To a dry two-necked 250 mL flask were added iodobenzene (4.1 g, 20 mmol), CuI (152 mg, 0.8 mmol) and PdCl2(PPh3)2 (140 mg, 0.2 mmol) under an argon atmosphere. Then Et3N (67 mL) and propyne (24 mL, 1.0 mol/L in THF, 24 mmol) were added to the system. The reaction mixture was stirred at 25 ℃ for 6 h. After completion of the reaction as indicated by thin layer chromatography (TLC), it was quenched with saturated aqueous NH4Cl, extracted with EtOAc (5 mL×3), washed with brine, dried over anhydrous Na2SO4, filtered and concentrated. The residue was purified by chromatography on silica gel (petroleum ether) to afford prop-1-yn-1-ylbenzene (2.0 g, 85% yield) as colorless oil.
To a solution of prop-1-yn-1-ylbenzene (2.0 g, 17.2 mmol) and dry CHCl3 (2.8 mL, 34.4 mmol) in dry THF (17 mL) at -78 ℃ was added dropwise nBuLi (10.4 mL, 2.5 mol/L in hexane, 25.8 mmol) for 1 h under an argon atmosphere. The reaction mixture was then stirred for 4 h at room temperature. Then the mixture was quenched with HCl (ω=37%, 10 mL), extracted with CH2Cl2 (10 mL×3), washed with brine, dried over anhydrous Na2SO4, filtered and concentrated. The residue was purified by chromatography (petroleum ether/ethyl acetate, V∶V=1∶1) to afford 1a (248 mg, 10% yield) as brown solid. Compounds 1b~1i, 1k, 1l, 1n, 1p~1t were prepared using the same method.
Substrates
1a,
[11a] 1b,
[11b] 1k[11c] are known compounds.
2-(4-(tert-Butyl)phenyl)-3-methylcycloprop-2-en-1-one (1c): Crystalline brown solid, 482 mg, 14% yield. m.p. 91~94 ℃; 1H NMR (600 MHz, CDCl3) δ: 7.66 (d, J=8.4 Hz, 2H), 7.48 (d, J=8.4 Hz, 2H), 2.41 (s, 3H), 1.28 (s, 9H); 13C NMR (151 MHz, CDCl3) δ: 156.7, 156.3, 154.5, 150.2, 130.9, 126.2, 121.0, 35.2, 31.0, 11.5; IR (KBr) ν: 1848, 1628, 1362, 1328, 1106, 846, 827, 682, 567 cm-1; HRMS (ESI) calcd for C14H16ONa [M+Na]+ 223.1093, found 223.1094.
2-(4-Chlorophenyl)-3-methylcycloprop-2-en-1-one (1d): Crystalline brown solid, 399 mg, 13% yield. m.p. 99~103 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.67 (d, J=8.2 Hz, 2H), 7.46 (d, J=8.2 Hz, 2H), 2.45 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 156.4, 153.7, 152.1, 138.9, 132.3, 129.8, 122.1, 11.7; IR (KBr) ν: 1847, 1630, 1587, 1400, 1089, 1011, 825, 680, 550, 459 cm-1; HRMS (ESI) calcd for C10H7OClNa [M+Na]+ 201.0078, found 201.0076.
2-(3-Chlorophenyl)-3-methylcycloprop-2-en-1-one (1e): Crystalline brown solid, 369 mg, 12% yield. m.p. 85~87 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.67 (s, 1H), 7.64~7.57 (m, 1H), 7.50~7.36 (m, 2H), 2.45 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 156.2, 153.6, 153.5, 135.2, 132.5, 130.63, 130.59, 129.1, 125.0, 11.8; IR (KBr) ν: 1863, 1831, 1639, 1467, 1426, 1313, 1158, 1067, 872, 813, 749, 690, 580 cm-1; HRMS (ESI) calcd for C10H7OClNa [M+Na]+ 201.0078, found 201.0072.
2-(3-Bromophenyl)-3-methylcycloprop-2-en-1-one (1f): Crystalline brown solid, 230 mg, 6% yield. m.p. 85~88 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.84 (s, 1H), 7.65 (dd, J=14.2, 7.6 Hz, 2H), 7.36 (t, J=7.9 Hz, 1H), 2.47 (s, 3H);13C NMR (101 MHz, CDCl3) δ: 156.3, 153.6, 153.5, 135.4, 133.6, 130.9, 129.6, 125.3, 123.2, 11.8; IR (KBr) ν: 1813, 1631, 1584, 1555, 1410, 1324, 1068, 877, 788, 730, 686, 571 cm-1; HRMS (ESI) calcd for C10H7OBrNa [M+Na]+ 244.9572, found 244.9570.
2-([1'-Biphenyl]-4-yl)-3-methylcycloprop-2-en-1-one (1g): Crystalline brown solid, 379 mg, 10% yield. m.p. 142~145 ℃; 1H NMR (600 MHz, CDCl3) δ: 7.80 (d, J=8.1 Hz, 2H), 7.70 (d, J=8.1 Hz, 2H), 7.59 (d, J=7.4 Hz, 2H), 7.44 (t, J=7.4 Hz, 2H), 7.38 (t, J=7.4 Hz, 1H), 2.46 (s, 3H); 13C NMR (151 MHz, CDCl3) δ: 156.7, 154.3, 151.2, 145.1, 139.5, 131.5, 129.0, 128.4, 127.8, 127.2, 122.4, 11.6; IR (KBr) ν: 1843, 1626, 1484, 1397, 1329, 1004, 842, 773, 730, 698, 565, 465 cm-1; HRMS (ESI) calcd for C16H12ONa [M+Na]+ 243.0780, found 243.0782.
2-Methyl-3-(thiophen-3-yl)cycloprop-2-en-1-one (1h): Crystalline brown solid, 129 mg, 5% yield. m.p. 38~40 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.02~7.96 (m, 1H), 7.44~7.31 (m, 2H), 2.34 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 155.4, 148.6, 147.4, 134.1, 127.7, 127.4, 124.5, 11.4; IR (KBr) ν: 3426, 3086, 1845, 1605, 1504, 1415, 1308, 1212, 860, 804, 605 cm-1; HRMS (ESI) calcd for C8H6OSNa M+Na]+ 173.0032, found 173.0030.
2-(4-Chloro-3-methylphenyl)-3-methylcycloprop-2-en-1-one (1i): Crystalline white solid, 497 mg, 15% yield. m.p. 111~114 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.57 (s, 1H), 7.48~7.43 (m, 1H), 7.43~7.38 (m, 1H), 2.42 (s, 3H), 2.35 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 156.5, 153.8, 151.7, 139.0, 137.5, 133.4, 130.0, 129.4, 122.0, 20.0, 11.7; IR (KBr) ν: 1845, 1623, 1473, 1399, 1316, 1278, 1136, 1044, 906, 819, 711, 579 cm-1; HRMS (ESI) calcd for C11H9O- ClNa [M+Na]+ 215.0234, found 215.0230.
2-Butyl-3-(naphthalen-1-yl)cycloprop-2-en-1-one (1l): Crystalline brown solid, 569 mg, 14% yield. m.p. 69~72 ℃; 1H NMR (400 MHz, CDCl3) δ: 8.64 (d, J=8.4 Hz, 1H), 8.03 (d, J=8.2 Hz, 1H), 7.95~7.85 (m, 2H), 7.72 (t, J=7.6 Hz, 1H), 7.63~7.53 (m, 2H), 2.93 (t, J=7.3 Hz, 2H), 1.93~1.78 (m, 2H), 1.59~1.46 (m, 2H), 0.98 (t, J=7.4 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 157.2, 154.4, 152.9, 133.7, 133.5, 132.7, 130.1, 128.5, 128.3, 127.2, 126.2, 125.3, 121.8, 28.7, 26.5, 22.6, 13.8; IR (KBr) ν: 2956, 1828, 1612, 1504, 1409, 1345, 1243, 804, 783, 732, 663, 617, 479 cm-1; HRMS (ESI) calcd for C17H16ONa [M+Na]+ 259.1093, found 259.1094.
2-(4-Chlorophenyl)-3-(3-chloropropyl)cycloprop-2-en-1-one (1n): Crystalline brown solid, 290 mg, 7% yield. m.p. 56~59 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.74 (d, J=8.3 Hz, 2H), 7.52 (d, J=8.3 Hz, 2H), 3.76 (t, J=6.2 Hz, 2H), 3.07 (t, J=7.1 Hz, 2H), 2.39~2.25 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 156.0, 154.0, 153.4, 138.8, 132.2, 129.5, 121.6, 43.6, 28.9, 23.8; IR (KBr) ν: 2944, 1842, 1624, 1585, 1482, 1402, 1291, 1088, 826, 720, 535 cm-1; HRMS (ESI) calcd for C12H10OCl2Na [M+Na]+ 263.0001, found 262.9999.
2-(4-Chlorophenyl)-3-(3-phenylpropyl)cycloprop-2-en- 1-one (1p): Crystalline brown solid, 632 mg, 13% yield. m.p. 42~45 ℃; 1H NMR (600 MHz, CDCl3) δ: 7.54 (d, J=8.3 Hz, 2H), 7.34 (d, J=8.3 Hz, 2H), 7.21~7.13 (m, 2H), 7.11~7.04 (m, 3H), 2.72~2.62 (m, 4H), 2.06~1.97 (m, 2H); 13C NMR (151 MHz, CDCl3) δ: 156.1, 155.3, 152.7, 140.2, 138.3, 132.0, 129.3, 128.2, 128.1, 125.9, 121.6, 34.7, 27.7, 25.5; IR (KBr) ν: 2928, 1837, 1623, 1480, 1400, 1289, 1084, 1009, 829, 745, 696, 534 cm-1; HRMS (ESI) calcd for C18H15OClNa [M+Na]+ 305.0704, found 305.0705.
2-(3-Chlorophenyl)-3-(3-(methoxymethoxy)propyl)cyclo-prop-2-en-1-one (1q): Brown liquid, 367 mg, 8% yield. 1H NMR (400 MHz, CDCl3) δ: 7.71 (s, 1H), 7.67~7.60 (m, 1H), 7.50~7.46 (m, 1H), 7.46~7.39 (m, 1H), 4.58 (s, 2H), 3.63 (t, J=5.9 Hz, 2H), 3.30 (s, 3H), 2.93 (t, J=7.3 Hz, 2H), 2.13~2.03 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 156.8, 156.6, 153.3, 135.3, 132.5, 130.8, 130.7, 129.3, 125.0, 96.5, 66.2, 55.4, 26.8, 23.8; IR (KBr) ν: 3446, 2932, 1839, 1622, 1587, 1148, 1107, 1033, 916, 793, 690 cm-1; HRMS (ESI) calcd for C14H15O3ClNa [M+Na]+ 289.0602, found 289.0601.
2-(3-(Benzyloxy)propyl)-3-(3-chlorophenyl)cycloprop-2-en-1-one (1r): Brown liquid, 538 mg, 10% yield. 1H NMR (400 MHz, CDCl3) δ: 7.74 (t, J=1.6 Hz, 1H), 7.65 (t, J=7.6 Hz, 1H), 7.54~7.49 (m, 1H), 7.46~7.41 (m, 1H), 7.34~7.25 (m, 5H), 4.51 (s, 2H), 3.62 (t, J=5.9 Hz, 2H), 2.97 (t, J=7.2 Hz, 2H), 2.17~2.07 (m, 2H); 13C NMR (151 MHz, CDCl3) δ: 156.9, 156.6, 153.2, 138.0, 135.2, 132.4, 130.7, 130.6, 129.2, 128.4, 127.6, 125.0, 73.1, 68.5, 26.7, 23.7; IR (KBr) ν: 2952, 1842, 1714, 1587, 1272, 1071, 788, 711 cm-1; HRMS (ESI) calcd for C19H17O2ClNa [M+Na]+ 335.0809, found 335.0805.
2-(2-(1,3-Dioxolan-2-yl)ethyl)-3-(3-chlorophenyl)cyclo-prop-2-en-1-one (1s): Brown liquid, 137 mg, 3% yield. 1H NMR (400 MHz, CDCl3) δ: 7.76 (s, 1H), 7.67 (d, J=7.4 Hz, 1H), 7.55~7.49 (m, 1H), 7.49~7.43 (m, 1H), 5.01 (t, J=4.0 Hz, 1H), 4.03~3.93 (m, 2H), 3.93~3.83 (m, 2H), 2.96 (t, J=7.3 Hz, 2H), 2.27~2.14 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 156.8, 156.6, 153.1, 135.4, 132.6, 131.0, 130.7, 129.4, 125.1, 102.7, 65.3, 30.4, 20.9; IR (KBr) ν: 3398, 1841, 1622, 1471, 1264, 1148, 1108, 1033, 916, 793, 732, 690 cm-1; HRMS (ESI) calcd for C14H13O3ClNa [M+Na]+ 287.0445, found 287.0440.
2-Cyclohexyl-3-phenylcycloprop-2-en-1-one (1t): Crystalline white solid, 402 mg, 11% yield. m.p. 49~52 ℃; 1H NMR (400 MHz, CDCl3) δ: 7.82~7.73 (m, 2H), 7.58~7.45 (m, 3H), 3.04~2.91 (m, 1H), 2.09~2.00 (m, 2H), 1.83~1.58 (m, 5H), 1.53~1.32 (m, 3H); 13C NMR (151 MHz, CDCl3) δ: 158.1, 157.0, 152.5, 131.9, 130.8, 128.7, 123.1, 35.9, 29.6, 25.1, 24.4; IR (KBr) ν: 2930, 2849, 1838, 1622, 1444, 1290, 1257, 1161, 1018, 897, 768, 690, 600, 466 cm-1; HRMS (ESI) calcd for C15H16ONa [M+Na]+ 235.1093, found 235.1095.
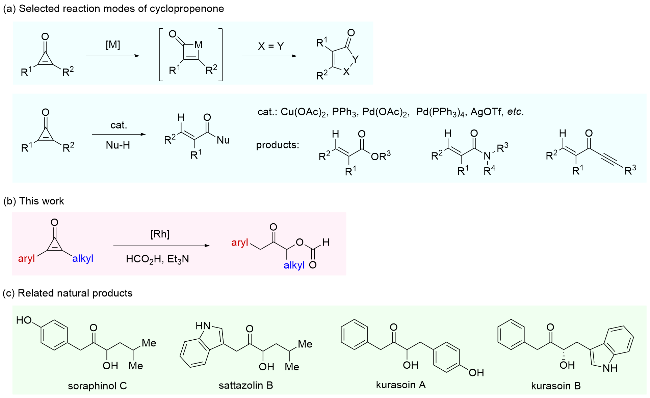
A mixture of 1a (29 mg, 0.2 mmol) and the Rh catalyst B (1.5 mg, 0.002 mmol) in CH2Cl2 (0.5 mL) was added to the mixture of formic acid (99%)/triethylamine (0.5 mL, V∶ V=1∶1, 30 equiv.). Then the mixture was stirred at 30 ℃ under an argon atmosphere. After completion of the reaction as indicated by TLC, it was quenched with brine, extracted with CH2Cl2 (10 mL×3), washed with brine, dried over anhydrous Na2SO4, filtered and concentrated. The residue was purified by chromatography (petroleum ether/ ethyl acetate, V∶V=5∶1) to afford 2a (28 mg, 72% yield, 6∶1 rr) as colorless oil. Compounds 2b~2t were prepared using the same method.
3-Oxo-4-phenylbutan-2-yl formate (2a): Colorless oil, 28 mg, 72% yield and 6∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.06 (s, 1H), 7.37~7.29 (m, 2H), 7.29~7.23 (m, 1H), 7.23~7.15 (m, 2H), 5.29 (q, J=7.0 Hz, 1H), 3.89~3.73 (m, 2H), 1.40 (d, J=7.0 Hz, 3H); 13C NMR (151 MHz, CDCl3) δ: 204.2, 160.0, 132.9, 129.6, 128.7, 127.3, 73.8, 45.5, 16.2; IR (KBr) ν: 1716, 1495, 1454, 1164, 1031, 699 cm-1; HRMS (ESI) calcd for C11H12O3Na [M+Na]+ 215.0679, found 215.0676.
4-(4-Methoxyphenyl)-3-oxobutan-2-yl formate (2b): Colorless oil, 15 mg, 34% yield and 1∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.09 (s, 1H), 7.12 (d, J=7.6 Hz, 2H), 6.87 (d, J=7.5 Hz, 2H), 5.30 (q, J=7.1 Hz, 1H), 3.79 (s, 3H), 3.76 (s, 2H), 1.41 (d, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 204.7, 160.1, 159.0, 130.8, 124.9, 114.3, 73.9, 55.4, 44.9, 16.5; IR (KBr) ν: 3358, 1717, 1613, 1512, 1456, 1247, 1166, 1032, 909, 737, 518 cm-1; HRMS (ESI) calcd for C12H14O4Na [M+Na]+ 245.0784, found 245.0785.
4-(4-(tert-Butyl)phenyl)-3-oxobutan-2-yl formate (2c): Colorless oil, 21 mg, 42% yield and 2∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.10 (s, 1H), 7.36 (d, J=8.2 Hz, 2H), 7.14 (d, J=8.1 Hz, 2H), 5.32 (q, J=7.0 Hz, 1H), 3.86~3.74 (m, 2H), 1.43 (d, J=7.0 Hz, 3H), 1.31 (s, 9H); 13C NMR (101 MHz, CDCl3) δ: 204.5, 160.1, 150.3, 129.8, 129.4, 125.8, 74.0, 45.2, 34.6, 31.4, 16.4; IR (KBr) ν: 2959, 1718, 1364, 1268, 1164, 1023, 810, 750, 554 cm-1; HRMS (ESI) calcd for C15H20O3Na [M+Na]+ 271.1305, found 271.1304.
4-(4-Chlorophenyl)-3-oxobutan-2-yl formate (2d): Colorless oil, 32 mg, 71% yield and 7∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.10 (s, 1H), 7.31 (d, J=8.3 Hz, 2H), 7.13 (d, J=8.3 Hz, 2H), 5.29 (q, J=7.0 Hz, 1H), 3.87~3.73 (m, 2H), 1.44 (d, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 203.9, 160.1, 133.4, 131.4, 131.1, 129.0, 74.1, 44.7, 16.4; IR (KBr) ν: 1717, 1492, 1164, 1090, 1016, 802, 751, 495 cm-1; HRMS (ESI) calcd for C11H11O3ClNa [M+Na]+ 249.0289, found 249.0285.
4-(3-Chlorophenyl)-3-oxobutan-2-yl formate (2e): Colorless oil, 36 mg, 79% yield and 10∶1 rr. 1H NMR (600 MHz, CDCl3) δ: 8.10 (s, 1H), 7.26~7.23 (m, 2H), 7.18 (s, 1H), 7.10~7.05 (m, 1H), 5.29 (q, J=7.0 Hz, 1H), 3.88~3.73 (m, 2H), 1.44 (d, J=7.0 Hz, 3H); 13C NMR (151 MHz, CDCl3) δ: 203.6, 160.1, 134.8, 134.6, 130.0, 129.9, 128.0, 127.7, 74.1, 44.9, 16.4; IR (KBr) ν: 1716, 1574, 1477, 1160, 1029, 870, 766, 683 cm-1; HRMS (ESI) calcd for C11H11- O3ClNa [M+Na]+ 249.0289, found 249.0281.
4-(3-Bromophenyl)-3-oxobutan-2-yl formate (2f): Colorless oil, 43 mg, 79% yield and 10∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.11 (s, 1H), 7.41 (d, J=7.9 Hz, 1H), 7.35 (s, 1H), 7.21 (t, J=7.8 Hz, 1H), 7.12 (d, J=7.6 Hz, 1H), 5.30 (q, J=7.0 Hz, 1H), 3.87~3.72 (m, 2H), 1.45 (d, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 203.6, 160.1, 135.1, 132.8, 130.6, 130.3, 128.4, 122.8, 74.1, 44.9, 16.4; IR (KBr) ν: 1716, 1569, 1475, 1161, 1028, 765; HRMS (ESI) calcd for C11H11O3BrNa [M+Na]+ 292.9784, found: 292.9781.
4-([1'-Biphenyl]-4-yl)-3-oxobutan-2-yl formate (2g): Colorless oil, 34 mg, 63% yield and 4∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.13 (s, 1H), 7.61~7.55 (m, 4H), 7.47~7.41 (m, 2H), 7.37~7.32 (m, 1H), 7.30~7.26 (m, 2H), 5.35 (q, J=6.9 Hz, 1H), 3.93~3.82 (m, 2H), 1.46 (d, J=7.0 Hz, 3H); 13C NMR (151 MHz, CDCl3) δ: 204.3, 160.1, 140.8, 140.4, 131.9, 130.2, 128.9, 127.6, 127.5, 127.2, 74.1, 45.3, 16.4; IR (KBr) ν: 1718, 1487, 1167, 1031, 758, 698 cm-1; HRMS (ESI) calcd for C17H16O3Na [M+Na]+ 291.0992, found 291.0990.
3-Oxo-4-(thiophen-3-yl)butan-2-yl formate (2h): Colorless oil, 22 mg, 56% yield and 2∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.10 (s, 1H), 7.31 (dd, J=5.0, 3.0 Hz, 1H), 7.16~7.10 (m, 1H), 6.97 (dd, J=5.0, 1.2 Hz, 1H), 5.36~5.26 (m, 1H), 3.92~3.79 (m, 2H), 1.43 (d, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 203.9, 160.1, 132.5, 128.7, 126.2, 123.6, 73.9, 40.1, 16.4; IR (KBr) ν: 1715, 1377, 1163, 1028, 763 cm-1; HRMS (ESI) calcd for C9H10O3SNa [M+Na]+ 221.0243, found 221.0242.
4-(4-Chloro-3-methylphenyl)-3-oxobutan-2-yl formate (2i): Colorless oil, 29 mg, 60% yield and 5∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.10 (s, 1H), 7.29 (d, J=8.1 Hz, 1H), 7.05 (s, 1H), 6.95 (d, J=8.1 Hz, 1H), 5.29 (q, J=7.0 Hz, 1H), 3.84~3.68 (m, 2H), 2.35 (s, 3H), 1.44 (d, J=7.0 Hz, 3H); 13C NMR (151 MHz, CDCl3) δ: 204.1, 160.1, 136.5, 133.5, 132.3, 131.3, 129.4, 128.4, 74.0, 44.7, 20.2, 16.4; IR (KBr) ν: 1717, 1481, 1161, 1032, 806 cm-1; HRMS (ESI) calcd for C12H13O3ClNa [M+Na]+ 263.0045, found 263.0041.
4-(3,5-Dichlorophenyl)-3-oxobutan-2-yl formate (2j): Colorless oil, 48 mg, 91% yield and 14∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.14 (s, 1H), 7.30 (s, 1H), 7.11 (s, 2H), 5.31 (q, J=7.0 Hz, 1H), 3.92~3.72 (m, 2H), 1.49 (d, J=7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 203.0, 160.0, 136.0, 135.1, 128.3, 127.6, 74.1, 44.3, 16.3; IR (KBr) ν: 3318, 1719, 1569, 1433, 1163, 1031, 907, 858, 796, 728 cm-1; HRMS (ESI) calcd for C11H10O3Cl2Na [M+Na]+ 282.9899, found 282.9892.
2-Oxo-1-phenylheptan-3-yl formate (2k): Colorless oil, 29 mg, 61% yield and 4∶1 rr. 1H NMR (400 MHz, CDCl3 ) δ: 8.12 (s, 1H), 7.39~7.31 (m, 2H), 7.31~7.26 (m, 1H), 7.24~7.16 (m, 2H), 5.23 (dd, J=7.3, 5.0 Hz, 1H), 3.89~3.73 (m, 2H), 1.85~1.68 (m, 2H), 1.37~1.23 (m, 4H), 0.93~0.82 (m, 3H); 13C NMR (151 MHz, CDCl3) 204.0, 160.4, 133.0, 129.8, 128.9, 127.4, 77.7, 46.1, 30.2, 27.3, 22.3, 13.9; IR (KBr) ν: 2957, 1719, 1455, 1167, 750, 701 cm-1; HRMS (ESI) calcd for C14H18O3Na [M+Na]+ 257.1148, found 257.1147.
1-(Naphthalen-1-yl)-2-oxoheptan-3-yl formate (2l): Colorless oil, 30 mg, 53% yield and 2∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.13 (s, 1H), 7.91~7.79 (m, 3H), 7.56~7.47 (m, 2H), 7.45 (t, J=7.6 Hz, 1H), 7.40~7.34 (m, 1H), 5.27 (t, J=6.2 Hz, 1H), 4.35~4.19 (m, 2H), 1.79~1.69 (m, 2H), 1.37~1.16 (m, 4H), 0.83 (t, J=7.1 Hz, 3H); 13C NMR (151 MHz, CDCl3) δ: 204.1, 160.5, 134.0, 132.4, 129.6, 128.9, 128.52, 128.47, 126.6, 126.1, 125.5, 123.9, 77.6, 44.4, 30.4, 27.2, 22.2, 13.8; IR (KBr) ν: 2956, 1718, 1168, 781 cm-1; HRMS (ESI) calcd for C18H20O3Na [M+Na]+ 307.1305, found 307.1306.
1-(3,5-Dichlorophenyl)-2-oxoheptan-3-yl formate (2m): Colorless oil, 54 mg, 89% yield and 14∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.14 (s, 1H), 7.28 (s, 1H), 7.07 (s, 2H), 5.19 (dd, J=7.3, 5.3 Hz, 1H), 3.86~3.67 (m, 2H), 1.90~1.73 (m, 2H), 1.43~1.27 (m, 4H), 0.95~0.85 (m, 3H); 13C NMR (151 MHz, CDCl3) δ: 202.7, 160.3, 136.1, 135.2, 128.4, 127.7, 77.9, 44.7, 30.2, 27.2, 22.4, 13.9; IR (KBr) ν: 2957, 1719, 1568, 1433, 1162, 857, 796, 738 cm-1; HRMS (ESI) calcd for C14H16O3Cl2Na [M+Na]+ 325.0369, found 325.0364.
6-Chloro-1-(4-chlorophenyl)-2-oxohexan-3-yl formate (2n): Colorless oil, 42 mg, 72% yield and 4∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.13 (s, 1H), 7.31 (d, J=8.3 Hz, 2H), 7.13 (d, J=8.3 Hz, 2H), 5.29~5.21 (m, 1H), 3.88~3.71 (m, 2H), 3.54 (t, J=6.2 Hz, 2H), 2.10~1.99 (m, 1H), 1.99~1.89 (m, 1H), 1.89~1.80 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 202.9, 160.1, 133.5, 131.12, 131.09, 129.0, 76.8, 45.1, 44.2, 27.9, 27.7; IR (KBr) ν: 1717, 1492, 1162, 1091, 1016, 751, 498, 425 cm-1; HRMS (ESI) calcd for C13H14O3Cl2Na [M+Na]+ 311.0212, found 311.0211.
6-Chloro-1-(3,4-dichlorophenyl)-2-oxohexan-3-yl formate (2o): Colorless oil, 49 mg, 75% yield and 8∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.14 (s, 1H), 7.41 (d, J=8.2 Hz, 1H), 7.29 (s, 1H), 7.03 (d, J=8.2 Hz, 1H), 5.25 (dd, J=7.6, 4.2 Hz, 1H), 3.86~3.71 (m, 2H), 3.56 (t, J=6.0 Hz, 2H), 2.13~2.02 (m, 1H), 2.02~1.92 (m, 1H), 1.92~1.83 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 202.4, 160.1, 132.9, 132.7, 131.8, 131.7, 130.7, 129.2, 76.8, 44.6, 44.1, 27.9, 27.6; IR (KBr) ν: 2933, 1721, 1472, 1165, 1032, 750, 440 cm-1; HRMS (ESI) calcd for C13H13O3Cl3Na [M+ Na]+ 344.9822, found 344.9820.
1-(4-Chlorophenyl)-2-oxo-6-phenylhexan-3-yl formate (2p): Colorless oil, 43 mg, 65% yield and 4∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.13 (s, 1H), 7.34~7.26 (m, 4H), 7.25~7.19 (m, 1H), 7.15 (d, J=7.4 Hz, 2H), 7.07 (d, J=8.3 Hz, 2H), 5.22 (dd, J=7.3, 4.7 Hz, 1H), 3.80~3.65 (m, 2H), 2.70~2.55 (m, 2H), 1.89~1.76 (m, 2H), 1.76~1.66 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 203.3, 160.3, 141.2, 133.4, 131.3, 131.0, 128.9, 128.6, 128.5, 126.2, 77.5, 45.0, 35.2, 29.8, 26.7; IR (KBr) ν: 2933, 1717, 1492, 1454, 1163, 1090, 1015, 802, 747, 699, 492 cm-1; HRMS (ESI) calcd for C19H19O3ClNa [M+Na]+ 353.0915, found 353.0918.
1-(3-Chlorophenyl)-6-(methoxymethoxy)-2-oxohexan-3- yl formate (2q): Colorless oil, 47 mg, 75% yield and 6∶1 rr. 1H NMR (600 MHz, CDCl3) δ: 8.14 (s, 1H), 7.29~7.26 (m, 2H), 7.20 (s, 1H), 7.11~7.05 (m, 1H), 5.27 (dd, J=8.5, 4.1 Hz, 1H), 4.60 (s, 2H), 3.85~3.75 (m, 2H), 3.56~3.51 (m, 2H), 3.35 (s, 3H), 2.02~1.94 (m, 1H), 1.93~1.84 (m, 1H), 1.72~1.66 (m, 2H); 13C NMR (151 MHz, CDCl3) δ: 203.0, 160.3, 134.8, 134.6, 130.0, 129.9, 128.0, 127.6, 96.6, 77.5, 66.7, 55.4, 45.3, 27.5, 25.4; IR (KBr) ν: 3421, 2934, 1717, 1476, 1150, 1108, 1035, 917, 765, 683 cm-1; HRMS (ESI) calcd for C15H19O5ClNa [M+Na]+ 337.0813, found: 337.0811.
6-(Benzyloxy)-1-(3-chlorophenyl)-2-oxohexan-3-yl for- mate (2r): Colorless oil, 53 mg, 74% yield and 6∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.12 (s, 1H), 7.39~7.28 (m, 5H), 7.27~7.24 (m, 2H), 7.19 (s, 1H), 7.09~7.03 (m, 1H), 5.26 (dd, J=8.0, 3.8 Hz, 1H), 4.50 (s, 2H), 3.84~3.71 (m, 2H), 3.48 (t, J=5.9 Hz, 2H), 2.05~1.95 (m, 1H), 1.94~1.83 (m, 1H), 1.75~1.67 (m, 2H); 13C NMR (151 MHz, CDCl3) δ: 203.0, 160.2, 138.3, 134.8, 134.5, 130.0, 129.9, 128.6, 128.0, 127.81, 127.78, 127.6, 77.5, 73.1, 69.1, 45.2, 27.4, 25.4; IR (KBr) ν: 3358, 1717, 1477, 1453, 1361, 1163, 1069, 747, 698 cm-1; HRMS (ESI) calcd for C20H21O4ClNa [M+Na]+ 383.1021, found 383.1020.
1-(3-Chlorophenyl)-5-(1,3-dioxolan-2-yl)-2-oxopentan-3-yl formate (2s): Colorless oil, 35 mg, 56% yield and 6∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.13 (s, 1H), 7.29~7.26 (m, 2H), 7.20 (s, 1H), 7.11~7.05 (m, 1H), 5.29 (dd, J=8.2, 4.2 Hz, 1H), 4.88 (t, J=4.2 Hz, 1H), 4.00~3.91 (m, 2H), 3.91~3.84 (m, 2H), 3.84~3.74 (m, 2H), 2.07~1.97 (m, 1H), 1.96~1.86 (m, 1H), 1.82~1.73 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 202.9, 160.2, 134.8, 134.5, 130.0, 129.9, 128.0, 127.6, 103.4, 77.3, 65.2, 65.1, 45.3, 29.0, 24.5; IR (KBr) ν: 2886, 1717, 1574, 1476, 1141, 1028, 941, 765, 683 cm-1; HRMS (ESI) calcd for C15H17O5ClNa [M+Na]+ 335.0657, found 335.0654.
1-Cyclohexyl-2-oxo-3-phenylpropyl formate (2t): Colorless oil, 26 mg, 50% yield and 3∶1 rr. 1H NMR (400 MHz, CDCl3) δ: 8.14 (s, 1H), 7.37~7.30 (m, 2H), 7.30~7.24 (m, 1H), 7.24~7.14 (m, 2H), 5.11 (d, J=4.3 Hz, 1H), 3.90~3.69 (m, 2H), 2.00~1.89 (m, 1H), 1.81~1.70 (m, 2H), 1.69~1.53 (m, 3H), 1.32~1.08 (m, 5H); 13C NMR (101 MHz, CDCl3) δ: 203.9, 160.5, 133.0, 129.8, 128.8, 127.3, 81.8, 46.7, 39.3, 29.5, 27.3, 26.2, 26.0, 25.9; IR (KBr) ν: 2928, 2855, 1719, 1452, 1168, 701 cm-1; HRMS (ESI) calcd for C16H20O3Na [M+Na]+ 283.1305, found 283.1304.
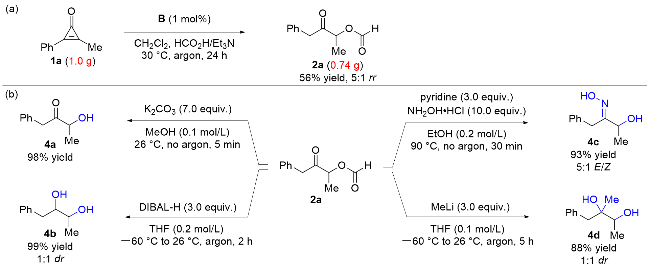
2a (19 mg, 0.1 mmol) and K2CO3 (97 mg, 0.7 mmol) were dissolved in MeOH (1 mL). The reaction mixture was stirred at 26 ℃ for 5 min. After completion of the reaction as indicated by TLC, it was quenched with saturated aqueous NH4Cl, extracted with EtOAc (5 mL×3), washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated. The residue was purified by chromatography on silica gel (petroleum ether/ethyl acetate, V∶V=3∶1) to afford 3-hydroxy-1-phenylbutan-2-one (4a) (16 mg, 98% yield) as colorless oil. 1H NMR (400 MHz, CDCl3) δ: 7.31~7.24 (m, 2H), 7.24~7.19 (m, 1H), 7.14 (d, J=7.1 Hz, 2H), 4.34~4.23 (m, 1H), 3.83~3.66 (m, 2H), 3.38 (d, J=4.8 Hz, 1H), 1.36 (d, J=7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 210.2, 133.1, 129.5, 128.9, 127.5, 72.4, 44.7, 19.9; IR (KBr) ν: 3388, 1714, 1496, 1454, 1050, 909, 730, 698 cm-1; HRMS calcd for C10H12O2Na (ESI) [M+Na]+ 187.0730, found 187.0735.
To a solution of 2a (19 mg, 0.1 mmol) in dry THF (1 mL) was added DIBAL-H (0.32 mL, 1.0 mol/L in THF, 0.32 mmol) at -60 ℃. After 30 min, the reaction warmed to room temperature and stirred for another 1.5 h. After completion of the reaction as indicated by TLC, it was quenched with saturated aqueous NH4Cl, extracted with EtOAc (5 mL×3), washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated. The residue was purified by chromatography on silica gel (petroleum ether/ethyl acetate, V∶V=2∶1) to afford 1-phenylbutane-2,3-diol (4b) (16 mg, 99% yield, 1∶1 dr) colorless oil. 1H NMR (400 MHz, CDCl3) δ: 7.38~7.28 (m, 4H), 7.28~7.21 (m, 6H), 3.91~3.78 (m, 2H), 3.73~3.64 (m, 1H), 3.63~3.56 (m, 1H), 2.94~2.86 (m, 1H), 2.86~2.79 (m, 1H), 2.74~2.63 (m, 2H), 2.01 (br, 4H), 1.32~1.21 (m, 6H); 13C NMR (101 MHz, CDCl3) δ: 138.4, 138.1, 129.6, 129.5, 128.82, 128.79, 126.73, 126.72, 76.8, 76.0, 70.1, 40.1, 38.2, 19.6, 17.3; IR (KBr) ν: 3352, 1642, 1495, 1454, 1053, 988, 731, 698 cm-1; HRMS (ESI) calcd for C10H14O2Na [M+Na]+ 189.0886, found 189.0882.
2a (19 mg, 0.1 mmol) and NH2OH•HCl (70 mg, 1 mmol) were dissolved in EtOH (0.5 mL), followed by the slow addition of pyridine (24 μL, 0.3 mmol). The reaction mixture was stirred at 90 ℃ for 30 min. After completion of the reaction as indicated by TLC, it was quenched with HCl (1 mol/L), extracted with EtOAc (5 mL×3), washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated. The residue was purified by column chromatography on silica gel (petroleum ether/ethyl acetate, V∶V=2∶1) to afford 3-hydroxy-1-phenylbutan-2-one oxime (4c) (17 mg, 93% yield, 5∶1 E/Z) as a colorless oil. 1H NMR (400 MHz, CDCl3) δ: 7.38~7.15 (m, 6H), 4.84 (q, J=6.7 Hz, 0.2H), 4.38 (q, J=6.4 Hz, 1H), 3.92 (d, J=14.4 Hz, 1H), 3.66 (d, J=14.4 Hz, 1H), 3.60 (s, 0.4H), 1.30 (d, J=6.5 Hz, 3H), 1.26 (d, J=6.6 Hz, 0.6H); 13C NMR (101 MHz, CDCl3) δ: 163.5, 161.5, 136.6, 136.3, 129.3, 129.0, 128.8, 127.0, 126.7, 68.5, 65.7, 37.2, 31.1, 21.4, 20.3; IR (KBr) ν: 3260, 1602, 1494, 1452, 1071, 962, 889, 728, 697 cm-1; HRMS (ESI) calcd for C10H13O2NNa [M+Na]+ 202.0838, found 202.0833.
To a solution of 2a (19 mg, 0.1 mmol) in THF (1 mL) was added MeLi (0.11 mL, 2.7 mol/L in THF, 0.3 mmol) at -60 ℃. After 1 h, the reaction mixture was warmed to room temperature and stirred for another 4 h. After completion of the reaction as indicated by TLC, it was quenched with saturated aqueous NH4Cl, extracted with EtOAc (5 mL×3), washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated. The residue was purified by chromatography on silica gel (petroleum ether/ethyl acetate, V∶V=2∶1) to afford 2-methyl-1-phenylbutane-2,3-diol (4d) (16 mg, 88% yield, 1∶1 dr) as a colorless oil. 1H NMR (400 MHz, CDCl3) δ: 7.35~7.28 (m, 4H), 7.26~7.19 (m, 6H), 3.72 (q, J=6.4 Hz, 1H), 3.65 (q, J=6.4 Hz, 1H), 2.98 (d, J=13.4 Hz, 1H), 2.82~2.72 (m, 2H), 2.59 (d, J=13.3 Hz, 1H), 1.24 (d, J=6.4 Hz, 3H), 1.20 (d, J=6.4 Hz, 3H), 1.07 (s, 6H); 13C NMR (101 MHz, CDCl3) δ: 137.1, 137.0, 130.9, 130.7, 128.5, 128.4, 126.8, 126.7, 75.0, 74.7, 74.0, 72.4, 45.1, 41.1, 23.6, 21.1, 17.5, 17.3; IR (KBr) ν: 3376, 2978, 1495, 1453, 1375, 1085, 910, 700 cm-1; HRMS (ESI) calcd for C11H16O2Na [M+Na]+ 203.1043, found 203.1040.
Supporting Information 1H NMR and
13C NMR spectra of compounds
1c~
1j,
1l~
1t,
2a~
2t, and
4a~
4d. The Supporting Information is available free of charge via the Internet at
http://sioc-journal.cn.