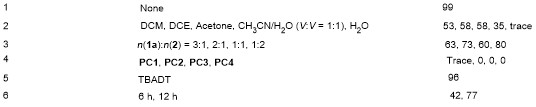
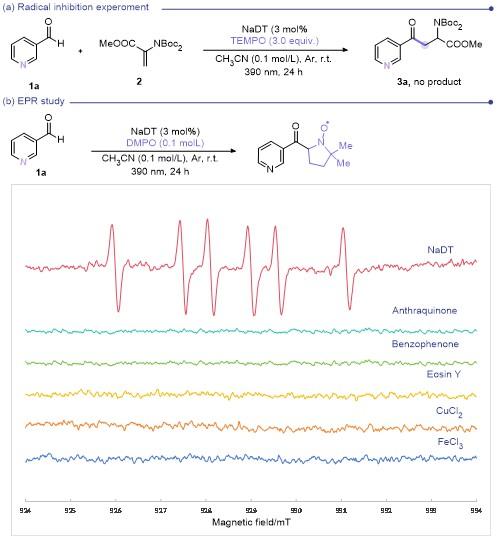
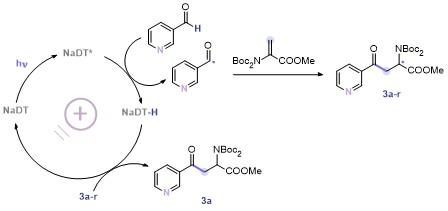
In a glovebox, 3-pyridinecarboxaldehyde (18.8 µL, 0.2 mmol), photocatalyst (3%) and DMPO (11.3 µL, 0.1 mmol) were added to a 5 mL round bottomed bottle with a stir bar, then CH3CN (1 mL) was added. After the mixture was stirred for 2 min under dark, 20 µL of the mixture was transferred to a capillary. The capillary was transferred to a ESR tube, the ESR tube was then adapted to the ESR instrument measure capacity, and measured in-situ.
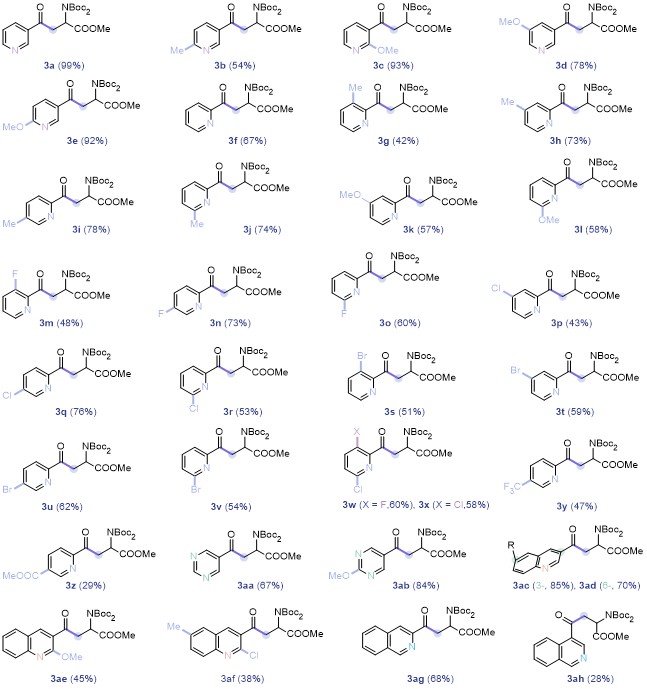
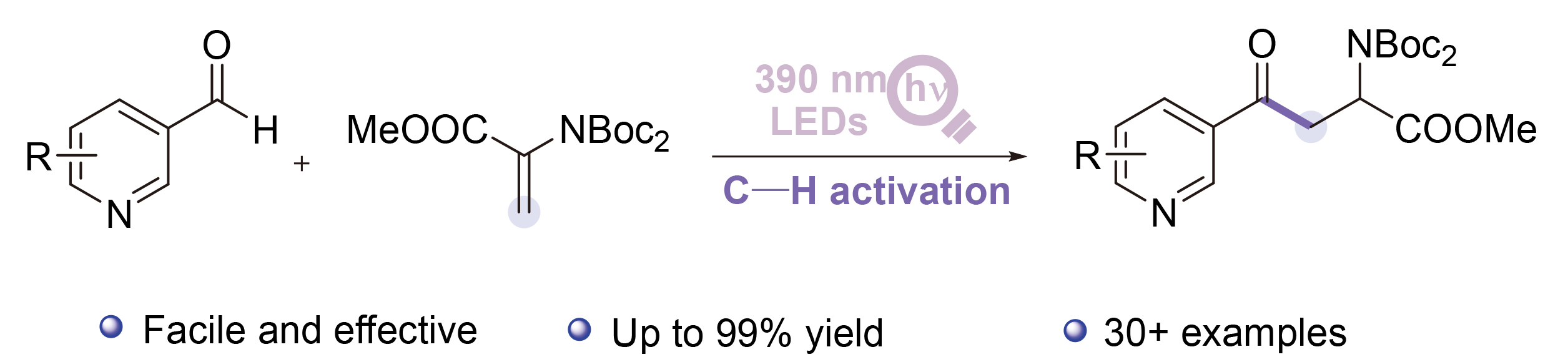
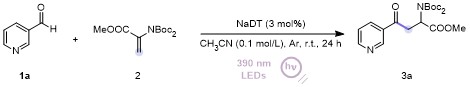
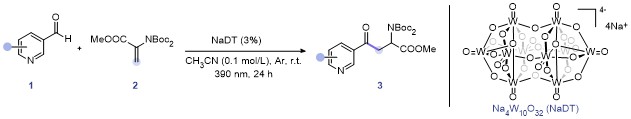
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-oxo-4-(py-ridin-3-yl)-butanoate (3a): 99% yield, faint yellow solid, m.p. 93.0~93.8 °C; 1H NMR (500 MHz, Chloroform-d) δ: 9.22~9.17 (m, 1H), 8.82~8.78 (m, 1H), 8.27 (dt, J=7.9, 2.0 Hz, 1H), 7.45 (dd, J=7.9, 4.7 Hz, 1H), 5.74 (dd, J=7.2, 5.3 Hz, 1H), 4.07 (dd, J=17.4, 7.2 Hz, 1H), 3.73 (s, 3H), 3.24 (dd, J=17.4, 5.4 Hz, 1H), 1.52 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 195.48, 170.58, 153.45, 151.86, 149.51, 135.68, 132.01, 123.71, 83.63, 54.41, 52.59, 40.06, 27.99. HRMS (ESI+) calcd for C20H28- N2O7Na (M+Na)+ 431.1789, found 431.1792.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-meth-yl- pyridin-3-yl)-4-oxobutanoate (3b): 54% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 9.08 (s, 1H), 8.16 (dd, J=8.1, 2.3 Hz, 1H), 7.28 (d, J=8.3 Hz, 1H), 5.74 (dd, J=7.2, 5.4 Hz, 1H), 4.04 (dd, J=17.4, 7.2 Hz, 1H), 3.72 (s, 3H), 3.22 (dd, J=17.4, 5.5 Hz, 1H), 2.65 (s, 3H), 1.52 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 195.19, 170.66, 163.33, 151.85, 149.10, 136.10, 129.63, 123.41, 83.60, 54.42, 52.58, 39.95, 28.01, 24.68. HRMS (ESI+) calcd for C21H30N2O7Na (M+Na)+ 445.1945, found 445.1949.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(2-metho-xypyridin-3-yl)-4-oxobutanoate (3c): 93% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.32 (dd, J=4.9, 2.0 Hz, 1H), 8.14 (dd, J=7.5, 2.1 Hz, 1H), 6.99 (dd, J=7.5, 4.8 Hz, 1H), 5.74 (dd, J=7.1, 5.4 Hz, 1H), 4.08~4.03 (m, 4H), 3.72 (s, 3H), 3.32 (dd, J=18.1, 5.3 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 196.65, 170.89, 161.99, 151.73, 150.93, 140.23, 121.07, 117.19, 83.30, 54.32, 53.81, 52.43, 44.76, 27.98. HRMS (ESI+) calcd for C21H30N2O8Na (M+Na)+ 461.1894, found 461.1889.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(5-metho-xypyridin-3-yl)-4-oxobutanoate (3d): 78% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.82-8.77 (m, 1H), 8.50 (d, J=2.9 Hz, 1H), 7.73 (dd, J=2.9, 1.7 Hz, 1H), 5.74 (dd, J=7.2, 5.4 Hz, 1H), 4.06 (dd, J=17.5, 7.2 Hz, 1H), 3.91 (s, 3H), 3.73 (s, 3H), 3.25 (dd, J=17.5, 5.4 Hz, 1H), 1.52 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 195.44, 170.60, 155.98, 151.84, 142.95, 141.77, 132.61, 118.07, 83.64, 55.78, 54.39, 52.61, 40.35, 28.01. HRMS (ESI+) calcd for C21H31N2O8 (M+H)+ 439.2075, found 439.2077.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-metho-xypyridin-3-yl)-4-oxobutanoate (3e): 92% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.81 (d, J=2.4 Hz, 1H), 8.16 (dd, J=8.8, 2.5 Hz, 1H), 6.79 (d, J=8.7 Hz, 1H), 5.74 (dd, J=7.0, 5.5 Hz, 1H), 4.01 (s, 3H), 4.00~3.95 (m, 1H), 3.72 (s, 3H), 3.19 (dd, J=17.2, 5.5 Hz, 1H), 1.52 (s, 18H); 13C NMR (126 MHz, Chloroform- d) δ: 194.23, 170.72, 166.86, 151.83, 149.07, 138.25, 126.47, 111.17, 83.48, 54.43, 54.08, 52.52, 39.62, 27.99. HRMS (ESI+) calcd for C21H30N2O8Na (M+Na)+ 461.1894, found 461.1892.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(2-brom-opyridin-3-yl)-4-oxobutanoate (3f): 53% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.45 (dd, J=4.8, 2.0 Hz, 1H), 7.84 (dd, J=7.6, 2.0 Hz, 1H), 7.37 (dd, J=7.6, 4.8 Hz, 1H), 5.70 (dd, J=8.0, 5.0 Hz, 1H), 3.93 (dd, J=17.3, 8.1 Hz, 1H), 3.73 (s, 3H), 3.29 (dd, J=17.3, 5.1 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 199.08, 170.61, 151.71, 151.37, 138.52, 137.76, 137.54, 122.77, 83.74, 54.90, 52.64, 43.73, 28.00. HRMS (ESI+) calcd for C20H27BrN2O7Na (M+Na)+ 509.0894, found 509.0899.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-oxo-4-(py-ridin-2-yl)butanoate (3g): 67% yield, faint yellow solid, m.p. 82.6~83.1 °C; 1H NMR (500 MHz, Chloroform-d) δ: 8.68 (dd, J=4.7, 1.6 Hz, 1H), 8.05 (d, J=7.5 Hz, 1H), 7.83 (td, J=7.7, 1.8 Hz, 1H), 7.47 (ddd, J=7.6, 4.7, 1.2 Hz, 1H), 5.72 (dd, J=7.4, 5.7 Hz, 1H), 4.34 (dd, J=17.5, 7.3 Hz, 1H), 3.72 (s, 3H), 3.44 (dd, J=17.6, 5.7 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 198.32, 170.74, 153.03, 151.65, 148.99, 136.83, 127.21, 121.93, 83.32, 54.74, 52.41, 39.19, 27.98. HRMS (ESI+) calcd for C20H28N2O7Na (M+Na)+ 431.1789, found 431.1794.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(3-methyl-pyridin-2-yl)-4-oxobutanoate (3h): 42% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.50 (dd, J=4.8, 1.6 Hz, 1H), 7.59~7.57 (m, 1H), 7.33 (dd, J=7.8, 4.6 Hz, 1H), 5.69 (dd, J=7.6, 5.6 Hz, 1H), 4.21 (dd, J=17.4, 7.5 Hz, 1H), 3.72 (s, 3H), 3.46 (dd, J=17.4, 5.6 Hz, 1H), 2.58 (s, 3H), 1.49 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 200.25, 170.86, 151.69, 151.49, 146.18, 140.04, 134.76, 126.12, 83.26, 54.88, 52.37, 41.19, 27.97, 20.17. HRMS (ESI+) calcd for C21H30N2O7Na (M+Na)+ 445.1945, found 445.1938.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(4-methyl-pyridin-2-yl)-4-oxobutanoate (3i): 73% yield, yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.54 (d, J=4.9 Hz, 1H), 7.89~7.86 (m, 1H), 7.29 (d, J=3.6 Hz, 1H), 5.71 (dd, J=7.4, 5.6 Hz, 1H), 4.31 (dd, J=17.6, 7.4 Hz, 1H), 3.71 (s, 3H), 3.42 (dd, J=17.6, 5.6 Hz, 1H), 2.42 (s, 3H), 1.50 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 198.56, 170.74, 152.82, 151.64, 148.85, 148.23, 128.08, 122.82, 83.29, 54.76, 52.39, 39.29, 27.97, 21.07. HRMS (ESI+) calcd for C21H31N2O7 (M+H)+ 423.2126, found 423.2131.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(5-methyl-pyridin-2-yl)-4-oxobutanoate (3j): 78% yield, yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.50 (d, J=2.1 Hz, 1H), 7.96 (d, J=8.0 Hz, 1H), 7.62 (dd, J=8.3, 2.0 Hz, 1H), 5.71 (dd, J=7.3, 5.7 Hz, 1H), 4.30 (dd, J=17.5, 7.3 Hz, 1H), 3.71 (s, 3H), 3.42 (dd, J=17.5, 5.7 Hz, 1H), 2.41 (s, 3H), 1.50 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 198.08, 170.75, 151.63, 150.73, 149.48, 137.70, 137.19, 121.70, 83.26, 54.77, 52.37, 39.11, 27.96, 18.73. HRMS (ESI+) calcd for C21H31N2O7 (M+H)+ 423.2126, found 423.2123.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-methyl-pyridin-2-yl)-4-oxobutanoate (3k): 73% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 7.85 (d, J=7.7 Hz, 1H), 7.70 (t, J=7.7 Hz, 1H), 7.32 (d, J=7.6 Hz, 1H), 5.73 (dd, J=7.2, 5.7 Hz, 1H), 4.35 (dd, J=17.6, 7.2 Hz, 1H), 3.72 (s, 3H), 3.44 (dd, J=17.6, 5.7 Hz, 1H), 2.60 (s, 3H), 1.51 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 198.60, 170.87, 157.98, 152.48, 151.65, 136.89, 126.84, 118.99, 83.27, 54.80, 52.39, 39.07, 27.96, 24.38. HRMS (ESI+) calcd for C21H30N2O7 (M H)+ 423.2126, found 423.2123.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(4-methox-ypyridin-2-yl)-4-oxobutanoate (3l): 57% yield, yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.48 (d, J=5.6 Hz, 1H), 7.58 (d, J=2.6 Hz, 1H), 6.98 (dd, J=5.6, 2.6 Hz, 1H), 5.71 (dd, J=7.5, 5.6 Hz, 1H), 4.32 (dd, J=17.6, 7.5 Hz, 1H), 3.90 (s, 3H), 3.72 (s, 3H), 3.41 (dd, J=17.7, 5.6 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, Chloroform-d) δ: 198.26, 170.71, 166.51, 154.82, 151.64, 150.15, 114.04, 107.07, 83.32, 55.48, 54.74, 52.40, 39.34, 27.97. HRMS (ESI+) calcd for C21H31N2O8 (M+H)+ 439.2075, found 439.2080.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-methox- ypyridin-2-yl)-4-oxobutanoate (3m): 58% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 7.72~7.65 (m, 2H), 6.94 (dd, J=8.1, 1.1 Hz, 1H), 5.76 (dd, J=6.9, 5.8 Hz, 1H), 4.31 (dd, J=17.9, 6.9 Hz, 1H), 3.99 (s, 3H), 3.73 (s, 3H), 3.40 (dd, J=17.9, 5.8 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.77, 170.90, 163.29, 151.66, 150.33, 139.13, 115.56, 115.24, 83.28, 54.46, 53.40, 52.44, 39.56, 27.97. HRMS (ESI+) calcd for C21- H30- N2O8Na (M+Na)+ 461.1894, found 461.1895.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(3-fluoro-pyridin-2-yl)-4-oxobutanoate (3n): 48% yield, white solid, m.p. 98.8~99.3 °C; 1H NMR (500 MHz, Chloroform-d) δ: 8.50 (d, J=4.1 Hz, 1H), 7.55~7.50 (m, 2H), 5.70 (dd, J=7.4, 5.7 Hz, 1H), 4.26 (dd, J=17.4, 7.4 Hz, 1H), 3.72 (s, 3H), 3.38 (dd, J=17.4, 5.7 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 196.02, 170.63, 159.69, 157.51, 151.61, 144.67, 144.63, 141.39, 128.67, 128.64, 125.67, 125.51, 83.40, 54.62, 52.47, 40.88, 27.97; 19F NMR (471 MHz, CDCl3) δ: -130.60. HRMS (ESI+) calcd for C20H27FN2O7Na (M+Na)+ 449.1695, found 449.1692.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(5-fluoro- pyridin-2-yl)-4-oxobutanoate (3o): 73% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.50 (d, J=2.7 Hz, 1H), 8.12 (dd, J=8.7, 4.6 Hz, 1H), 7.51 (td, J=8.3, 2.8 Hz, 1H), 5.72~5.68 (m, 1H), 4.32 (dd, J=17.5, 7.4 Hz, 1H), 3.72 (s, 3H), 3.38 (dd, J=17.5, 5.7 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 196.68, 170.69, 162.58, 160.48, 151.65, 149.59, 149.56, 137.50, 137.30, 123.98, 123.94, 123.51, 123.36, 83.37, 54.70, 52.43, 39.08, 27.97; 19F NMR (471 MHz, CDCl3) δ: -119.76. HRMS (ESI+) calcd for C20H28FN2O7 (M+H)+ 427.1875, found 427.1880.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-fluoro- pyridin-2-yl)-4-oxobutanoate (3p): 60% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 7.96~7.93 (m, 2H), 7.17~7.13 (m, 1H), 5.69 (dd, J=7.6, 5.4 Hz, 1H), 4.28 (dd, J=17.8, 7.6 Hz, 1H), 3.72 (s, 3H), 3.33 (dd, J=17.8, 5.5 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 196.74, 170.61, 163.48, 161.56, 151.61, 151.23, 151.14, 141.96, 141.90, 119.45, 119.42, 114.12, 113.83, 83.47, 54.59, 52.45, 39.07, 27.97; 19F NMR (471 MHz, CDCl3) δ: -66.40. HRMS (ESI+) calcd for C20H27- FN2O7Na (M+Na)+ 449.1695, found 449.1693.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(4-chloro-pyridin-2-yl)-4-oxobutanoate (3q): 43% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.57 (d, J=5.2 Hz, 1H), 8.04 (d, J=2.0 Hz, 1H), 7.47 (dd, J=5.2, 2.1 Hz, 1H), 5.70 (dd, J=7.4, 5.7 Hz, 1H), 4.33 (dd, J=17.6, 7.4 Hz, 1H), 3.72 (s, 3H), 3.38 (dd, J=17.6, 5.7 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.24, 170.63, 154.19, 151.65, 149.95, 145.43, 127.26, 122.44, 83.44, 54.71, 52.47, 39.23, 27.98; HRMS (ESI+) calcd for C20H27ClN2O7Na (M+Na)+ 465.1399, found 465.1401.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(5-chloro-pyridin-2-yl)-4-oxobutanoate (3r): 76% yield, yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.62 (d, J=2.4 Hz, 1H), 8.01 (d, J=8.4 Hz, 1H), 7.80 (dd, J=8.4, 2.4 Hz, 1H), 5.70 (dd, J=7.4, 5.7 Hz, 1H), 4.31 (dd, J=17.5, 7.3 Hz, 1H), 3.72 (s, 3H), 3.37 (dd, J=17.5, 5.7 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.21, 170.66, 151.65, 150.95, 147.98, 136.64, 136.16, 122.90, 83.41, 54.70, 52.45, 39.06, 27.97. HRMS (ESI+) calcd for C20H27ClN2O7Na (M+Na)+ 465.1399, found 465.1394.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-chloro-pyridin-2-yl)-4-oxobutanoate (3s): 53% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 7.97 (dd, J=7.6, 0.9 Hz, 1H), 7.80 (t, J=7.7 Hz, 1H), 7.51 (dd, J=7.9, 0.9 Hz, 1H), 5.70 (dd, J=7.7, 5.4 Hz, 1H), 4.34 (dd, J=17.8, 7.7 Hz, 1H), 3.72 (s, 3H), 3.35 (dd, J=17.8, 5.4 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 196.87, 170.64, 153.16, 151.61, 150.84, 139.50, 128.20, 120.44, 83.48, 54.64, 52.45, 38.98, 27.98. HRMS (ESI+) calcd for C20H27ClN2O7Na (M+Na)+ 465.1399, found 465.1401.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(3-bromo-pyridin-2-yl)-4-oxobutanoate (3t): 51% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.58 (dd, J=4.5, 1.4 Hz, 1H), 8.00 (dd, J=8.2, 1.4 Hz, 1H), 7.29 (dd, J=8.1, 4.6 Hz, 1H), 5.69 (dd, J=7.3, 5.7 Hz, 1H), 4.16 (dd, J=17.6, 7.4 Hz, 1H), 3.73 (s, 3H), 3.42 (dd, J=17.6, 5.7 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.46, 170.59, 151.75, 151.65, 147.20, 142.67, 126.77, 118.11, 83.38, 54.67, 52.51, 41.20, 27.99. HRMS (ESI+) calcd for C20H27BrN2O7Na (M+Na)+ 509.0894, found 509.0888.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(4-bromo-pyridin-2-yl)-4-oxobutanoate (3u): 59% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.49 (d, J=5.2 Hz, 1H), 8.20 (d, J=1.9 Hz, 1H), 7.64 (dd, J=5.2, 2.0 Hz, 1H), 5.69 (dd, J=7.4, 5.7 Hz, 1H), 4.32 (dd, J=17.6, 7.4 Hz, 1H), 3.72 (s, 3H), 3.38 (dd, J=17.6, 5.8 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.17, 170.61, 153.83, 151.64, 149.78, 133.99, 130.30, 125.49, 83.43, 54.69, 52.46, 39.19, 27.97. HRMS (ESI+) calcd for C20H27BrN2O7Na (M+Na)+ 509.0894, found 509.0890.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(5-bromo-pyridin-2-yl)-4-oxobutanoate (3v): 62% yield, yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.73 (dd, J=2.2, 0.8 Hz, 1H), 7.97 (dd, J=8.4, 2.2 Hz, 1H), 7.93 (dd, J=8.4, 0.8 Hz, 1H), 5.69 (dd, J=7.4, 5.7 Hz, 1H), 4.31 (dd, J=17.5, 7.3 Hz, 1H), 3.72 (s, 3H), 3.36 (dd, J=17.5, 5.7 Hz, 1H), 1.50 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.44, 170.66, 151.64, 151.28, 150.18, 139.58, 125.42, 123.21, 83.42, 54.70, 52.45, 39.03, 27.97. HRMS (ESI+) calcd for C20H27BrN2O7Na (M+Na)+ 509.0894, found 509.0890.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-bromo-pyridin-2-yl)-4-oxobutanoate (3w): 54% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.00 (dd, J=7.3, 1.2 Hz, 1H), 7.72~7.68 (m, 1H), 7.66 (dd, J=7.9, 1.2 Hz, 1H), 5.70 (dd, J=7.7, 5.4 Hz, 1H), 4.35 (dd, J=17.8, 7.7 Hz, 1H), 3.71 (s, 3H), 3.34 (dd, J=17.8, 5.4 Hz, 1H), 1.52 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 196.78, 170.67, 153.68, 151.60, 141.37, 139.18, 131.94, 120.78, 83.49, 54.64, 52.46, 38.96, 27.99. HRMS (ESI+) calcd for C20H27BrN2O7Na (M+Na)+ 509.0894, found 509.0893.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-chloro-3-fluoropyridin-2-yl)-4-oxobutanoate (3x): 60% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 7.55~7.50 (m, 2H), 5.67 (dd, J=7.8, 5.4 Hz, 1H), 4.25 (dd, J=17.5, 7.8 Hz, 1H), 3.72 (s, 3H), 3.29 (dd, J=17.5, 5.4 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 194.69, 194.66, 170.56, 158.65, 156.47, 151.57, 144.83, 144.80, 140.66, 140.61, 129.76, 129.71, 128.98, 128.81, 83.56, 54.56, 52.52, 40.49, 27.97; 19F NMR (471 MHz, CDCl3) δ: -122.91. HRMS (ESI+) calcd for C20H26ClF- N2O7 (M+K)+ 499.1044, found 499.1047.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(6-chloro-3-fluoropyridin-2-yl)-4-oxobutanoate (3y): 58% yield, faint yellow solid, m.p. 118.9~119.2 °C; 1H NMR (500 MHz, Chloroform-d) δ: 7.75 (d, J=8.4 Hz, 1H), 7.40 (d, J=8.4 Hz, 1H), 5.67 (dd, J=7.8, 5.5 Hz, 1H), 4.17 (dd, J=17.7, 7.8 Hz, 1H), 3.72 (s, 3H), 3.35 (dd, J=17.7, 5.5 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 195.57, 170.53, 151.61, 149.86, 148.26, 142.04, 129.44, 127.83, 83.53, 54.59, 52.54, 41.05, 27.99. HRMS (ESI+) calcd for C20H26Cl2N2O7Na (M+Na)+ 499.1009, found 499.1012.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-oxo-4-(5-(trifluoromethyl)pyridin-2yl)butanoate (3z): 47% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.95 (d, J=2.1 Hz, 1H), 8.17 (d, J=8.2 Hz, 1H), 8.08 (dd, J=8.3, 2.3 Hz, 1H), 5.72 (dd, J=7.5, 5.6 Hz, 1H), 4.39 (dd, J=17.6, 7.5 Hz, 1H), 3.72 (s, 3H), 3.38 (dd, J=17.6, 5.6 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.26, 170.61, 155.33, 151.66, 150.67, 146.09, 134.27, 124.70, 121.63, 83.52, 54.70, 52.50, 39.22, 27.97; 19F NMR (471 MHz, CDCl3) δ: -62.67. HRMS (ESI+) calcd for C21H27F3N2O7 (M+K)+ 499.1663, found 499.1661.
Methyl 6-(3-(bis(tert-butoxycarbonyl)amino)-4-metho-xy-4-oxobutanoyl)nicotinate (3aa): 29% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 9.27~9.25 (m, 1H), 8.42 (dd, J=8.2, 2.1 Hz, 1H), 8.11 (dd, J=8.1, 0.9 Hz, 1H), 5.71 (dd, J=7.4, 5.7 Hz, 1H), 4.37 (dd, J=17.6, 7.4 Hz, 1H), 3.99 (s, 3H), 3.72 (s, 3H), 3.42 (dd, J=17.6, 5.7 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.73, 170.64, 165.11, 155.38, 151.65, 150.25, 138.06, 128.66, 121.46, 83.46, 54.71, 52.72, 52.47, 39.36, 27.98. HRMS (ESI+) calcd for C22H30N2O9Na (M+Na)+ 489.1844, found 489.1842.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-oxo-4-(py-rimidin-5-yl)butanoate (3ab): 67% yield, faint yellow solid, m.p. 116.4~116.8 °C; H NMR (500 MHz, Chloroform-d) δ: 9.39 (s, 1H), 9.26 (s, 2H), 5.72 (dd, J=7.5, 5.2 Hz, 1H), 4.07 (dd, J=17.2, 7.5 Hz, 1H), 3.73 (s, 3H), 3.20 (dd, J=17.2, 5.3 Hz, 1H), 1.53 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 194.30, 170.42, 161.41, 156.70, 151.86, 129.47, 83.84, 54.40, 52.70, 40.11, 27.99. HRMS (ESI+) calcd for C19H28N3O7 (M+H)+ 410.1922, found 410.1925.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(2-methox-ypyrimidin-5-yl)-4-oxobutanoate (3ac): 84% yield, faint yellow solid, m.p. 112.1~112.3 °C; 1H NMR (500 MHz, Chloroform-d) δ: 9.09 (s, 2H), 5.71 (dd, J=7.3, 5.4 Hz, 1H), 4.11 (s, 3H), 3.98 (dd, J=17.2, 7.3 Hz, 1H), 3.73 (s, 3H), 3.15 (dd, J=17.2, 5.3 Hz, 1H), 1.52 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 193.08, 170.53, 167.18, 160.31, 151.85, 124.50, 83.71, 55.78, 54.34, 52.63, 39.75, 27.99. HRMS (ESI+) calcd for C20H30N3O8 (M+H)+ 440.2027, found 440.2021.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-oxo-4-(qu-inolin-3-yl)butanoate (3ad): 85% yield, yellow oil; 1H NMR (500 MHz, Chloroform-d) δ: 9.45 (d, J=2.1 Hz, 1H), 8.77 (d, J=2.1 Hz, 1H), 8.19 (d, J=8.5 Hz, 1H), 7.97 (dd, J=8.3, 1.5 Hz, 1H), 7.86 (ddd, J=8.5, 6.9, 1.4 Hz, 1H), 7.65 (ddd, J=8.1, 6.9, 1.1 Hz, 1H), 5.79 (dd, J=7.0, 5.5 Hz, 1H), 4.20 (dd, J=17.3, 7.1 Hz, 1H), 3.74 (s, 3H), 3.38 (dd, J=17.3, 5.6 Hz, 1H), 1.53 (s, 18H)l 13C NMR (126 MHz, CDCl3) δ: 195.48, 170.67, 151.99, 149.68, 148.96, 137.46, 132.19, 129.43, 129.36, 128.95, 127.68, 126.87, 83.65, 54.59, 52.63, 40.07, 28.03; HRMS (ESI+) calcd for C24H30N2O7Na (M+Na)+ 481.1945, found 481.1942.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-oxo-4-(qu-inolin-6-yl)butanoate (3ae): 70% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 9.04 (d, J=4.3 Hz, 1H), 8.50 (d, J=2.6 Hz, 1H), 8.31~8.27 (m, 2H), 8.19 (d, J=8.8 Hz, 1H), 7.50 (ddd, J=8.3, 4.2, 1.0 Hz, 1H), 5.78 (t, J=6.3 Hz, 1H), 4.20 (dd, J=17.3, 6.9 Hz, 1H), 3.74 (s, 3H), 3.41 (dd, J=17.4, 5.7 Hz, 1H), 1.53 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 195.99, 170.76, 152.70, 152.03, 150.10, 137.75, 134.51, 130.04, 129.70, 127.75, 127.52, 122.00, 83.52, 54.75, 52.57, 39.98, 28.02. HRMS (ESI+) calcd for C24H31N2O7 (M+H)+ 459.2126, found 459.2129.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(2-meth-oxyquinolin-3-yl)-4-oxobutanoate (3af): 45% yield, yellow solid, m.p. 114.2~114.5 °C; 1H NMR (500 MHz, Chloroform-d) δ: 8.55 (s, 1H), 7.84 (dd, J=21.7, 8.0 Hz, 2H), 7.70 (ddd, J=8.5, 7.0, 1.5 Hz, 1H), 7.42 (ddd, J=8.0, 6.9, 1.1 Hz, 1H), 5.78 (dd, J=7.2, 5.4 Hz, 1H), 4.17 (s, 3H), 4.11 (dd, J=18.0, 7.2 Hz, 1H), 3.73 (s, 3H), 3.40 (dd, J=18.0, 5.4 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 197.01, 170.88, 159.25, 151.73, 147.84, 141.66, 131.80, 129.13, 126.94, 124.83, 124.48, 123.08, 83.37, 54.46, 53.86, 52.46, 44.65, 27.99. HRMS (ESI+) calcd for C25H32N2O7Na (M+Na)+ 511.2051, found 511.2052.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(2-chloro-6-methylquinolin-3-yl)-4oxobutanoate (3ag): 38% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.34 (s, 1H), 7.94 (d, J=8.8 Hz, 1H), 7.65 (d, J=7.8 Hz, 2H), 5.76 (dd, J=7.9, 5.0 Hz, 1H), 4.05 (dd, J=17.4, 8.0 Hz, 1H), 3.74 (s, 3H), 3.36 (dd, J=17.4, 5.0 Hz, 1H), 2.55 (s, 3H), 1.52 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 198.08, 170.63, 151.75, 146.74, 144.70, 138.85, 138.05, 134.58, 132.91, 128.08, 127.30, 126.29, 83.69, 54.92, 52.62, 43.99, 28.00, 21.61. HRMS (ESI+) calcd for C25H31ClN2O7Na (M+Na)+ 529.1712, found 529.1717.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(isoqu-inolin-3-yl)-4-oxobutanoate (3ah): 68% yield, yellow solid, m.p. 112.5C~112.8 °C; 1H NMR (500 MHz, Chloroform-d) δ: 9.29 (s, 1H), 8.49 (s, 1H), 8.02 (dd, J=30.8, 7.6 Hz, 2H), 7.79~7.72 (m, 2H), 5.80 (dd, J=7.3, 5.6 Hz, 1H), 4.45 (dd, J=17.6, 7.3 Hz, 1H), 3.73 (s, 3H), 3.55 (dd, J=17.6, 5.7 Hz, 1H), 1.51 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 198.42, 170.82, 151.95, 151.71, 147.08, 135.51, 130.98, 130.26, 129.47, 128.63, 127.60, 120.57, 83.28, 54.84, 52.40, 39.89, 27.98. HRMS (ESI+) calcd for C24H31N2O7 (M+H)+ 459.2126, found 459.2120.
Methyl 2-(bis(tert-butoxycarbonyl)amino)-4-(isoquino-lin-4-yl)-4-oxobutanoate (3ai): 28% yield, yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 10.32 (s, 1H), 9.26 (d, J=8.5 Hz, 1H), 8.84 (s, 1H), 8.28 (d, J=8.4 Hz, 1H), 7.89 (ddd, J=8.4, 6.9, 1.2 Hz, 1H), 7.71 (ddd, J=8.4, 6.9, 1.3 Hz, 1H), 5.87 (dd, J=8.3, 5.3 Hz, 1H), 4.29 (dd, J=15.5, 5.3 Hz, 1H), 3.97 (dd, J=15.5, 8.3 Hz, 1H), 3.77 (s, 3H), 1.39 (s, 18H); 13C NMR (126 MHz, CDCl3) δ: 192.78, 170.85, 165.26, 152.17, 151.90, 132.98, 132.78, 128.30, 127.15, 125.49, 125.12, 123.81, 83.23, 57.58, 52.56, 35.90, 27.86. HRMS (ESI+) calcd for C24H31N2O7 (M+H)+ 459.2126, found 459.2131.
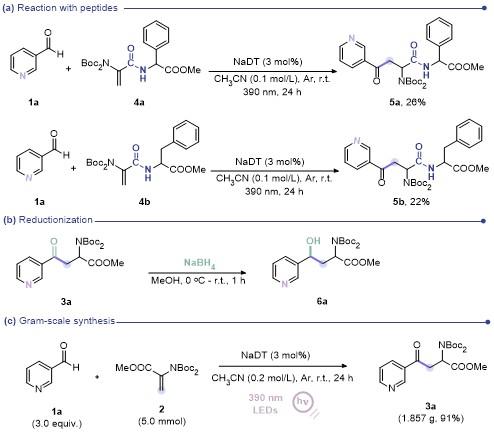
Methyl 2-(2-(bis(tert-butoxycarbonyl)amino)-4-oxo-4-(pyridin-3-yl)butanamido)-2-phenylacetate (5a): 26% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 9.19 (s, 1H), 8.79 (s, 1H), 8.26 (td, J=6.6, 1.7 Hz, 1H), 7.42 (dt, J=8.3, 4.2 Hz, 1H), 7.36~7.29 (m, 5H), 7.17~7.13 (m, 1H), 5.60~5.46 (m, 2H), 4.12 (ddd, J=17.9, 15.1, 6.5 Hz, 1H), 3.72 (d, J=11.6 Hz, 3H), 3.34 (ddd, J=55.0, 17.9, 6.1 Hz, 1H), 1.54 (s, 9H), 1.44 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 196.06, 195.98, 171.12, 170.88, 168.78, 153.48, 152.29, 151.96, 149.63, 136.50, 136.37, 135.82, 128.97, 128.93, 128.56, 128.50, 127.34, 127.09, 123.71, 84.18, 84.10, 56.51, 55.65, 55.47, 52.90, 52.78, 39.30, 39.15, 27.95, 27.86. HRMS (ESI+) calcd for C28H36N3O8 (M+H)+ 542.2497, found 542.2502.
Methyl (2-(bis(tert-butoxycarbonyl)amino)-4-oxo-4-(py-ridin-3-yl)butanoyl)phenylalaninate (5b): 22% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 9.23~9.18 (m, 1H), 8.80 (d, J=4.9 Hz, 1H), 8.27 (dd, J=8.0, 2.0 Hz, 1H), 7.43 (dd, J=8.0, 4.8 Hz, 1H), 7.29 (ddt, J=5.4, 3.7, 1.9 Hz, 2H), 7.25~7.22 (m, 1H), 7.11 (ddd, J=7.8, 6.3, 1.5 Hz, 2H), 6.55 (dd, J=13.0, 7.8 Hz, 1H), 5.46 (dt, J=12.7, 6.3 Hz, 1H), 4.86~4.81 (m, 1H), 4.11 (dd, J=17.8, 6.6 Hz, 1H), 3.67 (d, J=11.9 Hz, 3H), 3.35 (ddd, J=23.7, 17.9, 6.1 Hz, 1H), 3.19~2.95 (m, 3H), 1.50 (d, J=14.3 Hz, 17H); 13C NMR (126 MHz, CDCl3) δ: 196.12, 196.00, 171.58, 171.43, 169.19, 168.90, 153.51, 152.18, 152.10, 149.64, 135.80, 135.71, 135.61, 129.31, 129.19, 128.70, 128.61, 128.45, 127.20, 127.13, 123.71, 84.07, 83.95, 55.58, 55.37, 53.47, 53.40, 52.24, 52.22, 39.30, 39.04, 38.31, 38.05, 27.96, 27.90. HRMS (ESI+) calcd for C29H38N3O8 (M+H)+ 578.2473, found 578.2470.
Methyl (4S)-2-(bis(tert-butoxycarbonyl)amino)-4-hydr- oxy-4-(pyridin-3-yl)butanoate (6a): 33% yield, faint yellow oil. 1H NMR (500 MHz, Chloroform-d) δ: 8.71~8.58 (m, 2H), 7.87 (tt, J=8.0, 1.9 Hz, 1H), 7.39 (dd, J=7.7, 5.1 Hz, 1H), 5.14~4.69 (m, 2H), 3.74 (s, 3H), 3.14 (s, 1H), 2.78-2.40 (m, 1H), 2.29~2.04 (m, 1H), 1.51 (d, J=22.9 Hz, 18H); 13C NMR (126 MHz, CDCl3) δ: 1971.48, 171.03, 152.66, 152.05, 148.13, 146.98, 135.22, 124.15, 84.26, 83.71, 69.64, 67.98, 55.80, 55.32, 52.57, 52.50, 40.44, 39.81, 28.00, 27.98.
Supporting Information 1H NMR and
13C NMR spectra of compounds
3a~
6a. The Supporting Information is available free of charge via the Internet at
http://sioc- journal.cn.