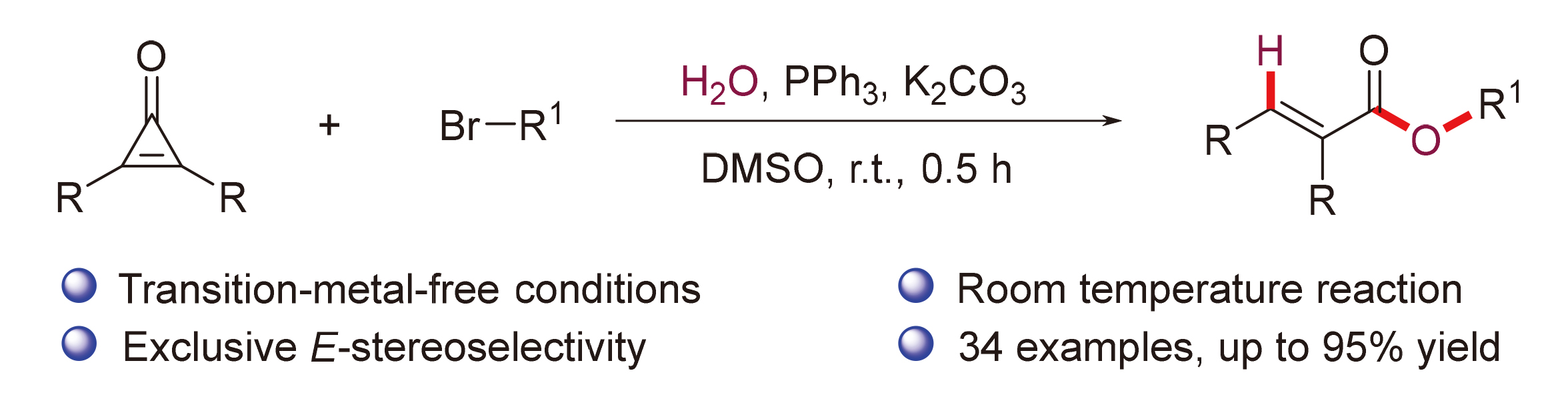
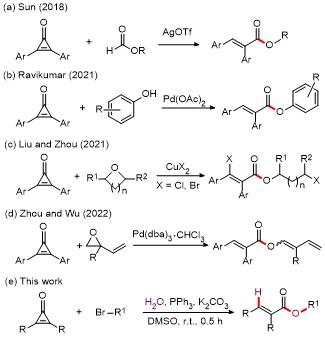
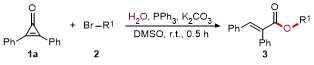
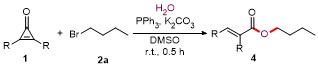
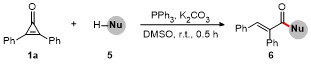
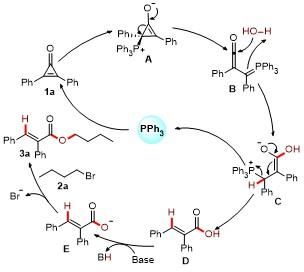
A mixture of cyclopropenones 1 (0.2 mmol), alkyl bromides 2 (0.3 mmol), PPh3 (5.2 mg, 0.02 mmol), K2CO3 (55.3 mg, 0.4 mmol), and DMSO (3.0 mL) was stirred at room temperature under air for 0.5 h. Following complete consumption of starting materials (verified by TLC analysis), the reaction underwent extraction using EtOAc (30 mL×2), followed by sequential washing with saturated NaCl solution (30 mL). The collected organic phase was dried over anhydrous Na2SO4 and evaporated under vacuum. Final purification of the residue through silica gel column chromatography (petroleum ether/ethyl acetate, V∶V 10∶1~15∶1) afforded products 3 and 4.
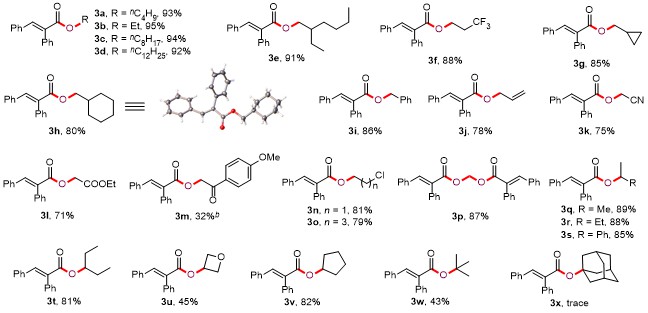
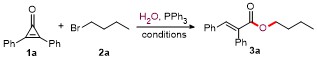
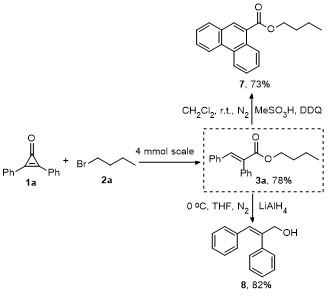
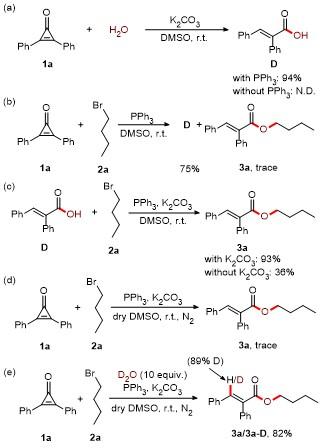
Butyl (E)-2,3-diphenylacrylate (3a): Yellow oil (52.1 mg, 93% yield). 1H NMR (300 MHz, CDCl3) δ: 7.83 (s, 1H), 7.36~7.33 (m, 3H), 7.24~7.11 (m, 5H), 7.04 (d, J=7.5 Hz, 2H), 4.20 (t, J=6.6 Hz, 2H), 1.68~1.59 (m, 2H), 1.41~1.32 (m, 2H), 0.91 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 167.8, 140.0, 135.9, 134.7, 132.8, 130.5, 129.7, 128.9, 128.5, 128.1, 127.7, 65.0, 30.6, 19.1, 13.7; HRMS (ESI) calcd for C19H21O2 [M+H]+ 281.1536, found 281.1532.
Ethyl (E)-2,3-diphenylacrylate (3b): Yellow oil (47.9 mg, 95% yield). 1H NMR (300 MHz, CDCl3) δ: 7.84 (s, 1H), 7.36~7.34 (m, 3H), 7.23~7.21 (m, 2H), 7.16 (t, J=6.6 Hz, 3H), 7.05~7.04 (m, 2H), 4.28 (q, J=6.9 Hz, 2H), 1.31 (t, J=6.6 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 167.7, 140.0, 135.8, 134.6, 132.7, 130.5, 129.7, 128.9, 128.5, 128.1, 127.7, 61.1, 14.2; HRMS (ESI) calcd for C17H17O2 [M+H]+ 253.1223, found 253.1219.
Octyl (E)-2,3-diphenylacrylate (3c): Yellow oil (63.3 mg, 94% yield). 1H NMR (300 MHz, CDCl3) δ: 7.83 (s, 1H), 7.37~7.32 (m, 3H), 7.23~7.10 (m, 5H), 7.04 (d, J=6.6 Hz, 2H), 4.19 (t, J=6.6 Hz, 2H), 1.66~1.59 (m, 2H), 1.28~1.26 (m, 10H), 0.88 (t, J=6.6 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 167.8, 140.0, 135.9, 134.6, 132.8, 130.5, 129.7, 128.9, 128.5, 128.1, 127.6, 65.3, 31.7, 29.12, 29.08, 28.5, 25.8, 22.6, 14.1; HRMS (ESI) calcd for C23H29O2 [M+H]+ 337.2162, found 337.2157.
Dodecyl (E)-2,3-diphenylacrylate (3d): White solid (72.2 mg, 92% yield). m.p. 31~32 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.83 (s, 1H), 7.37~7.33 (m, 3H), 7.24~7.11 (m, 5H), 7.04 (d, J=6.6 Hz, 2H), 4.19 (t, J=6.9 Hz, 2H), 1.66~1.61 (m, 2H), 1.26~1.25 (m, 18H), 0.88 (t, J=6.6 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 167.8, 140.0, 135.9, 134.7, 132.9, 130.5, 129.7, 128.9, 128.5, 128.1, 127.7, 65.3, 31.9, 29.63, 29.62, 29.52, 29.49, 29.3, 29.2, 28.6, 25.9, 22.7, 14.1; HRMS (ESI) calcd for C27H37O2 [M+H]+ 393.2788, found 393.2782.
2-Ethylhexyl (E)-2,3-diphenylacrylate (3e): Yellow oil (61.2 mg, 91% yield). 1H NMR (300 MHz, CDCl3) δ: 7.83 (s, 1H), 7.38~7.33 (m, 3H), 7.25~7.12 (m, 5H), 7.06 (d, J=6.9 Hz, 2H), 4.11 (d, J=5.7 Hz, 2H), 1.59~1.54 (m, 1H), 1.30~1.23 (m, 8H), 0.89~0.82 (m, 6H); 13C NMR (75 MHz, CDCl3) δ: 167.9, 139.9, 136.0, 134.7, 132.9, 130.6, 129.6, 128.9, 128.5, 128.1, 127.6, 67.5, 38.8, 30.5, 28.8, 23.9, 22.9, 14.0, 11.0; HRMS (ESI) calcd for C23H29- O2 [M+H]+ 337.2162, found 337.2161.
3,3,3-Trifluoropropyl (E)-2,3-diphenylacrylate (3f): Yellow oil (56.4 mg, 88% yield). 1H NMR (300 MHz, CDCl3) δ: 7.86 (s, 1H), 7.37~7.35 (m, 3H), 7.24~7.13 (m, 5H), 7.04 (d, J=6.9 Hz, 2H), 4.42 (t, J=6.6 Hz, 2H), 2.57~2.42 (m, 2H); 13C NMR (75 MHz, CDCl3) δ: 167.3, 141.1, 135.4, 134.4, 131.9, 130.7, 129.7, 129.2, 128.6, 128.2, 127.9, 125.8 (q, 1JC-F=274.5 Hz), 57.8 (q, 3JC-F=3.8 Hz), 33.3 (q, 2JC-F=29.3 Hz); HRMS (ESI) calcd for C18H16F3O2 [M+H]+ 321.1097, found 321.1091.
Cyclopropylmethyl (E)-2,3-diphenylacrylate (3g): Yellow solid (47.3 mg, 85% yield). m.p. 40~41 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.86 (s, 1H), 7.33~7.31 (m, 3H), 7.23~7.21 (m, 2H), 7.15~7.08 (m, 3H), 7.03 (d, J=7.2 Hz, 2H), 4.04 (d, J=6.9 Hz, 2H), 1.22~1.09 (m, 1H), 0.55~0.49 (m, 2H), 0.29~0.25 (m, 2H); 13C NMR (75 MHz, CDCl3) δ: 167.6, 139.9, 135.8, 134.5, 132.7, 130.4, 129.6, 128.8, 128.4, 128.0, 127.6, 69.5, 9.8, 3.1; HRMS (ESI) calcd for C19H19O2 [M+H]+ 279.1380, found 279.1374.
Cyclohexylmethyl (E)-2,3-diphenylacrylate (3h): Yellow solid (51.3 mg, 80% yield). m.p. 73~74 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.74 (s, 1H), 7.27~7.23 (m, 3H), 7.13~7.11 (m, 2H), 7.08~7.00 (m, 3H), 6.96 (d, J=7.2 Hz, 2H), 3.92 (d, J=6.0 Hz, 2H), 1.61~1.58 (m, 6H), 1.17~1.00 (m, 3H), 0.90~0.79 (m, 2H); 13C NMR (75 MHz, CDCl3) δ: 167.7, 139.9, 135.9, 134.6, 132.8, 130.5, 129.6, 128.8, 128.4, 128.0, 127.6, 70.2, 37.0, 29.5, 26.3, 25.6; HRMS (ESI) calcd for C22H25O2 [M+H]+ 321.1849, found 321.1849.
Benzyl (E)-2,3-diphenylacrylate (3i): Yellow solid (54.1 mg, 86% yield). m.p. 78~79 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.87 (s, 1H), 7.36~7.32 (m, 8H), 7.24~7.21 (m, 2H), 7.18~7.13 (m, 3H), 7.03 (d, J=6.9 Hz, 2H), 5.25 (s, 2H); 13C NMR (75 MHz, CDCl3) δ: 167.5, 140.6, 136.1, 135.7, 134.5, 132.4, 130.6, 129.7, 129.0, 128.6, 128.4, 128.1, 127.9, 127.8, 127.7, 66.7; HRMS (ESI) calcd for C22H19O2 [M+H]+ 315.1380, found 315.1378.
Allyl (E)-2,3-diphenylacrylate (3j): Yellow oil (41.2 mg, 78% yield). 1H NMR (300 MHz, CDCl3) δ: 7.87 (s, 1H), 7.36~7.35 (m, 3H), 7.24~7.13 (m, 5H), 7.05 (d, J=6.6 Hz, 2H), 6.00~5.88 (m, 1H), 5.26 (d, J=17.4 Hz, 1H), 5.20 (d, J=10.8 Hz, 1H), 4.71 (d, J=5.1 Hz, 2H); 13C NMR (75 MHz, CDCl3) δ: 167.4, 140.5, 135.7, 134.5, 132.4, 132.1, 130.6, 129.7, 129.0, 128.6, 128.2, 127.8, 117.7, 65.6; HRMS (ESI) calcd for C18H17O2 [M+H]+ 265.1223, found 265.1219.
Cyanomethyl (E)-2,3-diphenylacrylate (3k): White solid (39.5 mg, 75% yield). m.p. 84~85 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.92 (s, 1H), 7.39~7.36 (m, 3H), 7.23~7.20 (m, 3H), 7.15 (t, J=7.5 Hz, 2H), 7.04 (d, J=7.5 Hz, 2H), 4.77 (s, 2H); 13C NMR (75 MHz, CDCl3) δ: 166.0, 143.0, 134.6, 133.8, 130.8, 130.1, 129.7, 129.6, 128.8, 128.22, 128.19, 114.5, 48.8; HRMS (ESI) calcd for C17H14NO2 [M+H]+ 264.1019, found 264.1020.
2-Ethoxy-2-oxoethyl (E)-2,3-diphenylacrylate (3l): White solid (44.1 mg, 71% yield). m.p. 68~69 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.93 (s, 1H), 7.40~7.36 (m, 3H), 7.30~7.25 (m, 2H), 7.22~7.13 (m, 3H), 7.06 (d, J=6.9 Hz, 2H), 4.72 (s, 2H), 4.24 (q, J=7.2 Hz, 2H), 1.30 (t, J=7.2 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 167.8, 167.2, 141.7, 135.4, 134.4, 131.5, 130.7, 129.8, 129.3, 128.6, 128.2, 127.9, 61.4, 61.3, 14.1; HRMS (ESI) calcd for C19H19O4 [M+H]+ 311.1278, found 311.1274.
2-(4-Methoxyphenyl)-2-oxoethyl (E)-2,3-diphenylacryl- ate (3m): White solid (23.8 mg, 32% yield). m.p. 131~132 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.97 (s, 1H), 7.91 (d, J=8.7 Hz, 2H), 7.41~7.33 (m, 5H), 7.26~7.14 (m, 3H), 7.08 (d, J=7.5 Hz, 2H), 6.95 (d, J=9.0 Hz, 2H), 5.41 (s, 2H), 3.88 (s, 3H); 13C NMR (75 MHz, CDCl3) δ: 190.6, 167.3, 163.9, 141.5, 135.5, 134.5, 131.7, 130.7, 130.1, 129.9, 129.1, 128.6, 128.2, 127.9, 127.2, 114.0, 66.5, 55.5; HRMS (ESI) calcd for C24H21O4 [M+H]+ 373.1434, found 373.1433.
2-Chloroethyl (E)-2,3-diphenylacrylate (3n): White solid (46.5 mg, 81% yield). m.p. 49~50 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.88 (s, 1H), 7.35~7.31 (m, 3H), 7.23~7.20 (m, 2H), 7.16~7.08 (m, 3H), 7.03 (d, J=6.9 Hz, 2H), 4.40 (t, J=5.7 Hz, 2H), 3.65 (t, J=5.7 Hz, 2H); 13C NMR (75 MHz, CDCl3) δ: 167.1, 140.9, 135.3, 134.3, 131.8, 130.5, 129.6, 129.0, 128.4, 128.0, 127.7, 64.4, 41.4; HRMS (ESI) calcd for C17H16ClO2 [M+H]+ 287.0833, found 287.0833.
4-Chlorobutyl (E)-2,3-diphenylacrylate (3o): Yellow oil (49.7 mg, 79% yield). 1H NMR (300 MHz, CDCl3) δ: 7.84 (s, 1H), 7.37~7.33 (m, 3H), 7.22~7.19 (m, 2H), 7.16~7.10 (m, 3H), 7.04 (d, J=6.6 Hz, 2H), 4.22 (t, J=5.7 Hz, 2H), 3.47 (t, J=6.3 Hz, 2H), 1.84~1.74 (m, 4H); 13C NMR (75 MHz, CDCl3) δ: 167.6, 140.3, 135.8, 134.4, 132.4, 130.5, 129.5, 129.0, 128.5, 128.1, 127.7, 64.2, 44.3, 29.1, 25.9; HRMS (ESI) calcd for C19H20ClO2 [M+H]+ 315.1146, found 315.1144.
Methylene (2E,2'E)-bis(2,3-diphenylacrylate) (3p): White solid (40.1 mg, 87% yield). m.p. 155~156 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.83 (s, 2H), 7.30~7.28 (m, 6H), 7.17~7.06 (m, 10H), 6.98 (d, J=6.9 Hz, 4H), 5.94 (s, 2H); 13C NMR (75 MHz, CDCl3) δ: 166.3, 141.9, 135.1, 134.3, 131.4, 130.8, 129.8, 129.4, 128.7, 128.2, 128.0, 80.8; HRMS (ESI) calcd for C31H25O4 [M+H]+ 461.1747, found 461.1743.
Isopropyl (E)-2,3-diphenylacrylate (3q): White solid (47.4 mg, 89% yield). m.p. 57~58 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.80 (s, 1H), 7.35~7.32 (m, 3H), 7.22~7.19 (m, 2H), 7.17~7.09 (m, 3H), 7.03 (d, J=6.9 Hz, 2H), 5.18~5.09 (m, 1H), 1.27 (d, J=6.3 Hz, 6H); 13C NMR (75 MHz, CDCl3) δ: 167.2, 139.6, 135.9, 134.7, 133.2, 130.4, 129.7, 128.7, 128.4, 128.1, 127.6, 68.5, 21.8; HRMS (ESI) calcd for C18H19O2 [M+H]+ 267.1380, found 267.1378.
sec-Butyl (E)-2,3-diphenylacrylate (3r): Yellow oil (49.3 mg, 88% yield). 1H NMR (300 MHz, CDCl3) δ: 7.82 (s, 1H), 7.35~7.30 (m, 3H), 7.22~7.19 (m, 2H), 7.16~7.09 (m, 3H), 7.04 (d, J=6.9 Hz, 2H), 5.02~4.92 (m, 1H), 1.63~1.52 (m, 2H), 1.24 (d, J=6.3 Hz, 3H), 0.87 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 167.3, 139.6, 136.0, 134.7, 133.2, 130.4, 129.7, 128.7, 128.4, 128.0, 127.5, 73.0, 28.7, 19.3, 9.6; HRMS (ESI) calcd for C19H21O2 [M+H]+ 281.1536, found 281.1534.
1-Phenylethyl (E)-2,3-diphenylacrylate (3s): Yellow oil (55.8 mg, 85% yield). 1H NMR (300 MHz, CDCl3) δ: 7.87 (s, 1H), 7.33~7.19 (m, 10H), 7.13~7.06 (m, 3H), 7.03 (d, J=7.5 Hz, 2H), 6.01 (q, J=6.6 Hz, 1H), 1.52 (d, J=6.6 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 166.7, 141.7, 140.1, 135.8, 134.5, 132.7, 130.4, 129.6, 128.8, 128.4, 128.3, 128.0, 127.6, 127.5, 125.7, 72.9, 22.5; HRMS (ESI) calcd for C23H21O2 [M+H]+ 329.1536, found 329.1532.
Pentan-3-yl (E)-2,3-diphenylacrylate (3t): Yellow oil (47.7 mg, 81% yield). 1H NMR (300 MHz, CDCl3) δ: 7.83 (s, 1H), 7.36~7.32 (m, 3H), 7.23~7.19 (m, 2H), 7.17~7.09 (m, 3H), 7.04 (d, J=7.8 Hz, 2H), 4.94~4.88 (m, 1H), 1.63~1.53 (m, 4H), 0.87 (t, J=7.5 Hz, 6H); 13C NMR (75 MHz, CDCl3) δ: 167.6, 139.5, 136.1, 134.7, 133.2, 130.5, 129.6, 128.8, 128.4, 128.1, 127.5, 77.4, 26.3, 9.5; HRMS (ESI) calcd for C20H23O2 [M+H]+ 295.1693, found 295.1686.
Oxetan-3-yl (E)-2,3-diphenylacrylate (3u): White solid (25.2 mg, 45% yield). m.p. 86~87 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.89 (s, 1H), 7.32~7.30 (m, 3H), 7.22~7.20 (m, 2H), 7.15~7.08 (m, 3H), 7.04 (d, J=6.9 Hz, 2H), 5.51~5.44 (m, 1H), 4.84 (t, J=6.9 Hz, 2H), 4.62 (t, J=6.6 Hz, 2H); 13C NMR (75 MHz, CDCl3) δ: 166.5, 141.0, 135.0, 134.0, 131.3, 130.4, 129.4, 129.0, 128.4, 127.9, 127.7, 77.1, 68.2; HRMS (ESI) calcd for C18H17O3 [M+H]+ 281.1172, found 281.1169.
Cyclopentyl (E)-2,3-diphenylacrylate (3v): White solid (48.0 mg, 82% yield). m.p. 60~61 ℃; 1H NMR (300 MHz, CDCl3) δ: 7.78 (s, 1H), 7.35~7.31 (m, 3H), 7.21~7.10 (m, 5H), 7.04 (d, J=7.2 Hz, 2H), 5.31~5.26 (m, 1H), 1.92~1.83 (m, 2H), 1.74~1.56 (m, 6H); 13C NMR (75 MHz, CDCl3) δ: 167.4, 139.6, 135.9, 134.7, 133.2, 130.4, 129.7, 128.8, 128.3, 128.1, 127.5, 77.8, 32.6, 23.6; HRMS (ESI) calcd for C20H21O2 [M+H]+ 293.1536, found 293.1534.
tert-Butyl (E)-2,3-diphenylacrylate (3w): Yellow oil (24.1 mg, 43% yield). 1H NMR (300 MHz, CDCl3) δ: 7.73 (s, 1H), 7.38~7.32 (m, 3H), 7.21~7.12 (m, 5H), 7.03 (d, J=6.6 Hz, 2H), 1.50 (s, 9H); 13C NMR (75 MHz, CDCl3) δ: 166.9, 139.0, 136.2, 134.9, 134.3, 130.4, 129.8, 128.7, 128.4, 128.1, 127.5, 81.0, 28.1; HRMS (ESI) calcd for C19H21O2 [M+H]+ 281.1536, found 281.1532.
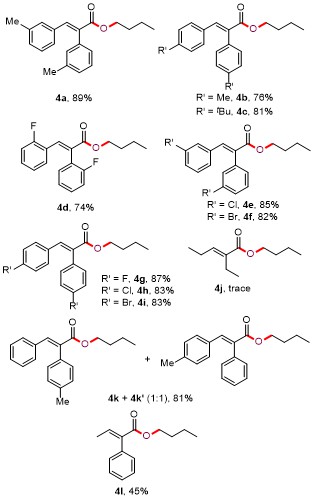
Butyl (E)-2,3-di-m-tolylacrylate (4a): Yellow oil (54.9 mg, 89% yield). 1H NMR (300 MHz, CDCl3) δ: 7.77 (s, 1H), 7.24 (t, J=7.5 Hz, 1H), 7.14 (d, J=7.2 Hz, 1H), 7.03~6.98 (m, 4H), 6.90 (s, 1H), 6.81 (s, 1H), 4.20 (t, J=6.6 Hz, 2H), 2.33 (s, 3H), 2.19 (s, 3H), 1.69~1.60 (m, 2H), 1.43~1.31 (m, 2H), 0.92 (t, J=7.2 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 168.0, 139.9, 138.0, 137.6, 135.9, 134.6, 132.7, 131.6, 130.2, 129.7, 128.4, 128.3, 127.9, 127.5, 126.7, 64.9, 30.6, 21.4, 21.2, 19.2, 13.7; HRMS (ESI) calcd for C21H25O2 [M+H]+ 309.1849, found 309.1846.
Butyl (E)-2,3-di-p-tolylacrylate (4b): Yellow oil (46.9 mg, 76% yield). 1H NMR (300 MHz, CDCl3) δ: 7.78 (s, 1H), 7.17~7.08 (m, 4H), 6.96 (s, 4H), 4.19 (t, J=6.9 Hz, 2H), 2.38 (s, 3H), 2.26 (s, 3H), 1.68~1.59 (m, 2H), 1.38~1.32 (m, 2H), 0.92 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 168.1, 139.8, 139.1, 137.2, 133.1, 132.0, 131.8, 130.5, 129.6, 129.2, 128.9, 64.9, 30.7, 21.33, 21.28, 19.1, 13.7; HRMS (ESI) calcd for C21H25O2 [M+H]+ 309.1849, found 309.1845.
Butyl (E)-2,3-bis(4-(tert-butyl)phenyl)acrylate (4c): Yellow oil (63.6 mg, 81% yield). 1H NMR (300 MHz, CDCl3) δ: 7.77 (s, 1H), 7.39 (d, J=8.4 Hz, 2H), 7.18~7.14 (m, 4H), 7.01 (d, J=8.4 Hz, 2H), 4.19 (t, J=6.6 Hz, 2H), 1.68~1.59 (m, 2H), 1.36 (s, 9H), 1.34~1.32 (m, 2H), 1.25 (s, 9H), 0.90 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 168.2, 152.3, 150.5, 139.6, 133.1, 131.9, 131.7, 130.5, 129.2, 125.4, 125.1, 64.9, 34.7, 34.6, 31.4, 31.1, 30.7, 19.2, 13.7; HRMS (ESI) calcd for C27H37O2 [M+H]+ 393.2788, found 393.2783.
Butyl (E)-2,3-bis(2-fluorophenyl)acrylate (4d): Yellow oil (46.8 mg, 74% yield). 1H NMR (300 MHz, CDCl3) δ: 8.01 (s, 1H), 7.20~7.13 (m, 1H), 7.07~7.00 (m, 1H), 6.97~6.94 (m, 2H), 6.90 (d, J=7.5 Hz, 1H), 6.84 (d, J=8.7 Hz, 1H), 6.70~6.62 (m, 2H), 4.09 (t, J=6.6 Hz, 2H), 1.54~1.45 (m, 2H), 1.28~1.16 (m, 2H), 0.77 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 166.3, 160.9 (d, 1JC-F=250.5 Hz), 160.1 (d, 1JC-F=245.3 Hz), 133.7 (d, 3JC-F=5.3 Hz), 131.3 (d, 4JC-F=3.0 Hz), 130.8 (d, 3JC-F=9.0 Hz), 129.8 (d, 2JC-F=27.8 Hz), 129.7 (d, 2JC-F=21.0 Hz), 128.8, 124.0 (d, 4JC-F=3.8 Hz), 123.5 (d, 4JC-F=3.8 Hz), 123.4 (d, 3JC-F=16.5 Hz), 122.4 (d, 3JC-F=12.0 Hz), 115.5 (d, 2JC-F=21.8 Hz), 115.4 (d, 2JC-F=21.8 Hz), 65.0, 30.4, 18.9, 13.4; HRMS (ESI) calcd for C19H19F2O2 [M+H]+ 317.1348, found 317.1344.
Butyl (E)-2,3-bis(3-chlorophenyl)acrylate (4e): Yellow oil (59.4 mg, 85% yield). 1H NMR (300 MHz, CDCl3) δ: 7.77 (s, 1H), 7.31~7.23 (m, 4H), 7.14 (d, J=8.1 Hz, 1H), 7.06 (d, J=6.9 Hz, 2H), 6.87 (d, J=7.8 Hz, 1H), 4.20 (t, J=6.6 Hz, 2H), 1.67~1.58 (m, 2H), 1.41~1.29 (m, 2H), 0.90 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 166.5, 138.9, 136.9, 135.8, 134.2, 134.0, 132.6, 130.2, 129.6, 129.5, 129.3, 128.9, 128.1, 127.9, 127.7, 65.1, 30.4, 18.9, 13.5; HRMS (ESI) calcd for C19H19Cl2O2 [M+H]+ 349.0757, found 349.0755.
Butyl (E)-2,3-bis(3-bromophenyl)acrylate (4f): Yellow oil (71.9 mg, 82% yield). 1H NMR (300 MHz, CDCl3) δ: 7.75 (s, 1H), 7.47 (d, J=7.5 Hz, 1H), 7.38 (s, 1H), 7.32 (d, J=7.2 Hz, 1H), 7.23 (s, 1H), 7.20 (d, J=7.8 Hz, 1H), 7.11 (d, J=7.5 Hz, 1H), 7.00 (t, J=7.8 Hz, 1H), 6.91 (d, J=7.5 Hz, 1H), 4.21 (t, J=6.6 Hz, 2H), 1.68~1.59 (m, 2H), 1.42~1.30 (m, 2H), 0.91 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 166.6, 138.9, 137.2, 136.1, 133.3, 132.6, 132.4, 131.9, 131.0, 130.0, 129.6, 128.5, 128.2, 122.4, 122.2, 65.2, 30.4, 19.0, 13.6; HRMS (ESI) calcd for C19H19Br2O2 [M+H]+ 436.9746, found 436.9746.
Butyl (E)-2,3-bis(4-fluorophenyl)acrylate (4g): Yellow oil (55.1 mg, 87% yield). 1H NMR (300 MHz, CDCl3) δ: 7.81 (s, 1H), 7.19~7.15 (m, 2H), 7.05~7.00 (m, 2H), 6.81 (t, J=8.7 Hz, 2H), 4.20 (t, J=6.6 Hz, 2H), 1.67~1.58 (m, 2H), 1.41~1.29 (m, 2H), 0.90 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 167.5, 162.8 (d, 1JC-F=249.8 Hz), 162.4 (d, 1JC-F=245.3 Hz), 139.2, 132.4 (d, 3JC-F=8.3 Hz), 131.55, 131.53 (d, 3JC-F=7.5 Hz), 130.7 (d, 4JC-F=3.0 Hz), 115.7 (d, 2JC-F=24.0 Hz), 115.4 (d, 2JC-F=24.8 Hz), 65.2, 30.6, 19.2, 13.7; HRMS (ESI) calcd for C19H19F2O2 [M+H]+ 317.1348, found 317.1342.
Butyl (E)-2,3-bis(4-chlorophenyl)acrylate (4h): Yellow oil (57.9 mg, 83% yield). 1H NMR (300 MHz, CDCl3) δ: 7.68 (s, 1H), 7.21 (d, J=8.4 Hz, 2H), 7.03~7.00 (m, 4H), 6.85 (t, J=8.4 Hz, 2H), 4.09 (t, J=6.6 Hz, 2H), 1.57~1.47 (m, 2H), 1.30~1.20 (m, 2H), 0.80 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 166.9, 139.0, 134.9, 133.8, 133.7, 132.7, 132.0, 131.5, 131.0, 128.7, 128.4, 65.1, 30.4, 19.0, 13.5; HRMS (ESI) calcd for C19H19Cl2O2 [M+H]+ 349.0757, found 349.0755.
Butyl (E)-2,3-bis(4-bromophenyl)acrylate (4i): Yellow oil (72.7 mg, 83% yield). 1H NMR (300 MHz, CDCl3) δ: 7.76 (s, 1H), 7.49 (d, J=8.4 Hz, 2H), 7.32 (d, J=8.4 Hz, 2H), 7.07 (d, J=8.4 Hz, 2H), 6.91 (d, J=8.4 Hz, 2H), 4.20 (t, J=6.6 Hz, 2H), 1.68~1.59 (m, 2H), 1.42~1.29 (m, 2H), 0.92 (t, J=7.5 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 167.1, 139.2, 134.4, 133.2, 132.3, 131.84, 131.82, 131.6, 131.4, 123.5, 122.2, 65.3, 30.6, 19.1, 13.7; HRMS (ESI) calcd for C19H19Br2O2 [M+H]+ 436.9746, found 436.9747.
Butyl (E)-3-phenyl-2-(p-tolyl)acrylate and butyl (E)-2- phenyl-3-(p-tolyl)acrylate (4k+4k'): Yellow oil (47.7 mg, 81% yield). 1H NMR (300 MHz, CDCl3) δ: 7.80 (s, 1H), 7.79 (s, 1H), 7.36~7.33 (m, 2H), 7.25~7.06 (m, 12H), 6.97~6.91 (m, 4H), 4.22~4.17 (m, 4H), 2.37 (s, 3H), 2.26 (s, 3H), 1.67~1.60 (m, 4H), 1.43~1.33 (m, 4H), 0.95~0.88 (m, 6H); 13C NMR (75 MHz, CDCl3) δ: 168.03, 167.96, 140.1, 139.7, 139.2, 137.4, 136.2, 134.9, 132.84, 132.81, 131.8, 130.6, 130.5, 129.7, 129.6, 129.2, 128.9, 128.8, 128.5, 128.1, 127.6, 65.0, 64.9, 30.7, 21.32, 21.28, 19.1, 13.7; HRMS (ESI) calcd for C20H23O2 [M+H]+ 295.1693, found 295.1692.
Butyl (E)-2-phenylbut-2-enoate (4l): Yellow oil (19.7 mg, 45% yield). 1H NMR (300 MHz, CDCl3) δ: 7.69 (s, 1H), 7.40~7.31 (m, 5H), 4.22 (t, J=6.6 Hz, 2H), 2.12 (d, J=0.9 Hz, 3H), 1.76~1.66 (m, 2H), 1.49~1.41 (m, 2H), 0.97 (t, J=7.2 Hz, 3H); 13C NMR (75 MHz, CDCl3) δ: 168.7, 138.6, 136.0, 129.6, 128.7, 128.3, 128.2, 64.8, 30.8, 19.3, 14.0, 13.8; HRMS (ESI) calcd for C14H19O2 [M+H]+ 219.1380, found 219.1376.