, 4H), 2.45~2.24 (m, 6H), 1.78~1.58 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.6, 138.1, 136.2, 135.6, 132.8, 132.7, 131.8, 130.8, 125.4, 125.3, 122.0, 121.8, 118.4, 70.5, 66.9, 56.2, 53.8, 41.7, 25.1; HRMS (ESI) calcd for C22H23BrN3O3 [M+H]+ 456.0923, found 456.0914.
6-[3-(咪唑基)丙基]-3-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4aE): 产率38%, m.p. 182~184 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.05~7.96 (m, 2H), 7.77 (dd, J=2.3, 8.5 Hz, 1H), 7.66~7.57 (m, 2H), 7.42 (s, 1H), 7.35~7.29 (m, 1H), 7.18 (d, J=7.6 Hz, 1H), 7.09 (d, J=5.4 Hz, 1H), 6.86 (t, J=1.2 Hz, 1H), 6.12 (s, 1H), 4.04~3.87 (m, 3H), 3.72~3.60 (m, 1H), 1.91~1.73 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.8, 163.2, 160.9, 158.9, 137.5, 137.2, 133.4, 132.8, 132.5, 130.9, 129.8, 125.3, 124.6, 122.4, 121.9, 121.1, 120.9, 119.0, 115.4, 115.2, 70.5, 44.6, 40.7, 30.3; HRMS (ESI) calcd for C21H18FN4O2 [M+H]+ 377.1414, found 377.1395.
6-[3-(咪唑基)丙基]-2-氟-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4bE): 产率32%, m.p. 190~193 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.08 (dd, J=2.4, 8.7 Hz, 1H), 7.96 (d, J=7.4 Hz, 1H), 7.77 (dd, J=2.4, 9.6 Hz, 1H), 7.68~7.56 (m, 2H), 7.41 (s, 1H), 7.19 (d, J=7.5 Hz, 1H), 7.05 (d, J=4.2 Hz, 1H), 7.01~6.94 (m, 1H), 6.85 (s, 1H), 6.14 (s, 1H), 4.01~3.87 (m, 3H), 3.64 (br, 1H), 1.80~1.83(m, 2H); 13C NMR (125 MHz, CDCl3) δ: 171.1, 168.5, 163.5, 140.3, 137.1, 133.5, 132.3, 132.0, 131.5, 130.9, 129.7, 129.4, 125.4, 124.6, 119.0, 116.3, 113.0, 112.8, 107.7, 107.4, 70.6, 44.6, 40.4, 30.3; HRMS (ESI) calcd for C21H18FN4O2 [M+H]+ 377.1414, found 377.1392.
6-[3-(咪唑基)丙基]-3-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4cE): 产率44%, m.p. 208~210 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.05 (d, J=2.5 Hz, 1H), 8.01~7.93 (m, 2H), 7.67~7.53 (m, 3H), 7.42 (s, 1H), 7.17 (d, J=7.6 Hz, 1H), 7.09~7.08 (m, 1H), 6.86~6.85 (m, 1H), 6.11 (s, 1H), 3.8~4.3 (m, 3H), 3.72~3.56 (m, 1H), 1.91~1.74 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 163.0, 137.5, 137.2, 135.1, 133.7, 133.5, 132.4, 131.0, 129.8, 128.8, 125.4, 124.7, 121.8, 121.4, 119.1, 70.4, 44.6, 40.6, 30.3; HRMS (ESI) calcd for C21H18Cl- N4O2 [M+H]+ 393.1118, found 393.1095.
6-[3-(咪唑基)丙基]-2-氯-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4dE): 产率56%, m.p. 187~189 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.07 (d, J=8.5 Hz, 1H), 7.80~7.98 (m, 2H), 7.66~7.57 (m, 2H), 7.42 (s, 1H), 7.26~7.24 (m, 1H), 7.18 (d, J=7.5 Hz, 1H), 7.09~7.07 (m, 1H), 6.86 (s, 1H), 6.12 (s, 1H), 4.03~3.88 (m, 3H), 3.65 (br, 1H), 1.91~1.76 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.6, 163.5, 139.9, 137.5, 137.2, 133.5, 132.3, 130.9, 130.3, 129.7, 125.8, 125.5, 124.7, 120.3, 119.0, 118.4, 70.5, 44.6, 40.5, 30.3; HRMS (ESI) calcd for C21H18ClN4O2 [M+H]+ 393.1118, found 393.1094.
6-[3-(咪唑基)丙基]-2-溴-6,6a-二氢异吲哚并[2,1-a]喹唑啉-5,11-二酮(4eE): 产率60%, m.p. 207~209 ℃; 1H NMR (500 MHz, CDCl3) δ: 8.22 (d, J=2.3 Hz, 1H), 7.99~7.97 (m, 2H), 7.71 (dd, J=2.3, 8.6 Hz, 1H), 7.67~7.58 (m, 2H), 7.42 (s, 1H), 7.18 (d, J=7.6 Hz, 1H), 7.08 (s, 1H), 6.86 (s, 1H), 6.12 (s, 1H), 4.04~3.88 (m, 3H), 3.69~3.61 (m, 1H), 1.90~1.75 (m, 2H); 13C NMR (125 MHz, CDCl3) δ: 164.7, 162.9, 137.4, 137.1, 136.5, 135.6, 133.5, 132.4, 131.8, 130.9, 129.7, 125.4, 124.7, 122.0, 121.6, 119.1, 118.6, 70.4, 44.6, 40.6, 30.3; HRMS (ESI) calcd for C21H18BrN4O2 [M+H]+ 437.0613, found 437.0586.
3-(3-氟-5,11-二氧代-6a,11-二氢-5H-异吲哚并[2,1- a]-6-喹唑啉基)丙酸(4aF): 产率64%, m.p. 203~205 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 12.36 (br, 1H), 8.07~7.99 (m, 2H), 7.95 (d, J=7.5 Hz, 1H), 7.82~7.86 (m, 1H), 7.77~7.75 (m, 1H), 7.70 (dd, J=2.5, 8.7 Hz, 1H), 7.50~7.62 (m, 1H), 6.59 (s, 1H), 4.01~3.85 (m, 2H), 2.62~2.60 (m, 1H), 2.28~2.20 (m, 1H); 13C NMR (125 MHz, DMSO-d6) δ: 172.9, 164.8, 162.3, 160.4, 158.4, 138.6, 133.7, 133.4, 132.2, 131.3, 126.7, 124.8, 122.9, 122.6, 121.1, 120.9, 114.8, 114.6, 70.6, 39.4, 33.0; HRMS (ESI) calcd for C18H12FN2O4 [M-H]- 339.0781, found 339.0797.
3-(2-氟-5,11-二氧代-6a,11-二氢-5H-异吲哚并[2,1- a]-6-喹唑啉基)丙酸(4bF): 产率44%, m.p. 233~235 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 12.35 (br, 1H), 8.05~8.03 (m, 2H), 7.95 (d, J=7.5 Hz, 1H), 7.81~7.85 (m, 1H), 7.71~7.78 (m, 2H), 7.12~7.18 (m, 1H), 6.59 (s, 1H), 4.03~3.79 (m, 2H), 2.52~2.62 (m, 1H), 2.36~2.20 (m, 1H); 13C NMR (125 MHz, DMSO-d6) δ: 172.9, 172.5, 166.0, 164.9, 164.0, 162.7, 138.6, 133.9, 132.1, 131.7, 131.3, 126.7, 125.0, 117.3, 113.0, 112.8, 107.3, 107.1, 70.7, 39.4, 32.9; HRMS (ESI) calcd for C18H12FN2O4 [M-H]- 339.0781, found 339.0793.
3-(3-氯-5,11-二氧代-6a,11-二氢-5H-异吲哚并[2,1- a]-6-喹唑啉基)丙酸(4cF): 产率56%, m.p. 245~247 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 12.39 (br, 1H), 8.06~8.03 (m, 2H), 7.97 (d, J=7.4 Hz, 1H), 7.92 (d, J=2.5 Hz, 1H), 7.87~7.85 (m, 1H), 7.83~7.72 (m, 2H), 6.59 (s, 1H), 4.03~3.83 (m, 2H), 2.61 (br, 1H), 2.34~2.17 (m, 1H); 13C NMR (125 MHz, DMSO-d6) δ: 172.8, 164.9, 162.3, 138.6, 135.8, 133.8, 133.6, 132.2, 131.3, 129.6, 128.1, 126.8, 124.9, 122.4, 70.6, 39.4, 32.9; HRMS (ESI) calcd for C18H12ClN2O4 [M-H]- 355.0486, found 355.0493.
3-(2-氯-5,11-二氧代-6a,11-二氢-5H-异吲哚并[2,1- a]-6-喹唑啉基)丙酸(4dF): 产率66%, m.p. 268~270 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 12.37 (br, 1H), 8.05~8.01 (m, 2H), 7.97 (dd, J=2.2, 7.8 Hz, 2H), 7.88~7.84 (m, 1H), 7.76 (t, J=7.5 Hz, 1H), 7.42 (dd, J=2.0, 8.4 Hz, 1H), 6.59 (s, 1H), 4.03~3.83 (m, 2H), 2.59~2.62 (m, 1H), 2.32~2.20 (m, 1H); 13C NMR (125 MHz, DMSO-d6) δ: 172.8, 164.9, 162.6, 138.6, 138.1, 137.9, 133.9, 132.0, 131.3, 130.7, 126.7, 125.6, 125.0, 119.9, 119.43, 70.66, 39.4, 32.9; HRMS (ESI) calcd for C18H12ClN2O4 [M- H]- 355.0486, found 355.0492.
3-(2-溴-5,11-二氧代-6a,11-二氢-5H-异吲哚并[2,1- a]-6-喹唑啉基)丙酸(4eF): 产率50%, m.p. 244~246 ℃; 1H NMR (500 MHz, DMSO-d6) δ: 8.06~8.01 (m, 2H), 7.96 (d, J=8.5 Hz, 2H), 7.91~7.83 (m, 2H), 7.76 (t, J=7.5 Hz, 1H), 6.58 (s, 1H), 4.00~3.86 (m, 2H), 2.60 (br, 1H), 2.30~2.22 (m, 1H); 13C NMR (125 MHz, DMSO-d6) δ: 172.8, 164.8, 162.2, 138.6, 136.3, 136.1, 133.8, 132.2, 131.3, 131.0, 126.7, 124.9, 122.6, 117.5, 70.5, 39.4, 32.9; HRMS (ESI) calcd for C18H12BrN2O4 [M-H]- 398.9980, found 398.9987.