

Chinese Journal of Organic Chemistry >
Progress in the Synthesis and Derivatization of Norcorrole
Received date: 2022-02-28
Revised date: 2022-04-13
Online published: 2022-04-29
Supported by
National Natural Science Foundation of China(22171076); Science and Technology Innovation Program of Hunan Province(2021RC5028)
In recent years, norcorrole has attracted extensive attention of scientific researchers, owing to its distinct paratropic ring current, high single molecule conductivity, narrow HOMO-LUMO gap, stable redox performance and unique chemical reactivity. The different synthetic methods of norcorrole in recent years are reviewed, with emphasis on the derivatization reactions in four aspects: insertion, substitution, redox and extension of π conjugated system, and the development of norcorrole is prospected.
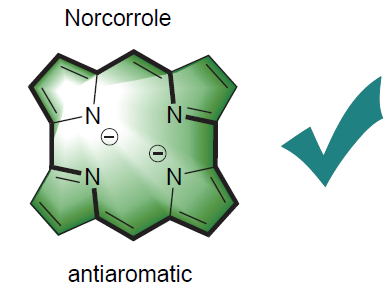
Key words: norcorrole; synthesis; derivatization; antiaromaticity
Sha Li , Yahan Sun , Yankui Meng , Xiaofang Li , Shaowei Zhang . Progress in the Synthesis and Derivatization of Norcorrole[J]. Chinese Journal of Organic Chemistry, 2022 , 42(8) : 2390 -2405 . DOI: 10.6023/cjoc202202039
| [1] | Proft, F. D.; Geerlings, P. Chem. Rev. 2001, 101, 1451. |
| [2] | Krygowski, T. M.; Cyrański, M. K. Chem. Rev. 2001, 101, 1385. |
| [3] | Schleyer, P. R.; Manoharan, M.; Wang, Z. X.; Kiran, B.; Jiao, H.; Puchta, R.; Hommes, N. E. Org. Lett. 2001, 3, 2465. |
| [4] | Liu, J.; Ma, J.; Zhang, K.; Ravat, P.; Machta, P.; Avdoshenko, S.; Hennersdorf, F.; Komber, H.; Pisula, W.; Weigand, J. J.; Popov, A. A.; Berger, R.; Müllen, K.; Feng, X. J. Am. Chem. Soc. 2017, 139, 7513. |
| [5] | Breslow, R.; Foss, F. W. J. Phys.: Condens. Matter. 2008, 20, 374104. |
| [6] | Nishinaga, T.; Uto, T.; Inoue, R.; Matsuura, A.; Treitel, N.; Rabinovitz, M.; Komatsu, K. Chem.-Eur. J. 2008, 14, 2067. |
| [7] | Fan, C.; Mercier, L. G.; Piers, W. E.; Tuononen, H. M.; Parvez, M. J. Am. Chem. Soc. 2010, 132, 9604. |
| [8] | Breslow, R.; Schneebeli, S. T. Tetrahedron. 2011, 67, 10171. |
| [9] | Ghosh, A.; Wasbotten, I. H.; Davis, W.; Swarts, J. C. Eur. J. Inorg. Chem. 2005, 22, 4479. |
| [10] | Sessler, J. L.; Tomat, E. Acc. Chem. Res. 2007, 40, 371. |
| [11] | Aviv-Harel, I.; Gross, Z. Chem.-Eur. J. 2009, 15, 8382. |
| [12] | Nozawa, R.; Yamamoto, K.; Shin, J. Y.; Hiroto, S.; Shinokubo, H. Angew. Chem., Int. Ed. 2015, 54, 8454. |
| [13] | Liu, B.; Yoshida, T.; Li, X.; Stępień, M.; Shinokubo, H.; Chmielewski, P. J. Angew. Chem., Int. Ed. 2016, 55, 13142. |
| [14] | Tu, X. M.; Xie, Q. J.; Jiang, S. Y.; Yao, S. Z. Biosens. Bioelectron. 2007, 22, 2819. |
| [15] | Suga, T.; Sugita, S.; Ohshiro, H.; Oyaizu, K.; Nishide. H. Adv. Mater. 2011, 23, 751. |
| [16] | Shin, J. Y.; Yamada, T.; Yoshikawa, H.; Awaga, K.; Shinokubo, H. Angew. Chem., Int. Ed. 2014, 53, 3096. |
| [17] | Deng, K.; Li, X.; Huang, H. Electrochim. Acta 2016, 204, 84. |
| [18] | Fujii, S.; Marqués-González, S.; Shin, J. Y.; Shinokubo, H.; Masuda, T.; Nishino, T.; Arasu, N. P.; Vázquez, H.; Kiguchi, M. Nat. Commun. 2017, 8, 15984. |
| [19] | Ukai, S.; Koo, Y. H.; Fukui, N.; Seki, S.; Shinokubo, H. Dalton Trans. 2020, 49, 14383. |
| [20] | Bröring, M.; Köhler, S.; Kleeberg, C. Angew. Chem., Int. Ed. 2008, 47, 5658. |
| [21] | Ito, T.; Hayashi, Y.; Shimizu, S.; Shin, J. Y.; Kobayashi, N.; Shinokubo, H. Angew. Chem., Int. Ed. 2012, 51, 8542. |
| [22] | Yoshida, T.; Sakamaki, D.; Seki, S.; Shinokubo, H. Chem. Commun. 2017, 53, 1112. |
| [23] | Liu, S.; Tanaka, H.; Nozawa, R.; Fukui, N.; Shinokubo, H. Chem.-Eur. J. 2019, 25, 7618. |
| [24] | Yoshida, T.; Takahashi, K.; Ide, Y.; Kishi, R.; Fujiyoshi, J.; Lee, S.; Hiraoka, Y.; Kim, D.; Nakano, M.; Ikeue, T.; Yamada, H.; Shinokubo, H. Angew. Chem., Int. Ed. 2018, 57, 2209. |
| [25] | Murakami, K.; Yamamoto, Y.; Yorimitsu, H.; Osuka, A. Chem.-Eur. J. 2013, 19, 9123. |
| [26] | Yonezawa, T.; Shafie, S. A.; Hiroto, S.; Shinokubo, H. Angew. Chem., Int. Ed. 2017, 56, 11822. |
| [27] | Zhang, S.; Zhang, D.; Liebeskind, L. S. J. Org. Chem. 1997, 62, 2312. |
| [28] | Yoshida, T.; Shafie, S. A.; Kawashima, H.; Fukui, N.; Shinokubo, H. Org. Lett. 2021, 23, 2826. |
| [29] | Kido, H.; Shin, J. Y.; Shinokubo, H. Angew. Chem., Int. Ed. 2013, 52, 13727. |
| [30] | Kira, M.; Ishida, S.; Iwamoto, T.; Kabuto, C. J. Am. Chem. Soc. 1999, 121, 9722. |
| [31] | Fukuoka, T.; Uchida, K.; Sung, Y. M.; Shin, J. Y.; Ishida, S.; Lim, J. M.; Hiroto, S.; Furukawa, K.; Kim, D.; Iwamoto, T.; Shinokubo, H. Angew. Chem., Int. Ed. 2014, 53, 1506. |
| [32] | Liu, S. Y.; Fukuoka, T.; Fukui, N.; Shin, J. Y.; Shinokubo, H. Org. Lett. 2020, 22, 4400. |
| [33] | Ren, D.; Smaga, O.; Fu, X.; Li, X.; Pawlicki, M.; Koniarz, S.; Chmielewski, P. J. Org. Lett. 2021, 23, 1032. |
| [34] | Horie, M.; Hayashi, Y.; Yamaguchi, S.; Shinokubo, H. Chem.-Eur. J. 2012, 18, 5919. |
| [35] | Li, X.; Sun, Y. H.; Yu, X. Y.; Tan, J. X. CN 112174972, 2021. |
| [36] | Yoshida, T.; Shinokubo, H. Mater. Chem. Front. 2017, 1, 1853. |
| [37] | Li, X.; Liu, B.; Yi, P.; Yi, R.; Yu, X.; Chmielewski, P. J. J. Org. Chem. 2011, 76, 2345. |
| [38] | Liu, B.; Li, X.; Zhang, J.; Chmielewski, P. J. Org. Biomol. Chem. 2013, 11, 4831. |
| [39] | Ren, D.; Fu, X.; Li, X.; Koniarz, S.; Chmielewski, P. J. Org. Chem. Front. 2019, 6, 2924. |
| [40] | Koley, D.; Colón, O. C.; Savinov, S. N. Org. Lett. 2009, 11, 4172. |
| [41] | Deng, Z.; Li, X.; Stępień, M.; Chmielewski, P. J. Chem.-Eur. J. 2016, 22, 4231. |
| [42] | Kawashima, H.; Hiroto, S.; Shinokubo, H. J. Org. Chem. 2017, 82, 10425. |
| [43] | Li, S.; Smaga, O.; Sun, Y.; Li, X.; Pawlicki, M.; Sukniewiczb, M.; Chmielewski, P. J. Org. Chem. Front. 2021, 8, 3639. |
| [44] | Shafie, S. A.; Kawashima, H.; Miyake, Y.; Shinokubo, H. ChemPlusChem 2019, 84, 623. |
| [45] | Liu, B.; Li, X.; Stępień, M.; Chmielewski, P. J. Chem.-Eur. J. 2015, 21, 7790. |
| [46] | Nozawa, R.; Yamamoto, K.; Hisaki, I.; Shin, J. Y.; Shinokubo, H. Chem. Commun. 2016, 52, 7106. |
| [47] | Whitlock, H. W.; Hanauer, R.; Oester, M. Y.; Bower, B. K. J. Am. Chem. Soc. 1969, 91, 7485. |
| [48] | Ukai, S.; Fukui, N.; Ikeue, T.; Shinokubo, H. Chem. Lett. 2022, 51, 590. |
| [49] | Silva, A. M. G.; Tomé, A. C.; Neves, M. G. P. M. S.; Silva, A. M. S.; Cavaleiro, J. A. S. J. Org. Chem. 2005, 70, 2306. |
| [50] | Li, X.; Zhuang, J.; Li, Y.; Liu, H.; Wang, S.; Zhu, D. Tetrahedron Lett. 2005, 46, 1555. |
| [51] | Li, X.; Chmielewski, P. J.; Xiang, J.; Xu, J.; Jiang, L.; Li, Y.; Liu, H.; Zhu, D. J. Org. Chem. 2006, 71, 9739. |
| [52] | Fu, X.; Meng, Y.; Li, X.; Stępień, M.; Chmielewski, P. J. Chem. Commun. 2018, 54, 2510. |
| [53] | Yokoi, H.; Wachi, N.; Hiroto, S.; Shinokubo, H. Chem. Commun. 2014, 50, 2715. |
| [54] | Li, X.; Meng, Y.; Yi, P.; Stępień, M.; Chmielewski, P. J. Angew. Chem., Int. Ed. 2017, 56, 10810. |
| [55] | Tanaka, T.; Osuka, A. Chem. Soc. Rev. 2015, 44, 943. |
| [56] | Tanaka, T.; Osuka, A. Chem.-Eur. J. 2018, 24, 17188. |
| [57] | Liu, S. Y.; Kawashima, H.; Fukui, N.; Shinokubo, H. Chem. Commun. 2020, 56, 6846. |
| [58] | Nozawa, R.; Tanaka, H.; Cha, W. Y.; Hong, Y.; Hisaki, I.; Shimizu, S.; Shin, J. Y.; Kowalczyk, T.; Irle, S.; Kim, D.; Shinokubo, H. Nat. Commun. 2016, 7, 13620. |
| [59] | Nozawa, R.; Kim, J.; Oh, J.; Lamping, A.; Wang, Y.; Shimizu, S.; Hisaki, I.; Kowalczyk, T.; Fliegl, H.; Kim, D.; Shinokubo, H. Nat. Commun. 2019, 10, 3576. |
| [60] | Kawashima, H.; Ukai, S.; Nozawa, R.; Fukui, N.; Fitzsimmons, G.; Kowalczyk, T.; Fliegl, H.; Shinokubo, H. J. Am. Chem. Soc. 2021, 143, 10676. |
| [61] | Ukai, S.; Takamatsu, A.; Nobuoka, M.; Tsutsui, Y.; Fukui, N.; Ogi, S.; Seki, S.; Yamaguchi, S.; Shinokubo, H. Angew. Chem., Int. Ed. 2022, 61, e202114230. |
/
| 〈 |
|
〉 |