

羌活中两个新的线型呋喃香豆素类成分及其抗炎活性
收稿日期: 2025-06-20
修回日期: 2025-07-15
网络出版日期: 2025-08-27
基金资助
国家自然科学基金(82304887)
四川省科技计划(2024ZYD0116)
Two New Linear Furocoumarins from Notopterygium incisum and Their Anti-inflammatory Activity
Received date: 2025-06-20
Revised date: 2025-07-15
Online published: 2025-08-27
Supported by
National Natural Science Foundation of China(82304887)
Sichuan Science and Technology Program(2024ZYD0116)
胡苏 , 赵丽莲 , 李昕宇 , 叶雨蓓 , 邓赟 , 黄莉钧 , 郭大乐 . 羌活中两个新的线型呋喃香豆素类成分及其抗炎活性[J]. 有机化学, 2026 , 46(1) : 300 -303 . DOI: 10.6023/cjoc202505002
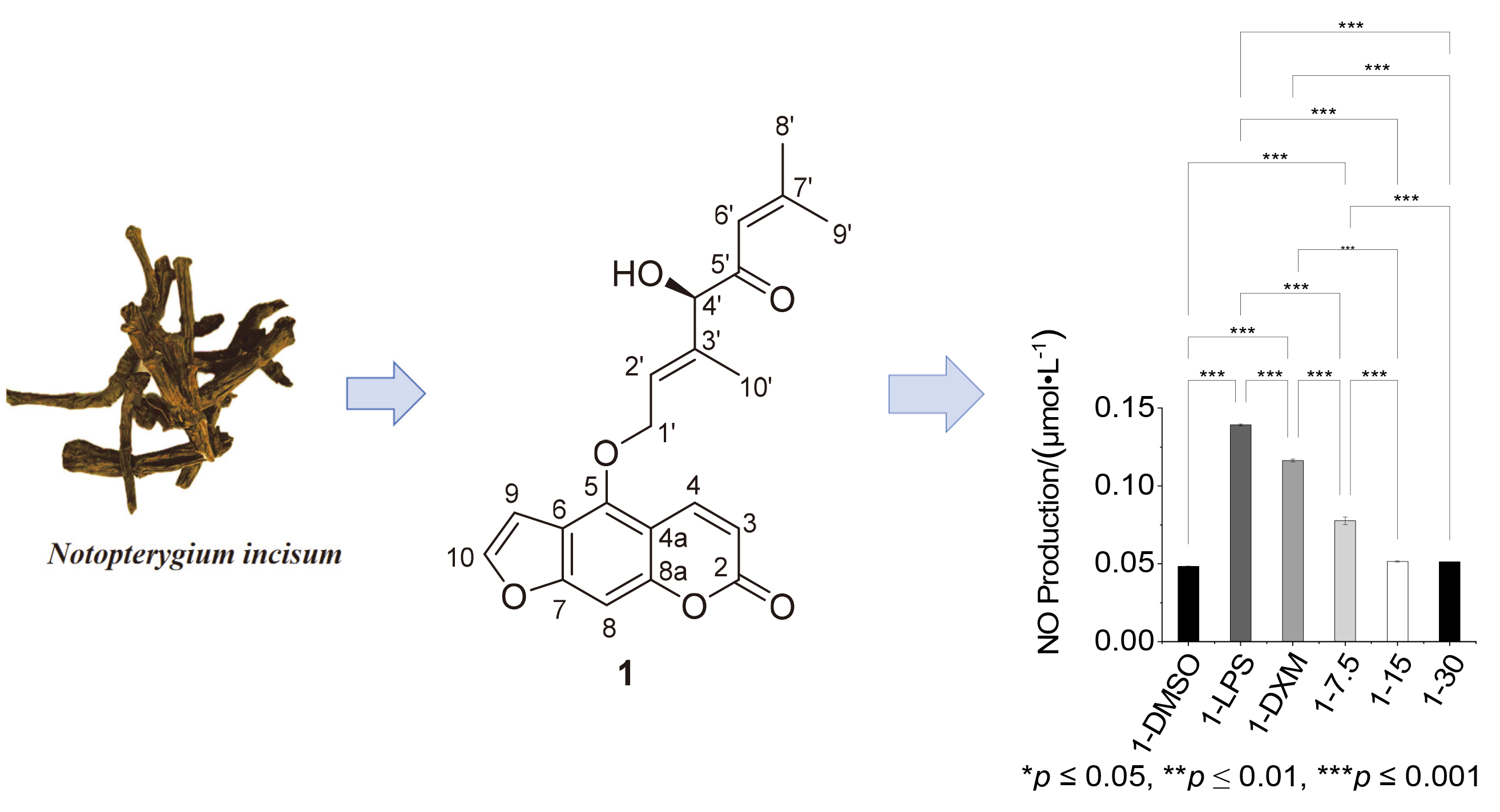
Two new linear furocoumarins were isolated from Notopterygium incisum using silica gel, gel Sephadex LH-20 column chromatography, and preparative high-performance liquid chromatography. The structures of the new compounds were elucidated through analysis of spectroscopic evidence, including data obtained from high-resolution electrospray ionization mass spectrometry (HRESIMS) and nuclear magnetic resonance (NMR). The absolute configurations of Notoprenylate L (1) and Nototerprinol K (2) were further confirmed using electronic circular dichroism (ECD) calculations. Compound 1 demonstrated the ability to inhibit the expression of nitric oxide (NO), a pro-inflammatory factor, in lipopolysaccharide-induced RAW264.7 macrophages at a concentration of 7.5 μmol/L, indicating its potential for anti-inflammatory activity.
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
(毕建平, 李平, 徐习习, 王婷, 李飞, 中国天然药物, 2018, 16, 926.)
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
/
| 〈 |
|
〉 |