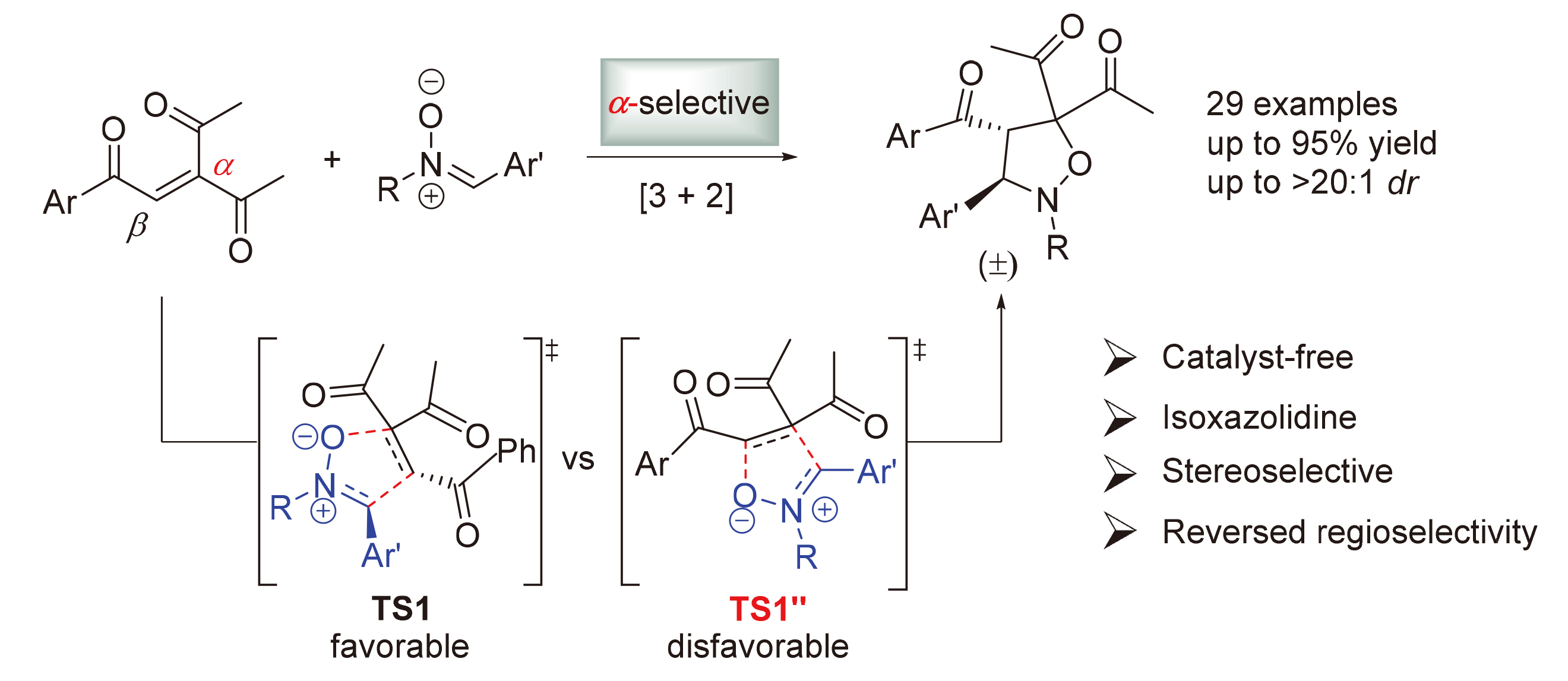
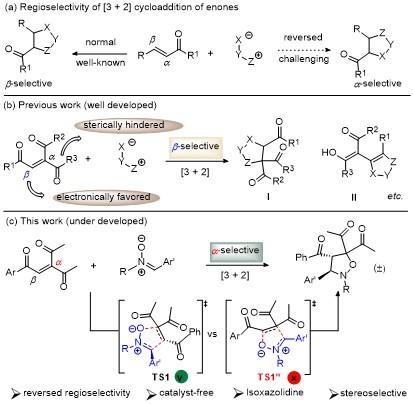
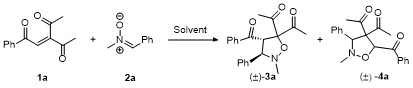
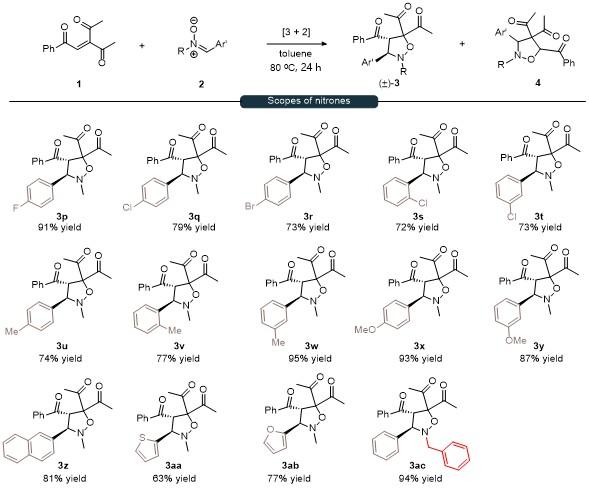
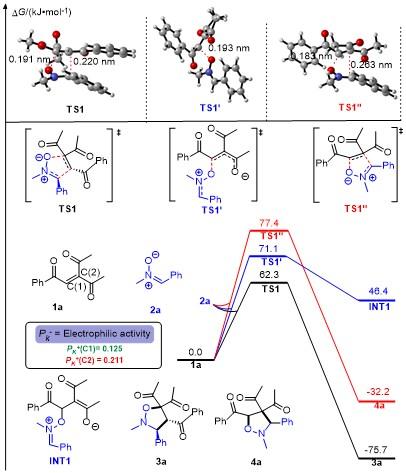
将三酮烯烃1 (0.2 mmol)、硝酮2 (mmol)和甲苯(1 mL)加入密封管中, 在80 ℃下搅拌24 h, 使用薄层色谱法检测反应. 反应完成后, 直接减压浓缩除去溶剂, 所得粗产物用柱层析纯化(石油醚/乙酸乙酯, V∶V=10∶1~6∶1), 得到目标产物3.
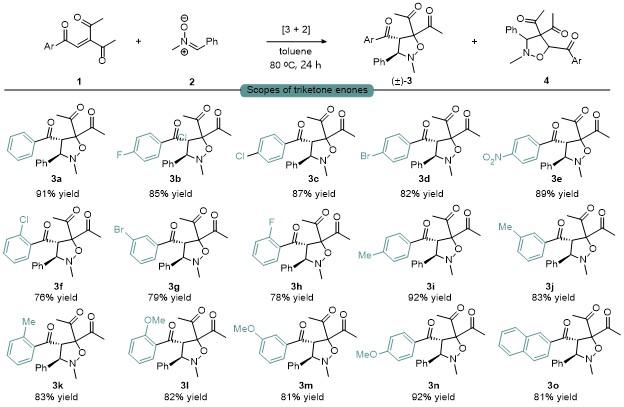
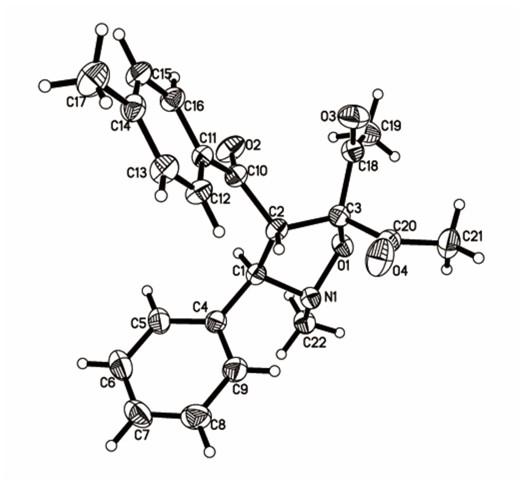
1,1'-(4-苯甲酰-2-甲基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3a): 白色固体, 产率91%. m.p. 98.2~99.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.77 (d, J=7.5 Hz, 2H), 7.48 (t, J=7.4 Hz, 1H), 7.31 (t, J=7.8 Hz, 2H), 7.29~7.25 (m, 3H), 7.22~7.15 (m, 2H), 5.51 (d, J=8.3 Hz, 1H), 3.96 (d, J=8.3 Hz, 1H), 2.71 (s, 3H), 2.36 (d, J=2.4 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.2, 202.9, 198.2, 136.4, 136.2, 133.8, 129.2, 128.9, 128.7, 128.5, 128.2, 97.5, 78.4, 63.8, 42.7, 28.3, 24.7; HRMS (ESI) calcd for C21H21NO4Na [M+Na]+ 374.1363, found 374.1367.
1,1'-(4-(4-氟苯甲酰)-2-甲基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3b): 白色固体, 产率85%. m.p. 152.7~ 153.4 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.88~7.73 (m, 2H), 7.32~7.27 (m, 3H), 7.23~7.13 (m, 2H), 6.98 (t, J=8.6 Hz, 2H), 5.44 (d, J=8.4 Hz, 1H), 3.98 (d, J=8.4 Hz, 1H), 2.71 (s, 3H), 2.36 (d, J=3.4 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.1, 203.0, 196.5, 166.2 (d, J=256.6 Hz), 136.1, 132.9 (d, J=2.8 Hz), 132.0 (d, J=9.7 Hz), 129.0, 128.8, 128.1, 115.7 (d, J=22.0 Hz), 97.4, 78.3, 63.8, 42.7, 28.3; 19F NMR (376 MHz, CDCl3) δ: -103.5; HRMS (ESI) calcd for C21H20FNO4Na [M+Na]+ 392.1268, found 392.1276.
1,1'-(4-(4-氯苯甲酰)-2-甲基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3c): 白色固体, 产率87%. m.p. 135.8~ 136.2 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.72 (d, J=8.6 Hz, 2H), 7.34~7.26 (m, 5H), 7.22~7.14 (m, 2H), 5.42 (d, J=8.4 Hz, 1H), 3.97 (d, J=8.4 Hz, 1H), 2.71 (s, 3H), 2.35 (d, J=3.2 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.0, 202.9, 197.0, 140.5, 136.0, 134.8, 130.6, 129.0, 128.9, 128.8, 128.1, 97.5, 78.3, 63.8, 42.7, 28.3, 24.7; HRMS (ESI) calcd for C21H20ClNO4Na [M+Na]+ 408.0973, found 408.0977.
1,1'-(4-(4-溴苯甲酰)-2-甲基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3d): 白色固体, 产率82%. m.p. 147.8~148.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.64 (d, J=8.6 Hz, 2H), 7.46 (d, J=8.6 Hz, 2H), 7.35~7.26 (m, 3H), 7.22~7.14 (m, 2H), 5.41 (d, J=8.4 Hz, 1H), 3.97 (d, J=8.4 Hz, 1H), 2.71 (s, 3H), 2.35 (d, J=3.1 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.0, 202.9, 197.3, 136.0, 135.2, 131.8, 130.6, 129.4, 129.0, 128.9, 128.1, 97.5, 78.3, 63.8, 42.7, 28.3, 24.7; HRMS (ESI) calcd for C21H21Br- NO4Na [M+Na]+ 452.0468, found 452.0473.
1,1'-(2-甲基-4-(4-硝基苯甲酰)-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3e): 白色固体, 产率89%. m.p. 110.6~111.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.17 (d, J=8.5 Hz, 2H), 7.97 (d, J=8.4 Hz, 2H), 7.37~7.27 (m, 3H), 7.24~7.13 (m, 2H), 5.44 (d, J=8.5 Hz, 1H), 4.01 (d, J=8.4 Hz, 1H), 2.73 (s, 3H), 2.38 (s, 6H); 13C NMR (100 MHz, CDCl3) δ: 203.8, 203.0, 197.4, 150.5, 141.0, 135.6, 130.1, 129.1, 129.1, 127.9, 123.6, 97.7, 78.4, 64.2, 42.7, 28.4, 24.8; HRMS (ESI) calcd for C21H20N2O6Na [M+ Na]+ 419.1213, found 419.1217.
1,1'-(4-(2-氯苯甲酰)-2-甲基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3f): 白色固体, 产率76%. m.p. 142.9~143.4 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.54 (d, J=7.6 Hz, 1H), 7.31~7.27 (m, 2H), 7.25~7.18 (m, 4H), 7.16~7.10 (m, 2H), 5.33 (d, J=8.5 Hz, 1H), 3.90 (d, J=8.5 Hz, 1H), 2.70 (s, 3H), 2.45 (s, 3H), 2.35 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 203.8, 202.9, 199.8, 137.4, 135.9, 132.5, 131.6, 130.7, 130.5, 128.8, 128.7, 127.8, 126.7, 97.5, 78.9, 67.4, 42.6, 28.5, 24.7; HRMS (ESI) calcd for C21H20ClNO4Na [M+Na]+ 408.0973, found 408.0981.
1,1'-(4-(3-溴苯甲酰)-2-甲基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3g): 白色固体, 产率79%. m.p. 125.3~126.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.87 (d, J=1.6 Hz, 1H), 7.69 (d, J=7.9 Hz, 1H), 7.60 (dd, J=8.0, 0.9 Hz, 1H), 7.36~7.27 (m, 3H), 7.25~7.12 (m, 3H), 5.41 (d, J=8.4 Hz, 1H), 3.92 (d, J=8.4 Hz, 1H), 2.71 (s, 3H), 2.37 (d, J=6.2 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 203.9, 202.8, 197.0, 138.1, 136.6, 135.9, 132.0, 130.1, 129.0, 128.9, 128.1, 127.9, 122.8, 97.5, 78.4, 64.0, 42.7, 28.3, 24.7; HRMS (ESI) calcd for C21H21BrNO4Na [M+Na]+ 452.0468, found 452.0472.
1,1'-(4-(2-氟苯甲酰)-2-甲基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3h): 白色固体, 产率78%. m.p. 153.2~154.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.68~7.58 (m, 1H), 7.48~7.36 (m, 1H), 7.30~7.22 (m, 3H), 7.20~7.07 (m, 3H), 6.99~6.89 (m, 1H), 5.45 (d, J=8.3 Hz, 1H), 3.87 (d, J=8.3 Hz, 1H), 2.69 (s, 3H), 2.40 (s, 3H), 2.35 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 203.6, 202.8, 197.4 (d, J=2.8 Hz), 161.1 (d, J=258.8 Hz), 135.0 (d, J=9.2 Hz), 131.0, 131.0, 128.8, 128.7, 128.0, 126.1 (d, J=10.2 Hz), 124.2 (d, J=3.7 Hz), 116.7 (d, J=22.7 Hz), 97.8, 78.9, 66.8, 66.8, 42.6, 28.4, 24.7; 19F NMR (376 MHz, CDCl3) δ: -111.0; HRMS (ESI) calcd for C21H20FNO4Na [M+Na]+ 392.1268, found 392.1274.
1,1'-(2-甲基-4-(4-甲基苯甲酰)-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3i): 白色固体, 产率91%. m.p. 151.2~152.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.67 (d, J=8.2 Hz, 2H), 7.32~7.23 (m, 4H), 7.2~7.16 (m, 2H), 7.11 (d, J=8.1 Hz, 2H), 5.47 (d, J=8.3 Hz, 1H), 3.97 (d, J=8.3 Hz, 1H), 2.71 (s, 3H), 2.34 (s, 6H), 2.33 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 204.2, 202.9, 197.5, 144.9, 136.3, 133.9, 129.3, 129.2, 128.9, 128.7, 128.2, 97.5, 78.3, 63.7, 42.7, 28.2, 24.7, 21.6; HRMS (ESI) calcd for C22H24NO4Na [M+Na]+ 388.1519, found 388.1523.
1,1'-(2-甲基-4-(3-甲基苯甲酰)-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3j): 白色固体, 产率83%. m.p. 111.8~112.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.56 (d, J=7.7 Hz, 1H), 7.47 (s, 1H), 7.33~7.26 (m, 4H), 7.23~7.13 (m, 3H), 5.49 (d, J=8.3 Hz, 1H), 3.88 (d, J=8.3 Hz, 1H), 2.70 (s, 3H), 2.36 (d, J=5.9 Hz, 6H), 2.23 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 204.2, 202.7, 198.2, 138.2, 136.3, 136.2, 134.6, 129.8, 128.9, 128.7, 128.4, 128.3, 126.5, 97.5, 78.5, 64.2, 42.7, 28.3, 24.7, 21.1; HRMS (ESI) calcd for C22H23NO4Na [M+Na]+ 388.1519, found 388.1529.
1,1'-(2-甲基-4-(2-甲基苯甲酰)-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3k): 白色固体, 产率83%. m.p. 145.6~147.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.46 (d, J=7.8 Hz, 1H), 7.31~7.22 (m, 4H), 7.19~7.10 (m, 3H), 7.07 (t, J=7.6 Hz, 1H), 5.37 (d, J=8.4 Hz, 1H), 3.87 (d, J=8.4 Hz, 1H), 2.70 (s, 3H), 2.42 (d, J=3.2 Hz, 6H), 2.35 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 204.0, 203.2, 201.5, 139.1, 136.9, 136.1, 132.1, 131.7, 130.3, 128.9, 128.7, 128.0, 125.7, 97.7, 79.3, 66.5, 42.7, 28.5, 24.8, 21.4; HRMS (ESI) calcd for C22H24NO4Na [M+Na]+ 388.1519, found 388.1522.
1,1'-(4-(2-甲氧基苯甲酰)-2-甲基-3-苯基异噁唑烷- 5,5-二基)双(乙-1-酮)(3l): 白色固体, 产率82%. m.p. 128.1~129.0 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.55~7.46 (m, 1H), 7.43~7.32 (m, 1H), 7.26~7.20 (m, 3H), 7.13~7.02 (m, 2H), 6.92 (t, J=7.5 Hz, 1H), 6.67 (d, J=8.4 Hz, 1H), 5.74 (d, J=7.9 Hz, 1H), 3.69 (d, J=7.9 Hz, 1H), 3.23 (s, 3H), 2.65 (s, 3H), 2.35 (d, J=15.6 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.4, 202.3, 201.1, 158.4, 137.5, 134.2, 130.8, 128.4, 128.3, 128.2, 127.7, 120.4, 111.3, 98.2, 79.0, 67.5, 54.6, 42.5, 28.2, 24.5; HRMS (ESI) calcd for C22H23NO5Na [M+Na]+ 404.1468, found 404.1479.
1,1'-(4-(3-甲氧基苯甲酰)-2-甲基-3-苯基异噁唑烷- 5,5-二基)双(乙-1-酮)(3m): 白色固体, 产率81%. m.p. 153.6~154.2 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.34 (d, J=7.7 Hz, 1H), 7.32~7.26 (m, 4H), 7.24~7.17 (m, 3H), 7.07~6.99 (m, 1H), 5.48 (d, J=8.3 Hz, 1H), 3.92 (d, J=8.3 Hz, 1H), 3.68 (s, 3H), 2.71 (s, 3H), 2.36 (d, J=4.5 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.3, 202.7, 197.8, 159.5, 137.5, 136.3, 129.5, 128.9, 128.8, 128.2, 122.0, 121.2, 112.5, 97.5, 78.4, 64.2, 55.2, 42.7, 28.3, 24.7. HRMS (ESI) calcd for C22H23NO5Na [M+Na]+ 404.1468, found 404.1472.
1,1'-(4-(4-甲氧基苯甲酰)-2-甲基-3-苯基异噁唑烷- 5,5-二基)双(乙-1-酮)(3n): 白色固体, 产率92%. m.p. 152.6~153.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.76 (d, J=8.9 Hz, 2H), 7.32~7.25 (m, 3H), 7.22~7.15 (m, 2H), 6.86~6.69 (m, 2H), 5.44 (d, J=8.3 Hz, 1H), 3.99 (d, J=8.3 Hz, 1H), 3.79 (s, 3H), 2.71 (s, 3H), 2.34 (s, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.4, 203.0, 196.0, 164.1, 136.4, 131.7, 129.4, 128.9, 128.7, 128.2, 113.7, 97.4, 78.2, 63.6, 55.4, 42.7, 28.2, 24.7; HRMS (ESI) calcd for C22H23NO5Na [M+Na]+ 404.1468, found 404.1475.
1,1'-(4-(2-萘甲酰基)-2-甲基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3o): 白色固体, 产率81%. m.p. 150.2~151.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.10 (s, 1H), 7.93~7.84 (m, 1H), 7.77 (t, J=7.5 Hz, 2H), 7.65 (d, J=8.1 Hz, 1H), 7.58~7.50 (m, 1H), 7.50~7.40 (m, 1H), 7.32~7.26 (m, 3H), 7.24~7.18 (m, 2H), 5.67 (d, J=8.3 Hz, 1H), 3.94 (d, J=8.2 Hz, 1H), 2.73 (s, 3H), 2.39 (d, J=7.3 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.4, 202.7, 197.7, 136.4, 135.8, 133.4, 132.3, 132.1, 130.0, 129.0, 128.9, 128.8, 128.3, 127.6, 126.7, 124.0, 97.6, 64.4, 42.8, 28.3, 24.7; HRMS (ESI) calcd for C25H23NO4Na [M+ Na]+ 424.1519, found 424.1526.
1,1'-(4-苯甲酰-3-(4-氟苯基)-2-甲基异噁唑烷-5,5-二基)双(乙-1-酮)(3p): 白色固体, 产率91%. m.p. 153.6~154.2 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.77 (d, J=7.5 Hz, 2H), 7.50 (t, J=7.4 Hz, 1H), 7.34 (t, J=7.8 Hz, 2H), 7.23~7.08 (m, 2H), 6.97 (t, J=8.6 Hz, 2H), 5.46 (d, J=8.3 Hz, 1H), 3.96 (d, J=8.3 Hz, 1H), 2.70 (s, 3H), 2.35 (d, J=3.4 Hz, 6H); 19F NMR (376 MHz, CDCl3) δ: -112.75; 13C NMR (100 MHz, CDCl3) δ: 204.0, 202.8, 198.0, 162.8 (d, J=247.8 Hz), 136.3, 134.0, 132.1 (d, J=3.2 Hz) 129.9 (d, J=8.2 Hz), 129.1, 128.5, 115.9 (d, J=21.6 Hz), 97.4, 63.6, 42.6, 28.3, 24.7; HRMS (ESI) calcd for C21H21F- NO4Na [M+Na]+ 392.1268, found 392.1273.
1,1'-(4-苯甲酰-3-(4-氯苯基)-2-甲基异噁唑烷-5,5-二基)双(乙-1-酮)(3q): 白色固体, 产率79%. m.p. 152.6~153.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.84~7.74 (m, 2H), 7.51 (t, J=7.4 Hz, 1H), 7.35 (t, J=7.8 Hz, 2H), 7.28~7.21 (m, 2H), 7.17~7.09 (m, 2H), 5.45 (d, J=8.3 Hz, 1H), 3.98 (d, J=8.3 Hz, 1H), 2.70 (s, 3H), 2.34 (d, J=1.7 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 203.9, 202.7, 197.9, 136.3, 134.9, 134.6, 134.0, 129.5, 129.1, 129.1, 128.6, 97.4, 63.5, 42.7, 28.3, 24.72; HRMS (ESI) calcd for C21H20ClNO4Na [M+Na]+ 408.0973, found 408.0976.
1,1'-(4-苯甲酰-3-(4-溴苯基)-2-甲基异噁唑烷-5,5-二基)双(乙-1-酮)(3r): 白色固体, 产率73%. m.p. 112.6~113.2 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.87~7.71 (m, 2H), 7.57~7.47 (m, 1H), 7.45~7.39 (m, 2H), 7.38~7.30 (m, 2H), 7.12~7.00 (m, 2H), 5.45 (d, J=8.3 Hz, 1H), 3.97 (d, J=8.3 Hz, 1H), 2.70 (s, 3H), 2.34 (d, J=1.2 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 203.9, 202.7, 197.9, 136.3, 135.4, 134.0, 132.1, 129.8, 129.2, 128.6, 122.8, 97.4, 77.5, 63.5, 42.7, 28.3, 24.7; HRMS (ESI) calcd for C21H20BrNO4Na [M+Na]+ 452.0468, found 452.0475.
1,1'-(4-苯甲酰-3-(2-氯苯基)-2-甲基异噁唑烷-5,5-二基)双(乙-1-酮)(3s): 白色固体, 产率72%. m.p. 152.6~153.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.82~7.71 (m, 2H), 7.53~7.46 (m, 1H), 7.47~7.41 (m, 1H), 7.37~7.26 (m, 3H), 7.25~7.16 (m, 2H), 5.45 (d, J=7.9 Hz, 1H), 4.61 (d, J=7.9 Hz, 1H), 2.76 (s, 3H), 2.37 (d, J=14.2 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 203.9, 202.6, 198.0, 136.6, 134.4, 134.3, 133.8, 129.6, 129.5, 129.0, 129.0, 128.5, 127.7, 97.9, 73.2, 63.1, 42.7, 28.4, 24.6; HRMS (ESI) calcd for C21H20ClNO4Na [M+Na]+ 408.0973, found 408.0978.
1,1'-(4-苯甲酰-3-(3-氯苯基)-2-甲基异噁唑烷-5,5-二基)双(乙-1-酮)(3t): 白色固体, 产率73%. m.p. 115.6~116.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.89~7.72 (m, 2H), 7.52 (t, J=7.4 Hz, 1H), 7.36 (t, J=7.8 Hz, 2H), 7.28~7.16 (m, 3H), 7.08 (d, J=7.4 Hz, 1H), 5.45 (d, J=8.3 Hz, 1H), 4.01 (d, J=8.3 Hz, 1H), 2.72 (s, 3H), 2.34 (d, J=4.4 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 203.8, 202.7, 197.8, 138.4, 136.3, 134.7, 134.0, 130.2, 129.2, 129.0, 128.6, 128.1, 126.3, 97.4, 77.4, 63.4, 42.8, 28.3, 24.7; HRMS (ESI) calcd for C21H20ClNO4Na [M+Na]+ 408.0973, found 408.0977.
1,1'-(4-苯甲酰-2-甲基-3-(对甲苯基)异噁唑烷-5,5-二基)双(乙-1-酮)(3u): 白色固体, 产率74%. m.p. 152.6~153.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.78 (d, J=7.4 Hz, 2H), 7.48 (t, J=7.4 Hz, 1H), 7.32 (t, J=7.8 Hz, 2H), 7.07 (s, 4H), 5.48 (d, J=8.3 Hz, 1H), 3.92 (d, J=8.3 Hz, 1H), 2.70 (s, 3H), 2.35 (d, J=3.0 Hz, 6H), 2.30 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 204.2, 202.9, 198.3, 138.6, 136.4, 133.8, 133.1, 129.6, 129.2, 128.5, 128.1, 97.5, 78.2, 63.8, 42.6, 28.3, 24.7, 21.1; HRMS (ESI) m/z calcd for C22H23NO4Na [M+Na]+388.1519, found 388.1526.
1,1'-(4-苯甲酰-2-甲基-3-(邻甲苯基)异噁唑烷-5,5-二基)双(乙-1-酮)(3v): 白色固体, 产率77%. m.p. 115.6~116.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.76 (d, J=7.6 Hz, 2H), 7.48 (t, J=7.4 Hz, 1H), 7.41~7.27 (m, 3H), 7.21 (t, J=7.5 Hz, 1H), 7.14 (t, J=7.4 Hz, 1H), 7.00 (d, J=7.5 Hz, 1H), 5.48 (d, J=8.3 Hz, 1H), 4.33 (d, J=8.3 Hz, 1H), 2.70 (s, 3H), 2.36 (d, J=4.0 Hz, 6H), 2.05 (s, 3H); 13C NMR (100 Hz, CDCl3) δ: 204.3, 202.8, 198.4, 136.8, 136.4, 134.4, 133.9, 130.4, 129.0, 128.5, 128.1, 127.7, 126.9, 97.7, 64.0, 42.6, 28.3, 24.7, 19.4; HRMS (ESI) calcd for C22H23NO4Na [M+Na]+ 388.1519, found 388.1528.
1,1'-(4-苯甲酰-2-甲基-3-(间甲苯基)异噁唑烷-5,5-二基)双(乙-1-酮)(3w): 白色固体, 产率95%. m.p. 145.6~146.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.78 (d, J=7.6 Hz, 2H), 7.47 (t, J=7.4 Hz, 1H), 7.31 (t, J=7.7 Hz, 2H), 7.16 (t, J=7.6 Hz, 1H), 7.11~7.04 (m, 1H), 7.01~6.93 (m, 2H), 5.49 (d, J=8.4 Hz, 1H), 3.95 (d, J=8.3 Hz, 1H), 2.71 (s, 3H), 2.35 (s, 6H), 2.27 (s, 3H); 13C NMR (100 MHz, CDCl3) δ: 204.1, 202.9, 198.2, 138.6, 136.5, 136.1, 133.8, 129.5, 129.2, 128.8, 128.7, 128.4, 125.2, 97.5, 78.4, 63.8, 42.8, 28.3, 24.8, 21.3; HRMS (ESI) calcd for C22H23NO4Na [M+Na]+ 388.1519, found 388.1523.
1,1'-(4-苯甲酰-3-(4-甲氧基苯基)-2-甲基异噁唑烷- 5,5-二基)双(乙-1-酮)(3x): 白色固体, 产率93%. m.p. 152.6~153.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.82~ 7.70 (m, 2H), 7.53~7.44 (m, 1H), 7.32 (t, J=7.8 Hz, 2H), 7.16~7.06 (m, 2H), 6.86~6.75 (m, 2H), 5.48 (d, J=8.3 Hz, 1H), 3.90 (s, 1H), 3.76 (s, 3H), 2.69 (s, 3H), 2.35 (d, J=6.5 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.2, 202.9, 198.3, 159.9, 136.4, 133.8, 129.4, 129.2, 128.5, 128.0, 114.2, 97.5, 78.0, 63.7, 55.2, 42.6, 28.3, 24.7; HRMS (ESI) calcd for C22H23NO5Na [M+Na]+ 404.1468, found 404.1471.
1,1'-(4-苯甲酰-3-(3-甲氧基苯基)-2-甲基异噁唑烷- 5,5-二基)双(乙-1-酮)(3y): 白色固体, 产率87%. m.p. 132.6~133.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.78 (d, J=7.4 Hz, 2H), 7.49 (t, J=6.5 Hz, 1H), 7.38~7.25 (m, 2H), 7.22~7.14 (m, 1H), 6.85~6.79 (m, 1H), 6.78~6.70 (m, 2H), 5.56~5.41 (m, 1H), 3.94 (d, J=7.9 Hz, 1H), 3.79~3.68 (m, 3H), 2.79~2.65 (m, 3H), 2.44~2.28 (m, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.0, 202.9, 198.2, 159.9, 137.8, 136.4, 133.8, 129.9, 129.2, 128.5, 120.4, 114.3, 113.3, 97.5, 78.2, 63.7, 55.2, 42.8, 28.3, 24.7; HRMS (ESI) calcd for C22H23NO5Na [M+Na]+ 404.1468, found 404.1473.
1,1'-(4-苯甲酰-2-甲基-3-(萘-2-基)异噁唑烷-5,5-二基)双(乙-1-酮)(3z): 白色固体, 产率81%. m.p. 137.2~138.0 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.83~7.76 (m, 4H), 7.75~7.70 (m, 1H), 7.60 (s, 1H), 7.51~7.40 (m, 3H), 7.39~7.33 (m, 1H), 7.30~7.22 (m, 2H), 5.62 (d, J=8.3 Hz, 1H), 4.17 (d, J=8.3 Hz, 1H), 2.74 (s, 3H), 2.39 (d, J=1.7 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 204.2, 202.9, 198.2, 136.4, 133.9, 133.6, 133.4, 133.1, 129.21, 129.0, 128.5, 128.0, 127.9, 127.7, 126.4, 126.4, 125.0, 97.6, 78.6, 63.5, 42.8, 28.4, 24.8; HRMS (ESI) calcd for C25H23NO4Na [M+Na]+ 424.1519, found 424.1526.
1,1'-(4-苯甲酰-2-甲基-3-(噻吩-2-基)异噁唑烷-5,5-二基)双(乙-1-酮)(3aa): 白色固体, 产率63%. m.p. 100.3~101.1 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.85 (d, J=7.6 Hz, 2H), 7.53 (t, J=7.4 Hz, 1H), 7.37 (t, J=7.8 Hz, 2H), 7.28 (s, 1H), 6.95 - 6.81 (m, 2H), 5.50 (d, J=8.0 Hz, 1H), 4.32 (d, J=8.0 Hz, 1H), 2.79 (s, 3H), 2.33 (s, 6H); 13C NMR (100 MHz, CDCl3) δ: 203.4, 202.5, 197.8, 138.9, 136.4, 134.0, 129.2, 128.6, 127.1, 126.8, 126.6, 97.4, 73.3, 64.0, 42.8, 28.2, 24.6; HRMS (ESI) calcd for C19H19NO4SNa [M+Na]+ 380.0927, found 380.0933.
1,1'-(4-苯甲酰-3-(呋喃-2-基)-2-甲基异噁唑烷-5,5-二基)双(乙-1-酮)(3ab): 白色固体, 产率77%. m.p. 135.6~136.3 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.90 (d, J=7.6 Hz, 2H), 7.54 (t, J=7.4 Hz, 1H), 7.40 (t, J=7.8 Hz, 3H), 6.30~6.25 (m, 2H), 5.73 (d, J=8.3 Hz, 1H), 4.10 (d, J=8.4 Hz, 1H), 2.81 (s, 3H), 2.32 (d, J=11.6 Hz, 6H); 13C NMR (100 MHz, CDCl3) δ: 203.4, 197.7, 148.1, 143.5, 136.3, 134.0, 129.2, 128.6, 110.5, 110.2, 97.5, 70.9, 59.7, 43.1, 28.2, 24.6; HRMS (ESI) calcd for C19H19N- O5Na [M+Na]+ 364.1155, found 364.1162.
1,1'-(4-苯甲酰-2-苄基-3-苯基异噁唑烷-5,5-二基)双(乙-1-酮)(3ac): 白色固体, 产率94%. m.p. 134.7~135.3 ℃; 1H NMR (400 MHz, DMSO-d6) δ: 7.66 (d, J=7.3 Hz, 2H), 7.60 (t, J=7.4 Hz, 1H), 7.41 (t, J=7.8 Hz, 2H), 7.38~7.30 (m, 7H), 7.28~7.23 (m, 2H), 5.37 (d, J=8.3 Hz, 1H), 4.20 (d, J=8.3 Hz, 1H), 3.87 (q, J=14.5 Hz, 2H), 2.30 (s, 3H), 1.98 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ: 204.2, 203.0, 198.4, 137.0, 136.7, 136.6, 134.6, 129.5, 129.9, 129.3, 129.2, 128.9, 128.6, 128.3, 127.9, 97.5, 76.0, 63.3, 59.0, 28.77, 24.9; HRMS (ESI) calcd for C27H26NO4Na [M+Na]+ 450.1676, found 450.1684.