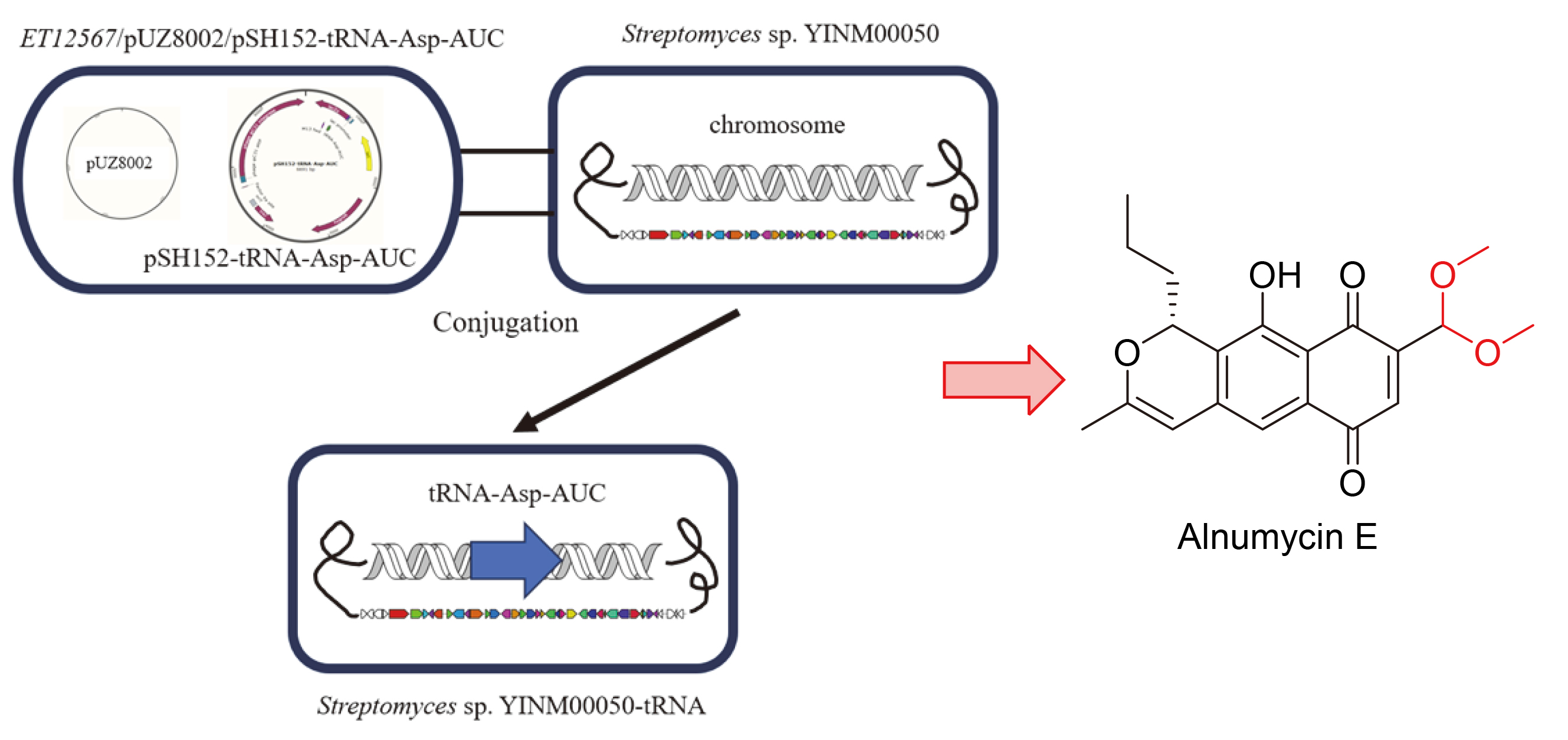
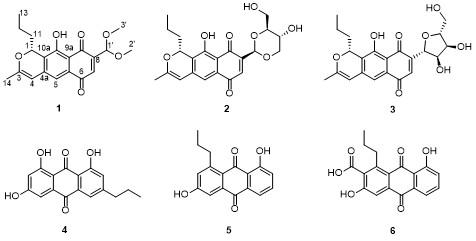
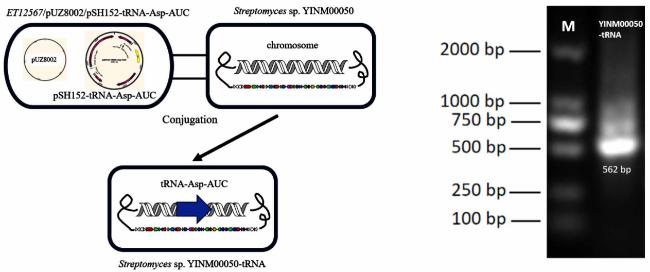
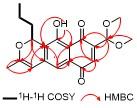
1 结果与讨论
表1 化合物1的1H NMR (600 MHz)和13C NMR (151 MHz)核磁数据(CD3OD)Table 1 1H NMR (600 MHz) and 13C NMR (151 MHz) data of compound 1 in CD3OD |
| Position | δC, type | δH, mult (J in Hz) |
|---|---|---|
| 1 | 73.0, CH | 5.58, dd (9.3, 3.5) |
| 3 | 158.5, C | — |
| 4 | 100.0, CH | 5.60, s |
| 4a | 139.6, C | — |
| 5 | 114.3, CH | 7.13, s |
| 5a | 131.4, C | — |
| 6 | 184.8, C | — |
| 7 | 135.2, CH | 6.99, s |
| 8 | 144.8, C | — |
| 9 | 187.8, C | — |
| 9a | 113.0, C | — |
| 10 | 157.1, C | — |
| 10a | 122.4, C | — |
| 11 | 34.9, CH2 | 1.96~2.02, m, overlapped 1.48~1.55, m, overlapped |
| 12 | 18.1, CH2 | 1.53~1.60, m, overlapped 1.45~1.52, m, overlapped |
| 13 | 13.8, CH3 | 0.96, t (7.1) |
| 14 | 20.4, CH3 | 1.96, s |
| 1' | 97.3, CH | 5.52, s |
| 2' | 54.2, CH3 | 3.44, s |
| 3' | 54.3, CH3 | 3.43, s |
| OH-10 | — | 12.25, s |
表2 化合物1的抗肿瘤活性(IC50±SD, μmol/L)Table 2 Anti-tumor activity of compound 1 (IC50±SD, μmol/L) |
| Compound | HL-60 | A549 | SMMC-7721 | MCF-7 | SW480 | BEAS-2B |
|---|---|---|---|---|---|---|
| 1 | 0.26±0.01 | 4.61±0.04 | 0.53±0.02 | 1.94±0.11 | 1.72±0.11 | >40 |
| DDP | 2.97±0.14 | 12.55±0.60 | 9.60±0.67 | 21.03±0.71 | 35.63±1.16 | >40 |
| Taxol | <0.008 | <0.008 | 0.28±0.02 | <0.008 | <0.008 | 0.46±0.01 |