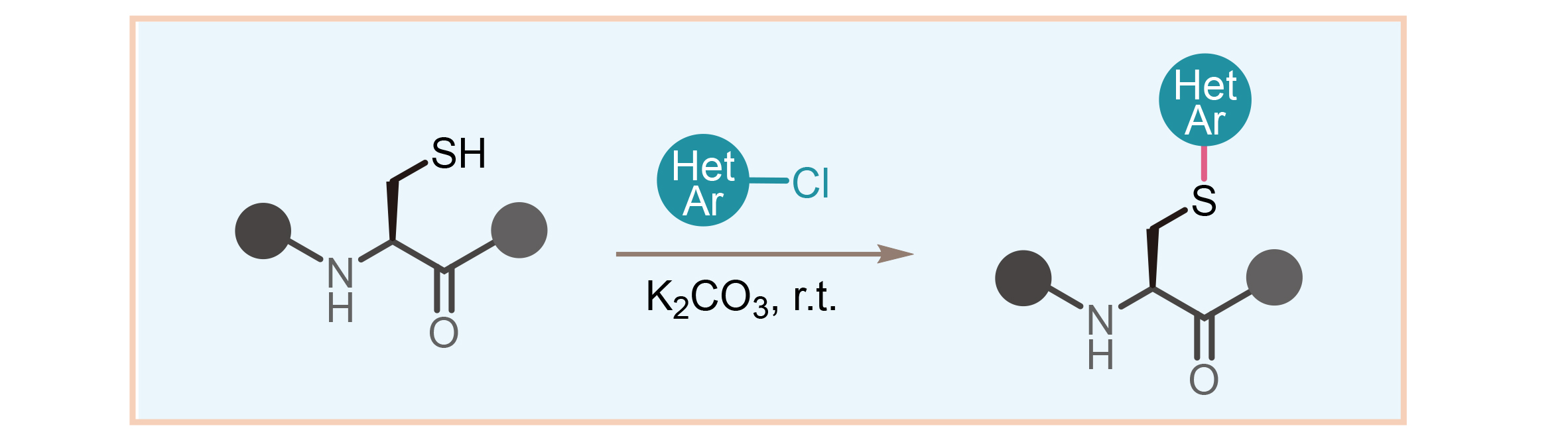
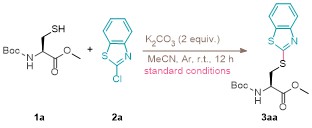
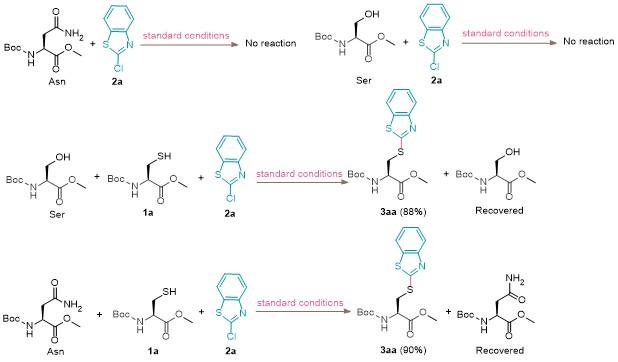
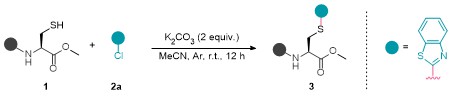
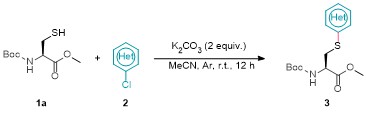
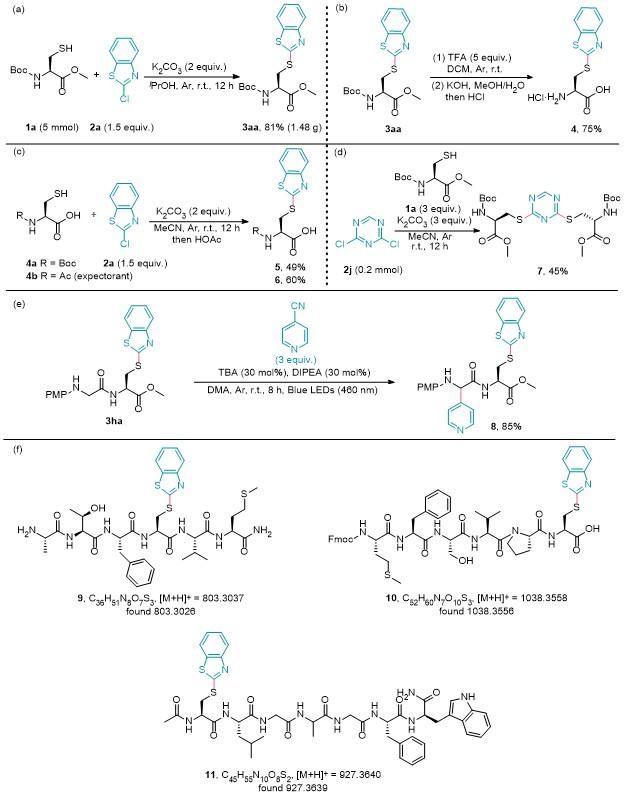
准确称量半胱氨酸衍生物1 (0.2 mmol), K2CO3 (0.4 mmol, 2 equiv.), 加入到装有搅拌棒的10 mL经干燥的反应管中. 然后将反应管置于双排管上抽真空, 并用氩气回填三次. 在氩气氛围下加入溶剂MeCN (2 mL)和氯代杂芳烃2 (1.5 equiv.). 常温搅拌12 h, 通过减压蒸馏移除溶剂, 在层析柱上进行分离(硅胶200~300目, 石油醚/乙酸乙酯, V∶V=10∶1~1∶1). 用旋转蒸发仪浓缩所需组分, 除去溶剂, 得到目标产物3. 此外, 化合物5和6经乙酸酸化后, 均通过此方法进行制备与分离. 化合物9~11在标准条件下反应生成, 待反应结束后对混合物溶液进行处理, 并通过HRMS进行分析, 与目标产物分子量相符合.
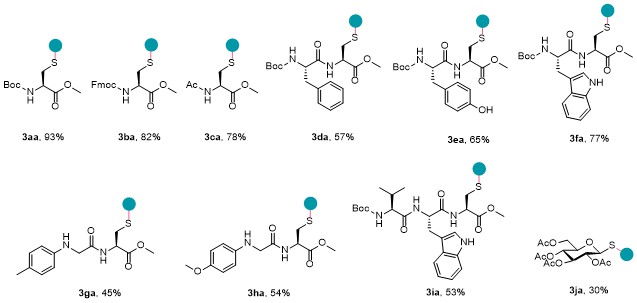
S-(苯并[d]噻唑-2-基)-N-(叔丁氧羰基)-L-半胱氨酸甲酯(3aa): 68.5 mg, 产率93%. 白色固体, m.p. 145~148 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.86 (d, J=8.1 Hz, 1H), 7.74 (d, J=8.0 Hz, 1H), 7.42 (t, J=7.7 Hz, 1H), 7.33~7.28 (m, 1H), 6.22 (d, J=7.7 Hz, 1H), 4.74 (q, J=6.0 Hz, 1H), 3.82 (d, J=5.3 Hz, 2H), 3.73 (s, 3H), 1.40 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 170.8, 165.8, 155.2, 152.7, 135.5, 126.1, 124.5, 121.5, 121.0, 80.0, 53.8, 52.6, 35.4, 28.2. HRMS (ESI-qTOF) calcd for C16H20N2O4S2K [M+K]+ 407.0496, found 407.0481.
N-(((9H-芴-9-基)甲氧基)羰基)-S-(苯并[d]噻唑-2-基)-L-半胱氨酸甲酯(3ba): 80.5 mg, 产率82%. 黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 7.86 (d, J=8.1 Hz, 1H), 7.75 (d, J=7.7 Hz, 1H), 7.72 (d, J=7.4 Hz, 2H), 7.50 (t, J=8.2 Hz, 2H), 7.42 (t, J=7.1 Hz, 1H), 7.37~7.30 (m, 4H), 7.17 (q, J=7.4 Hz, 2H), 6.90 (d, J=7.3 Hz, 1H), 4.79 (q, J=6.8 Hz, 1H), 4.37~4.35 (m, 2H), 4.15 (t, J=7.1 Hz, 1H), 3.85~3.84 (m, 2H), 3.77 (s, 3H); 13C NMR (151 MHz, CDCl3) δ: 170.6, 166.2, 156.0, 152.5, 143.7, 141.2, 135.5, 127.6, 127.0, 126.4, 125.1, 124.7, 121.5, 121.1, 120.0, 67.1, 54.8, 52.8, 47.1, 34.9. HRMS (ESI-qTOF) calcd for C26H23N2O4S2 [M+H]+ 491.1094, found 491.1096.
N-乙酰基-(苯并[d]噻唑-2-基)-L-半胱氨酸甲酯(3ca): 48.4 mg, 产率78%. 白色固体, m.p. 110~115 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.86 (d, J=8.1 Hz, 1H), 7.77 (d, J=7.9 Hz, 1H), 7.55 (d, J=6.5 Hz, 1H), 7.47~7.43 (m, 1H), 7.35~7.32 (m, 1H), 4.96 (q, J=6.4 Hz, 1H), 3.83~3.81 (m, 2H), 3.75 (s, 3H), 1.99 (s, 3H); 13C NMR (151 MHz, CDCl3) δ: 170.5, 170.1, 166.4, 152.5, 135.6, 126.4, 124.8, 121.3, 121.2, 53.2, 52.72, 34.8, 23.0. HRMS (ESI-qTOF) calcd for C13H15N2O3S2 [M+H]+ 311.0519, found 311.0517.
S-(苯并[d]噻唑-2-基)-N-((叔丁氧羰基)-L-苯丙氨酸基)-L-半胱氨酸甲酯(3da): 58.8 mg, 产率57%. 白色固体, m.p. 158~163 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.90~7.78 (m, 2H), 7.76 (d, J=8.0 Hz, 1H), 7.44 (t, J=7.7 Hz, 1H), 7.34 (t, J=7.6 Hz, 1H), 7.19~6.88 (m, 5H), 5.05~4.94 (m, 1H), 4.94~4.86 (m, 1H), 4.47~4.34 (m, 1H), 3.84~3.76 (m, 2H), 3.73 (s, 3H), 3.24~2.62 (m, 2H), 1.39 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 171.4, 170.0, 166.3, 155.3, 152.5, 136.3, 135.6, 129.3, 128.4, 126.8, 126.4, 124.8, 121.6, 121.1, 80.0, 55.7, 53.1, 52.6, 38.6, 34.8, 28.2. HRMS (ESI-qTOF) calcd for [M+H]+ C25H30N3O5S2 516.1621, found 516.1623.
S-(苯并[d]噻唑-2-基)-N-((叔丁氧羰基)-L-酪氨酸)-L-半胱氨酸甲酯(3ea): 69.1 mg, 产率65%. 白色固体, m.p. 135~137 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.19 (s, 1H), 7.71 (d, J=7.9 Hz, 2H), 7.66~7.62 (m, 1H), 7.51 (d, J=7.9 Hz, 1H), 7.35 (t, J=7.7 Hz, 1H), 7.30 (d, J=7.6 Hz, 1H), 7.26~7.22 (m, 1H), 7.10 (t, J=7.6 Hz, 1H), 7.05~6.99 (m, 1H), 5.19~5.08 (m, 1H), 4.87 (q, J=6.0 Hz, 1H), 4.50 (s, 1H), 3.77~3.66 (m, 2H), 3.65 (s, 3H), 3.20 (t, J=6.8 Hz, 2H), 1.39 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 171.8, 169.9, 166.4, 155.3, 155.0, 152.3, 135.5, 130.4, 127.4, 126.5, 124.8, 121.5, 121.2, 115.4, 80.2, 55.8, 53.2, 52.7, 37.8, 34.7, 28.2. HRMS (ESI-qTOF) calcd for C25H30N3O6S2 [M+H]+ 532.1571, found 532.1572.
S-(苯并[d]噻唑-2-基)-N-((叔丁氧羰基)-L-色氨酸)-L-半胱氨酸甲酯(3fa): 85.4 mg, 产率77%. 白色固体, m.p. 91~94 ℃; 1H NMR (400 MHz,CDCl3) δ: 7.90~7.78 (m, 2H), 7.76 (d, J=8.0 Hz, 1H), 7.44 (t, J=7.7 Hz, 1H), 7.34 (t, J=7.6 Hz, 1H), 7.17~6.95 (m, 5H), 5.05~4.94 (m, 1H), 4.92 (d, J=5.9 Hz, 1H), 4.42 (d, J=9.1 Hz, 1H), 3.84~3.75 (m, 2H), 3.73 (s, 3H), 3.30~2.66 (m, 2H), 1.39 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 171.9, 170.0, 166.1, 155.3, 152.4, 136.1, 135.4, 127.6, 126.4, 124.7, 123.3, 122.0, 121.5, 121.0, 119.5, 118.6, 111.0, 110.1, 79.9, 55.3, 52.9, 52.6, 34.8, 29.2, 28.2. HRMS (ESI-qTOF) calcd for C27H31N4O5S2 [M+H]+ 555.1730, found 555.1732.
S-(苯并[d]噻唑-2-基)-N-(对甲苯基甘油基)-L-半胱氨酸甲酯(3ga): 37.4 mg, 产率45%. 白色固体, m.p. 120~125 ℃; 1H NMR (400 MHz, CDCl3) 8.14 (d, J=7.6 Hz, 1H), 7.79 (d, J=8.1 Hz, 1H), 7.71 (d, J=8.0 Hz, 1H), 7.41 (t, J=7.1 Hz, 1H), 7.31 (t, J=7.0 Hz, 1H), 6.83 (d, J=8.1 Hz, 2H), 6.27 (d, J=8.3 Hz, 2H), 5.02~4.92 (m, 1H), 3.88~3.82 (m, 2H), 3.75 (s, 3H), 3.74~3.64 (m, 2H), 2.19 (s, 3H); 13C NMR (151 MHz, CDCl3) δ: 171.6, 170.3, 166.2, 152.5, 144.7, 135.7, 129.5, 128.2, 126.2, 124.5, 121.5, 121.1, 113.3, 53.1, 52.7, 49.4, 33.9, 20.4. HRMS (ESI-qTOF) calcd for C20H21N3O3S2Na [M+Na]+ 438.0917, found 438.0931.
S-(苯并[d]噻唑-2-基)-N-((4-甲氧基苯基)甘油基)-L-半胱氨酸甲酯(3ha): 46.6 mg, 产率54%. 白色固体, m.p. 97~100 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.23 (d, J=7.5 Hz, 1H), 7.79 (d, J=8.1 Hz, 1H), 7.70 (d, J=7.9 Hz, 1H), 7.43~7.39 (m, 1H), 7.33~7.29 (m, 1H), 6.83~6.77 (m, 1H), 6.60 (d, J=8.9 Hz, 2H), 6.28 (d, J=8.9 Hz, 2H), 4.98~4.93 (m, 1H), 3.89~3.83 (m, 2H), 3.75 (s, 3H), 3.72 (s, 1H), 3.70 (s, 3H), 3.64 (d, J=17.4 Hz, 1H); 13C NMR (151 MHz, CDCl3) δ: 171.7, 170.3, 166.2, 153.0, 152.4, 141.0, 135.7, 126.1, 124.5, 121.4, 121.1, 114.5, 114.4, 55.7, 53.2, 52.7, 49.9, 33.8. HRMS (ESI-qTOF) calcd for C20H22N3O4S2 [M+H]+ 432.1046, found 432.1058.
S-(苯并[d]噻唑-2-基)-N-(叔丁氧羰基)-L-缬氨酸-L-色氨酰基-L-半胱氨酸甲酯(3ia): 70.0 mg, 产率57%. 白色固体, m.p. 157~159 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.19 (s, 1H), 7.73~7.68 (m, 2H), 7.66 (d, J=8.1 Hz, 1H), 7.63 (d, J=7.8 Hz, 1H), 7.35 (t, J=7.6 Hz, 1H), 7.29 (d, J=8.3 Hz, 1H), 7.23 (d, J=8.0 Hz, 1H), 7.15 (d, J=2.3 Hz, 1H), 7.12~7.01 (m, 2H), 6.75 (d, J=7.6 Hz, 1H), 4.97 (d, J=8.0 Hz, 1H), 4.86 (q, J=5.8 Hz, 1H), 4.82~4.72 (m, 1H), 3.99~3.88 (m, 1H), 3.78~3.67 (m, 2H), 3.67 (s, 3H), 3.38~3.08 (m, 2H), 2.24~2.05 (m, 1H), 1.39 (s, 9H), 0.91 (d, J=6.8 Hz, 3H), 0.81 (d, J=6.8 Hz, 3H); 13C NMR (151 MHz, CDCl3) δ: 171.2, 169.8, 165.9, 155.9, 152.4, 136.1, 135.3, 128.8, 127.5, 126.4, 124.7, 123.7, 122.0, 121.5, 121.0, 119.6, 118.5, 111.1, 109.9, 79.9, 65.5, 60.0, 53.9, 52.6, 34.8, 30.7, 28.2, 19.2, 17.3. HRMS (ESI-qTOF) calcd for C32H40N5O6S2 [M+H]+ 654.2415, found 654.2415.
(2R,3R,4S,5R,6S)-2-(乙酰氧基甲基)-6-(苯并[d]噻唑-2-基硫基)四氢-2H-吡喃-3,4,5-三酰基三乙酸酯(3ja): 29.9 mg, 产率30%. 白色固体, m.p. 131~134 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.94 (d, J=8.1 Hz, 1H), 7.80 (d, J=7.8 Hz, 1H), 7.51~7.42 (m, 1H), 7.40~7.32 (m, 1H), 5.58 (d, J=10.3 Hz, 1H), 5.36 (t, J=9.3 Hz, 1H), 5.28~5.14 (m, 2H), 4.31 (dd, J=12.5, 4.9 Hz, 1H), 4.19 (dd, J=12.5, 2.3 Hz, 1H), 3.98~3.89 (m, 1H), 2.06 (s, 3H), 2.05 (s, 3H), 2.05 (s, 3H), 2.03 (s, 3H); 13C NMR (151 MHz, CDCl3) δ: 170.6, 170.1, 169.4, 169.4, 161.9, 152.7, 135.8, 126.4, 125.0, 122.3, 121.0, 84.0, 76.3, 73.7, 69.7, 68.1, 61.8, 20.7, 20.6, 20.6. HRMS (ESI-qTOF) calcd for C21H24NO9S2 [M+H]+ 498.0887, found 498.0886.
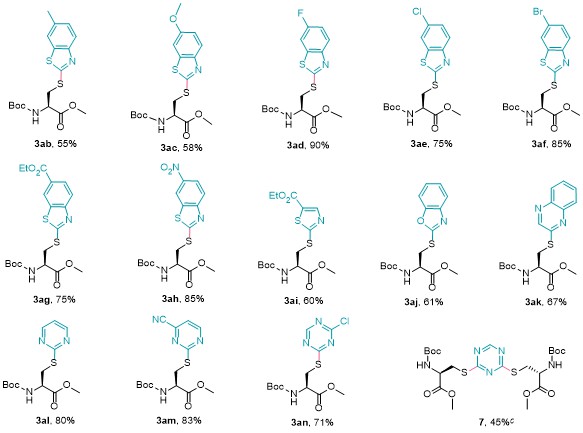
N-(叔丁氧羰基)-S-(6-甲基苯并[d]噻唑-2-基)-L-半胱氨酸甲酯(3ab): 42.1 mg, 产率55%. 白色固体, m.p. 143~144 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.74 (d, J=8.3 Hz, 1H), 7.53 (s, 1H), 7.23 (d, J=8.3 Hz, 1H), 6.29 (d, J=7.5 Hz, 1H), 4.72 (q, J=6.0 Hz, 1H), 3.79 (dd, J=5.3, 2.3 Hz, 2H), 3.72 (s, 3H), 2.45 (s, 3H), 1.41 (s, 9H); 13C NMR (151 MHz, cdcl3) δ: 170.8, 164.4, 155.3, 150.8, 135.7, 134.7, 127.6, 121.0, 120.8, 79.9, 53.9, 52.6, 35.4, 28.2, 21.4. HRMS (ESI-qTOF) calcd for C17H23N2O4S2[M+H]+ 383.1094, found 383.1094.
N-(叔丁氧羰基)-S-(6-甲氧基苯并[d]噻唑-2-基)-L-半胱氨酸甲酯(3ac): 46.2 mg, 产率58%. 白色固体, m.p. 150~152 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.75 (d, J=8.8 Hz, 1H), 7.21 (d, J=2.4 Hz, 1H), 7.05~6.98 (m, 1H), 6.28 (d, J=7.5 Hz, 1H), 4.72 (q, J=5.9 Hz, 1H), 3.85 (s, 3H), 3.77 (d, J=5.1 Hz, 2H), 3.72 (s, 3H), 1.41 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 170.8, 162.4, 157.3, 155.2, 147.3, 136.9, 122.0, 115.0, 104.0, 79.9, 55.7, 53.8, 52.6, 35.6, 28.2. HRMS (ESI-qTOF) calcd for C17H23N2O5S2 [M+H]+ 399.1043, found 399.1043.
N-(叔丁氧羰基)-S-(6-氟苯并[d]噻唑-2-基)-L-半胱氨酸甲酯(3ad): 69.6 mg, 产率90%. 白色固体, m.p. 145~148 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.80 (dd, J=8.9, 4.8 Hz, 1H), 7.44 (dd, J=8.0, 2.6 Hz, 1H), 7.19~7.09 (m, 1H), 6.09 (d, J=7.7 Hz, 1H), 4.74 (q, J=6.1 Hz, 1H), 3.84~3.77 (m, 2H), 3.73 (s, 3H), 1.41 (s, 9H); 13C NMR (151 MHz, CDCl3) 170.76, 165.32 (d, J=2.5 Hz), 160.76, 159.13, 155.16, 149.40 (d, J=5.4 Hz), 136.45 (d, J=10.9 Hz), 122.29 (d, J=9.3 Hz), 114.53 (d, J=24.5 Hz), 107.47 (d, J=27.1 Hz), 80.03, 77.21, 77.00, 76.79, 53.69, 52.63, 35.49, 28.19; 19F NMR (376 MHz, CDCl3) δ: -116.55. HRMS (ESI-qTOF) calcd for C16H20FN2O4S2 [M+H]+ 387.0843, found 387.0843.
S-(6-溴苯并[d]噻唑-2-基)-N-(叔丁氧羰基)-L-半胱氨酸甲酯(3ae): 76.1 mg, 产率85%. 黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 7.87 (d, J=1.9 Hz, 1H), 7.70 (d, J=8.6 Hz, 1H), 7.53~7.50 (m, 1H), 6.01 (d, J=7.8 Hz, 1H), 4.74 (d, J=6.6 Hz, 1H), 3.87~3.81 (m, 2H), 3.73 (s, 3H), 1.41 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 170.7, 166.6, 155.2, 151.6, 137.1, 129.6, 123.5, 122.5, 118.0, 80.1, 53.6, 52.7, 35.4, 28.2. HRMS (ESI-qTOF) calcd for C16H20BrN2O4S2 [M+H]+ 444.9896, found 444.9892.
(R)-2-((2-((叔丁氧羰基)氨基)-3-甲氧基-3-氧代丙基)硫代)苯并噻唑-6-羧酸乙酯(3af): 66.1 mg, 产率75%. 黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 8.46 (s, 1H), 8.11 (d, J=8.6 Hz, 1H), 7.87 (d, J=8.5 Hz, 1H), 6.05 (d, J=7.8 Hz, 1H), 4.76 (q, J=6.3 Hz, 1H), 4.41 (q, J=7.2 Hz, 2H), 3.93~3.85 (m, 2H), 3.75 (s, 3H), 1.42 (t, J=7.1 Hz, 1H), 1.41 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 170.7, 169.9, 165.9, 155.5, 155.2, 135.4, 127.5, 126.7, 123.0, 121.0, 80.1, 61.2, 53.6, 52.6, 35.4, 28.2, 14.3. HRMS (ESI-qTOF) calcd for C19H25N2O6S2 [M+H]+ 441.1149, found 441.1147.
N-(叔丁氧羰基)-S-(6-硝基苯并[d]噻唑-2-基)-L-半胱氨酸甲酯(3ag): 70.3 mg, 产率85%. 白色固体, m.p. 183~184 ℃. 1H NMR (400 MHz, CDCl3) δ: 8.68 (d, J=2.3 Hz, 1H), 8.31 (dd, J=9.0, 2.3 Hz, 1H), 7.91 (d, J=9.0 Hz, 1H), 5.83 (d, J=7.8 Hz, 1H), 4.78 (q, J=6.5 Hz, 1H), 4.02~3.81 (m, 2H), 3.77 (s, 3H), 1.41 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 173.0, 170.6, 156.4, 155.0, 144.2, 135.7, 121.9, 121.3, 117.4, 80.2, 53.2, 52.7, 35.5, 28.2. HRMS (ESI-qTOF) calcd for C16H20N3O6S2 [M+H]+ 414.0788, found 414.0787.
N-(叔丁氧羰基)-S-(4-氯苯并[d]噻唑-2-基)-L-半胱氨酸甲酯(3ah): 60.4 mg, 产率75%. 白色固体, m.p. 115~118 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.62 (d, J=8.0 Hz, 1H), 7.43 (d, J=6.8 Hz, 1H), 7.23 (t, J=8.0 Hz, 1H), 6.30 (d, J=7.5 Hz, 1H), 4.77 (q, J=5.9 Hz, 1H), 3.84 (d, J=5.2 Hz, 2H), 3.74 (s, 3H), 1.41 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 170.7, 167.5, 155.3, 149.8, 136.8, 126.5, 126.4, 125.1, 119.4, 80.0, 54.0, 52.5, 35.5, 28.2. HRMS (ESI-qTOF) calcd for C16H20ClN2O4S2 [M+H]+ 403.0548, found 403.0546.
(R)-2-((2-((叔丁氧羰基)氨基)-3-甲氧基-3-氧代丙基)硫代)噻唑-5-羧酸乙酯(3ai): 46.9 mg, 产率60%. 淡黄色油状液体. 1H NMR (400 MHz, CDCl3) δ: 8.19 (s, 1H), 5.55 (d, J=7.9 Hz, 1H), 4.70 (d, J=6.8 Hz, 1H), 4.37 (q, J=7.1 Hz, 2H), 3.76 (s, 3H), 3.74~3.65 (m, 2H), 1.43 (s, 9H), 1.39~1.35 (m, 3H); 13C NMR (151 MHz, CDCl3) δ: 170.4, 161.1, 160.5, 154.9, 145.1, 134.5, 80.2, 61.2, 53.0, 52.7, 34.9, 28.1, 14.2. HRMS (ESI-qTOF) calcd for C15H22N2O6S2Na [M+Na]+ 413.0811, found 413.0815.
S-(苯并[d]噁唑-2-基)-N-(叔丁氧羰基)-L-半胱氨酸甲酯(3aj): 43.0 mg, 产率61%. 白色固体, m.p. 95~97 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.58 (d, J=7.6 Hz, 1H), 7.44 (d, J=6.7 Hz, 1H), 7.31~7.27 (m, 1H), 7.26~7.23 (m, 1H), 5.78~5.76 (m, 1H), 4.76 (d, J=6.3 Hz, 1H), 3.86~3.81 (m, 2H), 3.75 (s, 3H), 1.40 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 170.6, 164.0, 152.1, 141.7, 124.4, 124.2, 118.5, 110.0, 80.2, 53.5, 52.7, 34.5, 28.2. HRMS (ESI-qTOF) calcd for C16H21N2O5S [M+H]+ 353.1166, found 353.1164.
N-(叔丁氧羰基)-S-(喹喔啉-2-基)-L-半胱氨酸甲酯(3ak): 48.7 mg, 产率67%. 白色固体, m.p. 114~115 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.62 (d, J=1.8 Hz, 1H), 8.02 (d, J=8.0 Hz, 1H), 7.97 (d, J=8.3 Hz, 1H), 7.71 (t, J=7.6 Hz, 1H), 7.68~7.59 (m, 1H), 6.26 (d, J=7.7 Hz, 1H), 4.76 (d, J=6.3 Hz, 1H), 3.86~3.77 (m, 2H), 3.73 (s, 3H), 1.40 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 171.1, 155.2, 154.7, 144.4, 142.1, 140.1, 130.3, 129.2, 128.5, 127.6, 79.9, 53.8, 52.5, 31.8, 28.2. HRMS (ESI-qTOF) calcd for C17H22N3O4S [M+H]+ 364.1326, found 364.1323.
N-(叔丁氧羰基)-S-(嘧啶-2-基)-L-半胱氨酸甲酯(3al): 50.1 mg, 产率80%. 无色油状液体. 1H NMR (400 MHz, CDCl3) δ: 8.52 (d, J=4.9 Hz, 2H), 7.02 (t, J=4.8 Hz, 1H), 5.73 (d, J=7.9 Hz, 1H), 4.67 (d, J=6.2 Hz, 1H), 3.75 (s, 3H), 3.72~3.54 (m, 2H), 1.41 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 171.4, 171.3, 157.2, 155.2, 116.9, 79.8, 53.6, 52.4, 32.7, 28.2. HRMS (ESI-qTOF) calcd for C13H20N3O4S [M+H]+ 314.1169, found 314.1166.
N-(叔丁氧羰基)-S-(4-氰基嘧啶-2-基)-L-半胱氨酸甲酯(3am): 56.1 mg, 产率83%. 白色固体, m.p. 108~110 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.74 (d, J=4.9 Hz, 1H), 7.34 (d, J=4.8 Hz, 1H), 5.48 (d, J=8.3 Hz, 1H), 4.70 (d, J=6.8 Hz, 1H), 3.78 (s, 3H), 3.76~3.50 (m, 2H), 1.43 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 173.5, 171.0, 158.8, 155.0, 141.2, 119.4, 114.9, 80.1, 52.7, 52.6, 33.2, 28.1. HRMS (ESI-qTOF) calcd for C14H18N4O4SNa [M+Na]+ 361.0941, found 361.0940.
N-(叔丁氧羰基)-S-(4-氯-1,3,5-三嗪-2-基)-L-半胱氨酸甲酯(3an): 49.5 mg, 产率71%. 白色固体, m.p. 141~143 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.68 (s, 1H), 5.45 (d, J=8.1 Hz, 1H), 4.76~4.67 (m, 1H), 3.82 (d, J=4.7 Hz, 1H), 3.79 (s, 3H), 3.55 (dd, J=14.1, 6.8 Hz, 1H), 1.43 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 184.0, 170.7, 170.0, 165.7, 154.9, 80.4, 52.8, 52.6, 32.9, 28.2. HRMS (ESI-qTOF) calcd for C12H18ClN4O4S [M+H]+ 349.0732, found 349.0735.
将三氟乙酸(75 μL)加入到含有化合物3aa (65.1 mg, 0.2 mmol)的二氯甲烷(DCM) (0.5 mL)溶液中, 所得混合物于室温下搅拌30 min. 反应完成后, 除去溶剂, 用水和二氯甲烷(10 mL)萃取. 合并水层, 去除溶剂, 将所得粗产物溶解于甲醇(1 mL)中, 并随后加入氢氧化钾水溶液(1 mL, 1 mol/L). 所得混合物在室温下搅拌1 h. 通过薄层色谱(TLC)监测反应终点. 反应完成后, 蒸除溶剂, 并用水溶解, DCM萃取. 向水层加入1 mol/L盐酸调节pH至1. 用乙酸乙酯洗涤以去除脂溶性杂质. 收集水相并在减压下浓缩, 得到S-(苯并[d]噻唑-2-基)-L-半胱氨酸盐酸盐(4) 38.2 mg, 产率75%. 白色固体, m.p. 230~233 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.88 (s, 2H), 8.82~8.64 (m, 1H), 8.04 (d, J=8.0 Hz, 1H), 7.88 (d, J=8.1 Hz, 1H), 7.63~7.45 (m, 1H), 7.39 (t, J=7.6 Hz, 1H), 4.39 (s, 1H), 4.05~3.87 (m, 2H); 13C NMR (151 MHz, DMSO-d6) δ: 169.0, 165.2, 152.4, 134.9, 126.6, 124.9, 122.1, 121.4, 51.5, 32.4. HRMS (ESI-qTOF) calcd for C10H11N2O2S2 [M+H]+ 255.0256, found 255.0258.
S-(苯并[d]噻唑-2-基)-N-(叔丁氧羰基)-L-半胱氨酸(5): 34.7 mg, 产率49%. 白色固体, m.p. 145~148 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.77 (d, J=8.1 Hz, 1H), 7.56 (d, J=7.8 Hz, 1H), 7.31~7.13 (m, 2H), 6.91~6.61 (m, 1H), 4.63~4.42 (m, 1H), 3.99~3.53 (m, 2H), 1.30 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 176.72, 167.24, 156.52, 152.57, 135.30, 125.92, 124.16, 121.35, 120.77, 79.83, 77.21, 77.00, 76.79, 55.56, 36.30, 28.28. HRMS (ESI-qTOF) calcd for C15H19N2O4S2 [M+H]+ 355.0781, found 355.0780.
N-乙酰基-(苯并[d]噻唑-2-基)-L-半胱氨酸(6): 35.6 mg, 产率60%. 白色固体, m.p. 197~198 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.61 (d, J=5.2 Hz, 1H), 7.85 (d, J=8.4 Hz, 1H), 7.79 (d, J=7.5 Hz, 1H), 7.54~7.45 (m, 1H), 7.43~7.34 (m, 1H), 4.90~4.79 (m, 1H), 3.88~3.64 (m, 2H), 2.03 (s, 3H); 13C NMR (151 MHz, CDCl3) δ: 173.04, 170.89, 168.97, 151.65, 135.34, 126.74, 125.20, 121.42, 120.85, 77.21, 77.00, 76.79, 55.26, 34.97, 22.70. HRMS (ESI-qTOF) calcd for C12H13N2O3S2 [M+H]+ 297.0362, found 297.0363.
3,3'-(1,3,5-三嗪-2,4-二基)双(磺胺二基)(2R,2'R)-双(2-((叔丁氧羰基)氨基)丙酸二甲酯)(7): 59.1 mg, 产率54%. 白色固体, m.p. 152~155 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.48 (s, 1H), 5.49 (d, J=8.1 Hz, 2H), 4.66 (q, J=6.8 Hz, 2H), 3.79 (d, J=2.4 Hz, 2H), 3.76 (s, 6H), 3.49 (dd, J=14.2, 6.8 Hz, 2H), 1.43 (s, 18H); 13C NMR (151 MHz, CDCl3) δ: 180.7, 170.9, 163.1, 155.0, 129.7, 80.2, 52.9, 52.7, 32.3, 28.2. HRMS (ESI-qTOF) calcd for C21- H34N5O8S2 [M+H]+ 548.1843, found 548.1843.
S-(苯并[d]噻唑-2-基)-N-(2-((4-甲氧基苯基)氨基)-2-(吡啶-4-基)乙酰基)-L-半胱氨酸甲酯(8): 86.5 mg, 产率71%. 白色固体, m.p. 170~172 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 8.26 (d, J=8.0 Hz, 1H), 7.66 (d, J=5.9 Hz, 2H), 7.14 (d, J=8.0 Hz, 1H), 6.99 (d, J=8.6 Hz, 1H), 6.64~6.61 (m, 2H), 6.54~6.48 (m, 1H), 5.83 (d, J=9.1 Hz, 2H), 5.77 (d, J=9.0 Hz, 2H), 5.11 (d, J=7.7 Hz, 1H), 4.16 (d, J=7.6 Hz, 1H), 3.99~3.89 (m, 1H), 3.07 (dd, J=13.7, 4.9 Hz, 1H), 2.85~2.77 (m, 2H), 2.75 (s, 3H), 2.71 (s, 3H); 13C NMR (151 MHz, DMSO-d6) δ: 170.5, 170.1, 165.7, 152.5, 151.6, 149.5, 147.6, 140.9, 134.6, 126.4, 124.6, 122.6, 121.8, 121.2, 114.6, 114.4, 60.8, 55.2, 52.3, 51.3, 33.6. HRMS (ESI-qTOF) calcd for C25H25N4O4S2 [M+H]+ 509.1312, found 509.1310.
辅助材料(Supporting Information) 化合物
3,
4~
8的
1H NMR,
13C NMR和
19F NMR图谱, 以及化合物
9~
11的高分辨质谱图. 这些材料可以免费从本刊网站(
http://sioc-journal.cn/)上下载.