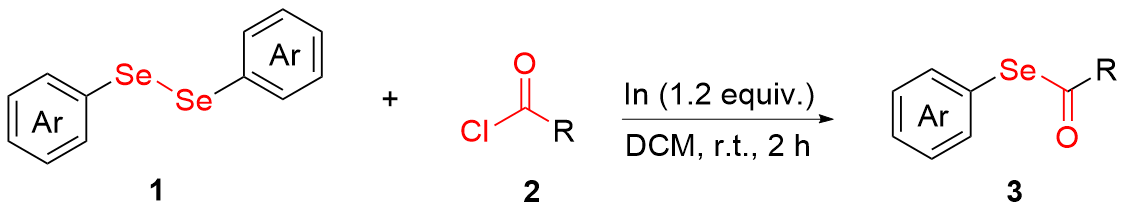
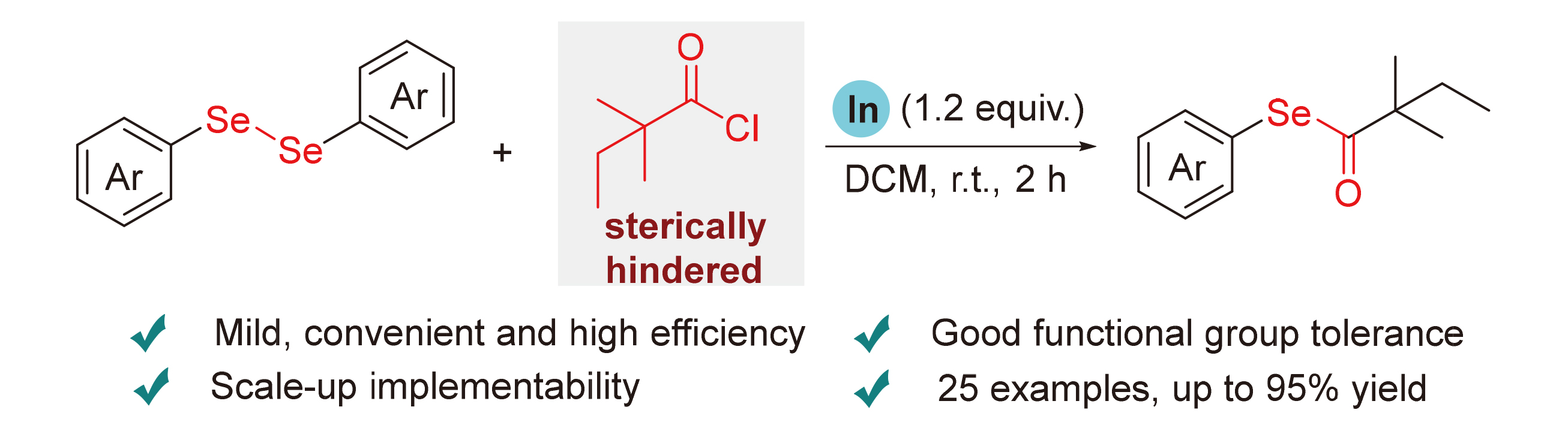
To a solution of 1 (0.2 mmol, 1.0 equiv.) and indium (0.24 mmol, 325 mesh powder, 1.2 equiv.) in dried DCM (1.0 mL) in a 15 mL round bottom flask, 2 (0.40 mmol, 2.0 equiv) was slowly added under N2 atmosphere. The mixture was stirred at room temperature for 2 h. After completion of the reaction (as monitored by TLC), the reaction mixture was diluted with 10 mL of DCM, then successively washed with water and brine (15 mL×3), dried over anhydrous sodium sulfate, and filtered. The solvent was evaporated under vacuum. Purification was performed by a column chromatography on silica gel (eluents: petroleum ether/ethyl acetate, V∶V=60∶1) to afford the desired compounds 3.
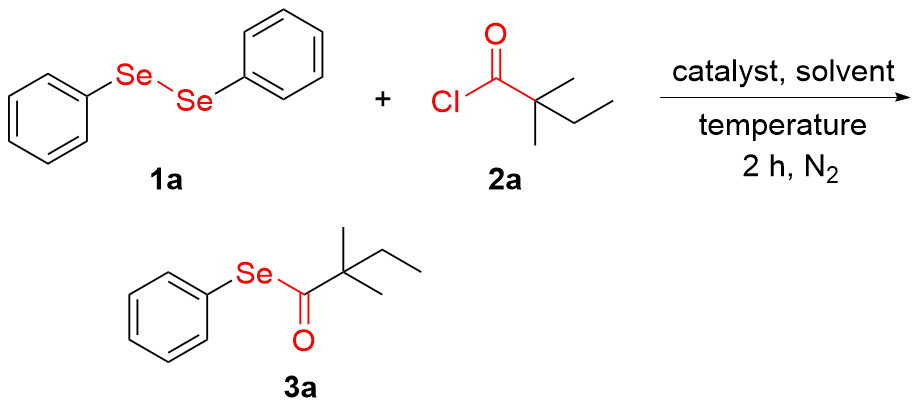
Se-Phenyl-2,2-dimethylbutaneselenoate (3a): Colorless oil, 48.6 mg, 95% yield. 1H NMR (400 MHz, CDCl3) δ: 7.50~7.48 (m, 2H), 7.39~7.36 (m, 3H), 1.72 (q, J=7.6 Hz, 2H), 1.27 (s, 6H), 0.96 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.8, 136.5 (2C), 129.3 (2C), 128.8, 126.4, 53.5, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C12H17OSe [M+H]+ 257.0445, found 257.0443
Se-(o-Tolyl)-2,2-dimethylbutaneselenoate (3b): Colorless oil, 46.5 mg, 86% yield. 1H NMR (400 MHz, CDCl3) δ: 7.49 (d, J=7.6 Hz, 1H), 7.29 (d, J=5.2 Hz, 2H), 7.15~7.12 (m, 1H), 2.35 (s, 3H), 1.70 (q, J=8.4 Hz, 2H), 1.24 (s, 6H), 0.94 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.3, 142.5, 138.0, 130.3, 129.6, 127.4, 126.6, 53.5, 33.6, 24.6 (2C), 23.0, 9.1; HRMS (ESI) calcd for C13H19OSe [M+H]+ 271.0601, found 271.0584
Se-(2-Chlorophenyl)-2,2-dimethylbutaneselenoate (3c): Colorless oil, 42.9 mg, 74% yield. 1H NMR (400 MHz, CDCl3) δ: 7.60 (d, J=6.8 Hz, 1H), 7.52 (d, J=8.0 Hz, 1H), 7.36 (t, J=8.8 Hz, 1H), 7.26~7.22 (m, 1H), 1.73 (q, J=7.6 Hz, 2H), 1.28 (s, 6H), 0.97 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 205.9, 139.5, 139.1, 130.7, 129.9, 127.3, 127.2, 53.7, 33.6, 24.5 (2C), 9.1; HRMS (ESI) calcd for C12H16ClOSe [M+H]+ 291.0055, found 291.0038
Se-(2-bromophenyl)-2,2-dimethylbutaneselenoate (3d): Colorless oil, 50.8 mg, 76% yield. 1H NMR (400 MHz, CDCl3) δ: 7.64 (s, 1H), 7.52 (d, J=8.0 Hz, 1H), 7.42 (d, J=7.6 Hz, 1H), 7.26 (td, J=8.0, 2.0 Hz, 1H), 1.70~1.65 (m, 2H), 1.26 (d, J=1.2 Hz, 6H), 0.95 (td, J=7.6, 1.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 206.8, 138.8, 135.0, 131.8, 130.5, 128.1, 122.7, 53.7, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C12H16BrOSe [M+H]+ 334.9550, found 334.9543
Se-(m-Tolyl)-2,2-dimethylbutaneselenoate (3e): Colorless oil, 48.1 mg, 89% yield. 1H NMR (400 MHz, CDCl3) δ: 7.33 (s, 1H), 7.30 (d, J=6.8 Hz, 2H), 7.22 (d, J=6.8 Hz, 1H), 2.38 (s, 3H), 1.73 (q, J=7.6 Hz, 2H), 1.28 (s, 6H), 0.97 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 208.1, 139.1, 137.1, 133.5, 129.6, 129.1, 126.1, 53.6, 33.6, 24.7 (2C), 21.4, 9.1; HRMS (ESI) calcd for C13H19O- Se [M+H]+ 271.0601, found 271.0584
Se-(3-Isopropylphenyl)-2,2-dimethylbutaneselenoate (3f): Colorless oil, 54.3 mg, 91% yield. : 1H NMR (400 MHz, CDCl3) δ: 7.40 (d, J=8.0 Hz, 2H), 7.25 (d, J=8.0 Hz 2H), 2.97~2.87 (m, 1H), 1.70 (q, J=7.6 Hz, 2H), 1.27 (d, J=6.0 Hz, 12H), 0.95 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 208.4, 149.7, 136.5 (2C), 127.6 (2C), 123.2, 53.5, 34.1, 33.7, 24.7 (2C), 24.0 (2C), 9.1; HRMS (ESI) calcd for C15H23OSe [M+H]+ 299.0914, found 299.0918
Se-(3-(tert-butyl)phenyl)-2,2-dimethylbutaneselenoate (3g): Colorless oil, 51.8 mg, 83% yield. 1H NMR (400 MHz, CDCl3) δ: 7.48 (s, 1H), 7.41~7.38 (m, 1H), 7.32~7.28 (m, 2H), 1.71 (q, J=7.6 Hz, 2H), 1.33 (s, 9H), 1.26 (s, 6H), 0.95 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.9, 152.2, 133.5, 133.4, 128.9, 126.1, 125.9, 53.6, 34.9, 33.7, 31.4 (3C), 24.7 (2C), 9.1; HRMS (ESI) calcd for C16H25OSe [M+H]+ 313.1071, found 313.1097
Se-(3-(Trifluoromethyl)phenyl)-2,2-dimethylbutaneselenoate (3h): Colorless oil, 52.5 mg, 81% yield. 1H NMR (400 MHz, CDCl3) δ: 7.75 (s, 1H), 7.67 (t, J=8.4 Hz, 2H), 7.50 (t, J=8.0 Hz, 1H), 1.72 (q, J=7.2 Hz, 2H), 1.27 (s, 6H), 0.96 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 208.5, 137.8, 137.6, 137.5, 134.0, 130.6, 122.9, 53.5, 33.7, 24.7 (2C), 9.0; HRMS (ESI) calcd for C13H16F3OSe [M+H]+ 325.0318, found 325.0311
Se-(3-Fluorophenyl)-2,2-dimethylbutaneselenoate (3i): Colorless oil, 47.7 mg, 87% yield. 1H NMR (400 MHz, CDCl3) δ: 7.36~7.30 (m, 1H), 7.26~7.21 (m, 2H), 7.09~7.05 (m, 1H), 1.70 (q, J=7.6 Hz, 2H), 1.25 (s, 6H), 0.94 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.0, 163.8 (d, 1JCF=247.8 Hz), 132.1 (d, 4JCF=3.1 Hz), 130.4 (d, 3JCF=7.9 Hz), 127.8 (d, 3JCF=7.2 Hz), 123.5 (d, 2JCF=21.8 Hz), 116.0 (d, 2JCF=20.8 Hz), 53.7, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C12H16FOSe [M+H]+ 275.0350, found 275.0369
Se-(3-Chlorophenyl)-2,2-dimethylbutaneselenoate (3j): Colorless oil, 47.6 mg, 82% yield. 1H NMR (400 MHz, CDCl3) δ: 7.48 (s, 1H), 7.36 (d, J=7.6 Hz, 2H), 7.29 (t, J=7.6 Hz, 1H), 1.69 (q, J=7.6 Hz, 2H), 1.24 (s, 6H), 0.94 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 206.8, 136.1, 134.6, 134.5, 130.2, 129.0, 127.8, 53.7, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C12H16ClOSe [M+H]+ 291.0055, found 291.0038
Se-(3-Bromophenyl)-2,2-dimethylbutaneselenoate (3k): Colorless oil, 52.1 mg, 78% yield. 1H NMR (400 MHz, CDCl3) δ: 7.64 (t, J=1.6 Hz, 1H), 7.52~7.49 (m, 1H), 7.41(dt, J=7.6, 1.2 Hz, 1H), 7.25 (t, J=8.0 Hz, 1H), 1.70 (q, J=7.2 Hz, 2H), 1.25 (s, 6H), 0.94 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 206.9, 138.8, 135.0, 131.8, 130.6, 128.1, 122.7, 53.7, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C12H16BrOSe [M+H]+ 334.9550, found 334.9574
Se-(p-Tolyl)-2,2-dimethylbutaneselenoate (3l): Colorless oil, 48.6 mg, 90% yield. 1H NMR (400 MHz, CDCl3) δ: 7.36 (dt, J=8.4, 2.0 Hz, 2H), 7.17 (d, J=7.6 Hz, 2H), 2.35 (s, 3H), 1.68 (q, J=7.6 Hz, 2H), 1.23 (s, 6H), 0.92 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 208.1, 138.8, 136.4 (2C), 130.1 (2C), 122.7, 53.4, 33.6, 24.6 (2C), 21.4, 9.1; HRMS (ESI) calcd for C13H19OSe [M+H]+ 271.0601, found 271.0584
Se-(4-(tert-Butyl)phenyl)-2,2-dimethylbutaneselenoate (3m): Colorless oil, 53.7 mg, 86% yield. 1H NMR (400 MHz, CDCl3) δ: 7.40 (s, 4H), 1.70 (q, J=7.6 Hz, 2H), 1.32 (s, 9H), 1.25 (s, 6H), 0.94 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 208.5, 151.9, 136.2 (2C), 126.5 (2C), 122.9, 53.5, 34.8, 33.7, 31.4 (3C), 24.7 (2C), 9.1; HRMS (ESI) calcd for C16H25OSe [M+H]+ 313.1071, found 313.1060
Se-(4-(Trifluoromethoxy)phenyl)-2,2-dimethylbutanese-lenoate (3n): Colorless oil, 60.5 mg, 89% yield. 1H NMR (400 MHz, CDCl3) δ: 7.50 (d, J=8.4 Hz, 2H), 7.22 (d, J=8.4 Hz, 2H), 1.70 (q, J=7.2 Hz, 2H), 1.25 (s, 6H), 0.94 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.3, 149.7, 138.0 (2C), 124.7, 121.7 (2C), 119.3, 53.7, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C13H16F3O2Se [M+H]+ 341.0268; found:341.0276
Se-(4-(Trifluoromethoxy)phenyl)-2,2-dimethylbutanese-lenoate (3o): Colorless oil, 51.6 mg, 89% yield. 1H NMR (400 MHz, CDCl3) δ: 7.40 (d, J=8.4 Hz, 2H), 7.34 (d, J=8.4 Hz, 2H), 1.70 (q, J=7.6 Hz, 2H), 1.25 (s, 6H), 0.93 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.3, 137.8 (2C), 135.3, 129.5 (2C), 124.6, 53.7, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C12H16ClOSe [M+H]+ 291.0055, found 291.0068
Se-(4-Bromophenyl)-2,2-dimethylbutaneselenoate (3p): Colorless oil, 55.4 mg, 83% yield. 1H NMR (400 MHz, CDCl3) δ: 7.50 (dd, J1=8.4 Hz, J2=2.4 Hz, 2H), 7.34 (dd, J1=8.4 Hz, J2=2.4 Hz, 2H), 1.69~1.63 (m, 2H), 1.24 (d, J=2.0 Hz, 6H), 0.94 (td, J1=10.0 Hz, J2=2.4Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.2, 138.0 (2C), 132.5 (2C), 125.3, 123.5, 53.7, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C12H16BrOSe [M+H]+ 334.9550, found 334.9532
Se-(3,5-Dimethylphenyl)-2,2-dimethylbutaneselenoate (3q): Colorless oil, 49.4 mg, 87% yield. 1H NMR (400 MHz, CDCl3) δ: 7.11 (s, 2H), 7.00 (s, 1H), 2.32 (s, 6H), 1.70 (q, J=7.6 Hz, 2H), 1.25 (s, 6H), 0.95 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 208.4, 138.9, 134.1 (2C), 130.7 (2C), 125.7, 53.5, 33.7, 24.7 (2C), 21.3 (2C), 9.1; HRMS (ESI) calcd for C14H21OSe [M+H]+ 285.0758, found 285.0737
Se-(4-chloro-3-methylphenyl)-2,2-dimethylbutanesele-noate (3r): Colorless oil, 50.5 mg, 83% yield. 1H NMR (400 MHz, CDCl3) δ: 7.34 (d, J=10.4 Hz, 2H), 7.24 (d, J=8.0 Hz, 1H), 2.38 (s, 3H), 1.70 (q, J=7.2 Hz,2H), 1.25 (s, 6H), 0.99 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.6, 138.7, 137.2, 135.5, 135.1, 129.8, 124.3, 53.6, 33.6, 24.6 (2C), 20.1, 9.1; HRMS (ESI) calcd for C13H18ClOSe [M+H]+ 305.0211, found 305.0208
Se-(Naphthalen-1-yl)-2,2-dimethylbutaneselenoate (3s): Colorless oil, 25.7 mg, 42% yield. 1H NMR (400 MHz, CDCl3) δ: 8.19 (d, J=7.6 Hz, 1H), 7.94 (d, J=8.4 Hz, 1H), 7.87~7.78 (m, 2H), 7.55~7.44 (m, 3H), 1.77 (q, J=7.6 Hz,2H), 1.32 (s, 6H), 0.99 (t, J=7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.2, 137.0, 135.2, 134.2, 133.1, 130.4, 128.8, 127.9, 127.0, 126.3, 126.0, 53.8, 33.7, 24.7 (2C), 9.2; HRMS (ESI) calcd for C16H19OSe [M+ H]+ 307.0601, found 307.0591
Se-(Naphthalen-2-yl)-2,2-dimethylbutaneselenoate (3t): Colorless oil, 41.0 mg, 67% yield. 1H NMR (400 MHz, CDCl3) δ: 8.04 (s, 1H), 7.87 (t, J=8.4 Hz, 3H), 7.55~7.50 (m, 3H), 1.75 (q, J=7.6 Hz,2H), 1.30 (s, 6H), 0.99 (t, J=7.6 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ: 207.9, 136.2, 134.0, 133.1 (2C), 128.6, 127.9 (2C), 126.8, 126.4, 123.8, 53.6, 33.6, 24.6 (2C), 9.1; HRMS (ESI) calcd for C16H19O- Se [M+H]+ 307.0601, found 307.0590
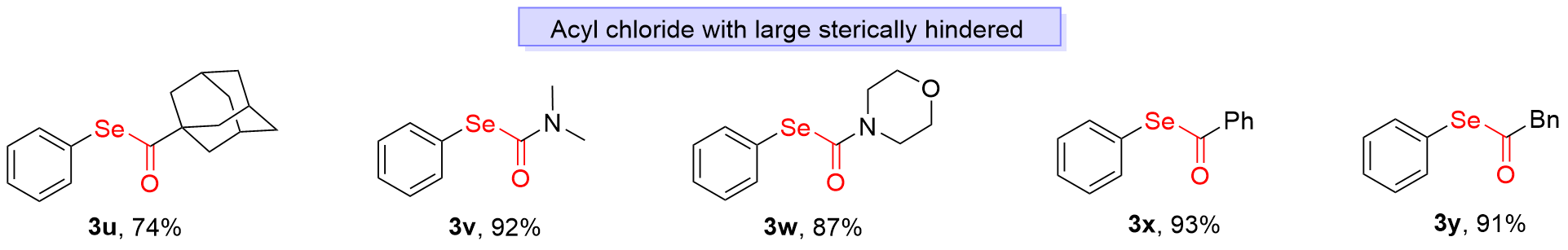
Se-Phenyl (3R,5R,7R)-adamantane-1-carboselenoate (3u): Colorless oil, 47.4 mg, 74% yield. 1H NMR (400 MHz, CDCl3) δ: 7.50~7.48 (m, 2H), 7.38~7.34 (m, 3H), 2.11 (s, 3H), 2.00 (d, J=2.4 Hz, 6H), 1.80 (m, 6H); 13C NMR (100 MHz, CDCl3) δ: 207.8, 136.5 (2C), 129.2 (2C), 128.7, 126.2, 52.1, 39.2 (3C), 36.5 (3C), 28.3 (3C); HRMS (ESI) calcd for C17H21OSe [M+H]+ 321.0758, found 321.0746
Se-Phenyl dimethylcarbamoselenoate (3v): Colorless oil, 42.1 mg, 92% yield. 1H NMR (400 MHz, CDCl3) δ: 7.62 (dd, J1=7.6 Hz, J2=1.6 Hz, 2H), 7.41~7.34 (m, 3H), 3.01 (s, 6H); 13C NMR (100 MHz, CDCl3) δ: 164.3, 136.6 (2C), 129.0 (2C), 128.7, 126.8, 37.2, 36.8; HRMS (ESI) calcd for C9H12NOSe [M+H]+ 230.0084, found 230.0076
Se-Phenyl morpholine-4-carboselenoate (3w): Colorless oil, 47.2 mg, 87% yield. 1H NMR (400 MHz, CDCl3) δ: 7.61 (d, J=6.4 Hz, 2H), 7.43~7.35 (m, 3H), 3.70~3.46 (m, 8H); 13C NMR (100 MHz, CDCl3) δ: 163.9, 136.8 (2C), 129.2 (2C), 129.1, 126.2, 66.6 (4C); HRMS (ESI) calcd for C11H14NO2Se [M+H]+ 272.0190, found 272.0201
Se-Phenyl benzoselenoate (3x): Colorless oil, 48.7 mg, 93% yield. 1H NMR (400 MHz, CDCl3) δ: 7.96 (d, J=7.2 Hz, 2H), 7.63~7.60 (m, 2H), 7.52 (t, J=8.0 Hz, 3H), 7.45~7.42 (m, 3H); 13C NMR (100 MHz, CDCl3) δ: 193.5, 138.6, 136.5 (2C), 134.0, 129.5 (2C), 129.2, 129.1 (2C), 127.5 (2C), 125.9; HRMS (ESI) calcd for C13H11OSe [M+H]+ 262.9975, found 262.9973
Se-Phenyl 2-phenylethaneselenoate (3y): Colorless oil, 50.2 mg, 91% yield. 1H NMR (400 MHz, CDCl3) δ: 7.49~7.47 (m, 2H), 7.41~7.33 (m, 8H), 3.94 (s, 2H); 13C NMR (100 MHz, CDCl3) δ: 198.9, 135.9 (2C), 132.7, 130.2 (2C), 129.4 (2C), 129.0, 128.9 (2C), 128.0, 126.7, 53.7; HRMS (ESI) calcd for C14H13OSe [M+H]+ 277.0132, found 277.0134.