

咪唑类化合物的合成研究进展
收稿日期: 2025-07-20
修回日期: 2025-09-01
网络出版日期: 2025-10-15
基金资助
国家自然科学基金(22201012)
陕西省创新能力支撑计划——青年科技新星项目(2024ZC-KJXX-039)
陕西高校优秀青年人才支持计划(2025QJ-02(内))
Progress in the Synthesis of Imidazole Compounds
Received date: 2025-07-20
Revised date: 2025-09-01
Online published: 2025-10-15
Supported by
National Natural Science Foundation of China(22201012)
Innovation Capability Support Program of Shaanxi—Young Science and Technology Star Project(2024ZC-KJXX-039)
Support Program for Outstanding Young Talents in Shaanxi Universities(2025QJ-02(内))
赵咪娜 , 唐嘉一 . 咪唑类化合物的合成研究进展[J]. 有机化学, 2026 , 46(2) : 455 -474 . DOI: 10.6023/cjoc202507027
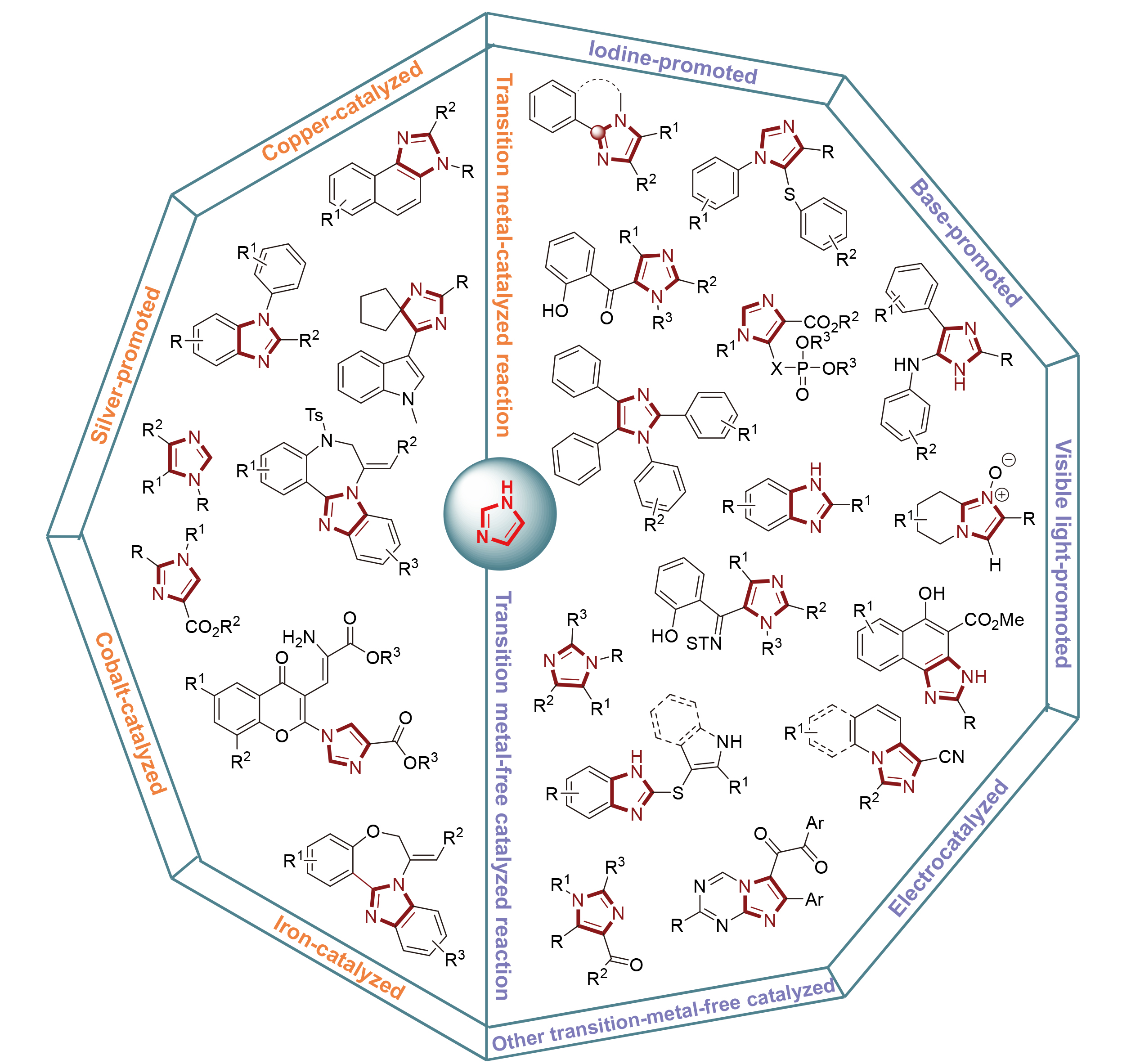
Imidazole compounds are a class of substances with important structural skeletons, which are not only the components of histidine, but also the components of ribonucleic acid and purine of DNA. Imidazole itself has a special “acid-base amphoteric” aromatic heterocycle structure and good electron transfer ability, so it is widely used in the fields of medicine and materials. Based on the research reports in the past five years, the common synthesis strategies are summarized according to the types of catalysts, and the future development trend of this field is prospected.
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
(赵咪娜, 杨梓墨, 杨得锁, 有机化学, 2022, 42, 111.)
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
(刘梦琴, 陈贻庭, 张榉元, 周和烨, 秦涛, 刘彬, 有机化学, 2025, 45, 1283.)
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
/
| 〈 |
|
〉 |