化学学报 ›› 2021, Vol. 79 ›› Issue (1): 71-80.DOI: 10.6023/A20080392 上一篇 下一篇
研究论文
李金华a, 卓庆德b, 卓凯玥b, 陈大发a,*( ), 夏海平a,b,*(
), 夏海平a,b,*( )
)
投稿日期:2020-08-26
发布日期:2020-09-27
通讯作者:
陈大发, 夏海平
作者简介:基金资助:
Jinhua Lia, Qingde Zhuob, Kaiyue Zhuob, Dafa Chena,*( ), Haiping Xiaa,b,*(
), Haiping Xiaa,b,*( )
)
Received:2020-08-26
Published:2020-09-27
Contact:
Dafa Chen, Haiping Xia
Supported by:文章分享

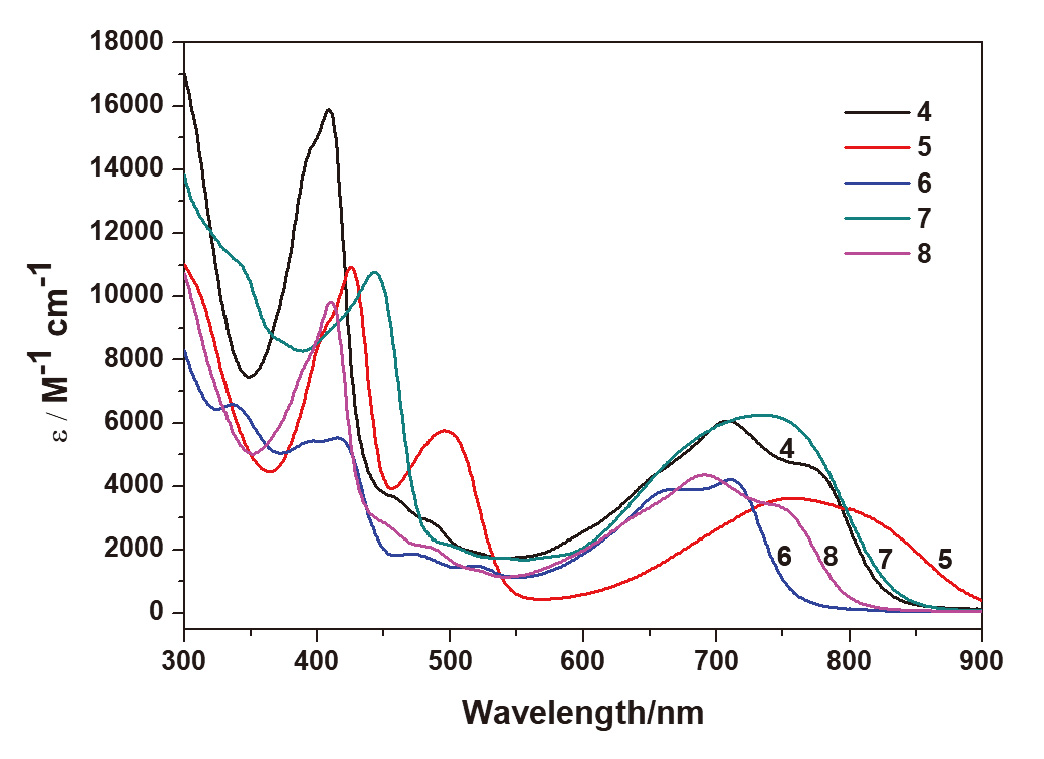
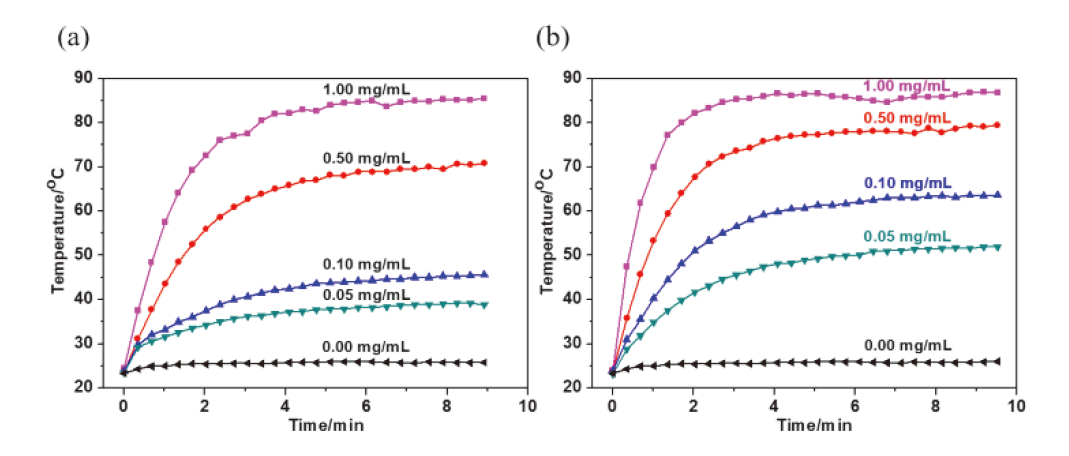
碳龙配合物由一条碳数不少于7的碳链配体鳌合过渡金属而成, 代表了一系列全新的π共轭分子骨架基元, 并表现出独特的光物理性质, 具有很好的应用前景. 通过合理设计多炔化合物, 实现了“一锅法”构筑具有大π共轭体系的铱杂碳龙配合物 4和 5, 首次将碳龙配合物拓展至金属铱体系. 此外, 还研究了 4和 5的配体取代反应, 制备了铱杂碳龙配合物 6~ 9. 所制备的一系列铱杂碳龙配合物在紫外-可见区具有很好的吸收, 部分甚至在近红外区也表现出一定的吸收特性. 光热性能研究表明, 化合物 5具有较好的近红外光热效果: 在808 nm的激光照射下, 含有 5(0.1 mg•mL -1)的乙醇/水溶液在10 min内温度可升高近40 ℃, 这为后续铱杂碳龙配合物的应用研究提供了可能性.
李金华, 卓庆德, 卓凯玥, 陈大发, 夏海平. 铱杂碳龙配合物的合成及反应性[J]. 化学学报, 2021, 79(1): 71-80.
Jinhua Li, Qingde Zhuo, Kaiyue Zhuo, Dafa Chen, Haiping Xia. Synthesis and Reactivity Studies of Irida-carbolong Complexes[J]. Acta Chimica Sinica, 2021, 79(1): 71-80.


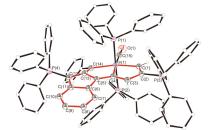
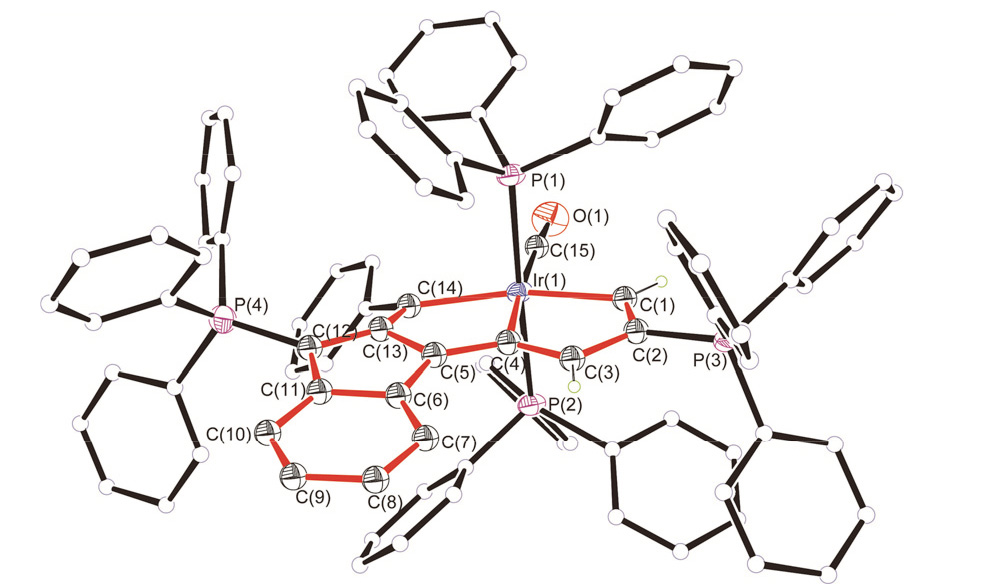
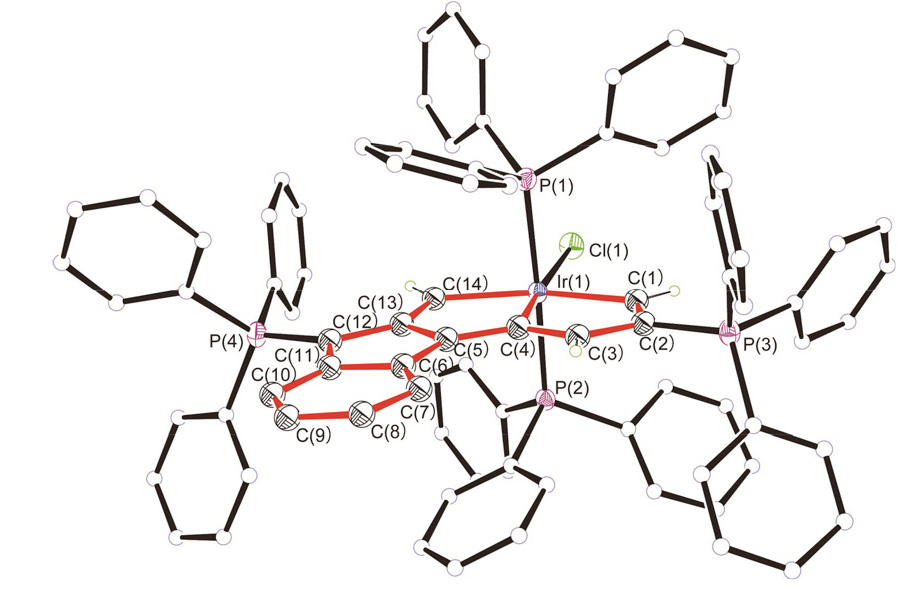
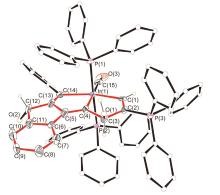
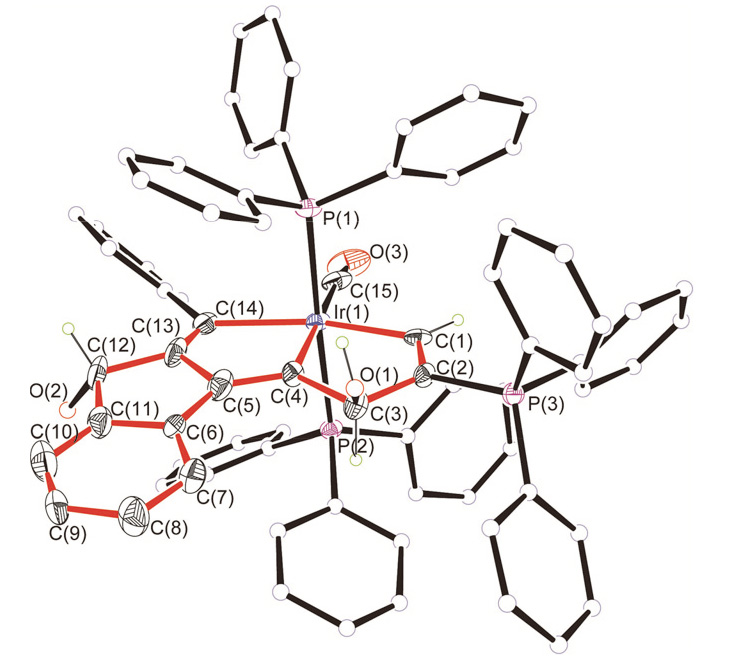

| [1] |
Selected reviews: (a) He, G.; Xia, H.; Jia, G. Chin. Sci. Bull. 2004, 49, 1543.
|
|
Landorf, C.W.; Haley, M.M. Angew. Chem., Int. Ed. 2006, 45, 3914.
|
|
|
Chen, J.; Jia, G. Coord. Chem. Rev. 2013, 257, 2491.
|
|
|
Cao, X.-Y.; Zhao, Q.; Lin, Z.; Xia, H. Acc. Chem. Res. 2014, 47, 341.
|
|
|
Frogley, B.J.; Wright, L.J. Coord. Chem. Rev. 2014, 270-271, 151.
|
|
|
Fernández, I.; Frenking, Gernot.; Merino, G. Chem. Soc. Rev. 2015, 44, 6452.
|
|
|
Frogley, B.J.; Wright, L.J. Chem. -Eur. J. 2018, 24, 2025.
|
|
|
Hua, Y.; Zhang, H.; Xia, H. Chin. J. Org. Chem. 2018, 38, 11 . (in Chinese)
|
|
|
华煜辉, 张弘, 夏海平, 有机化学, 2018, 38, 11.
|
|
| [2] |
Selected reviews: (a) Jia, G.Acc. Chem. Res. 2004, 37, 479.
|
|
Chen, J.; He, G.; Jia, G. Chin. J. Org. Chem. 2013, 33, 792 . (in Chinese)
|
|
|
陈江溪, 何国梅, 贾国成, 有机化学, 2013, 33, 792.
|
|
|
Jia, G. Organometallics 2013, 32, 6852.
|
|
| [3] |
Paneque, M.; Posadas, C.M.; Poveda, M.L.; Rendón, N.; Salazar, V.; Oñate, E.; Mereiter, K. J. Am. Chem. Soc. 2003, 125, 9898.
|
|
Paneque, M.; Posadas, C.M.; Poveda, M.L.; Rendón, N.; Santos, L.L.; Álvarez, E.; Salazar, V.; Mereiter, K.; Oñate, E. Organometallics 2007, 26, 2403.
|
|
|
Vivancos, Á.; Hernández, Y. A. Paneque, M.; Poveda, M. L.; Salazar, V.; Álvarez, E. Organometallics 2015, 34, 177.
|
|
| [4] |
He, G.; Chen, J.; Xia, H.; Sci. Bull. 2016, 61, 430.
|
|
Zhou, X.; Zhang, H. Chem. Eur. J. 2018, 24, 8962.
|
|
|
Wang, H.; Zhou, X.; Xia, H. Chin. J. Chem. 2018, 36, 93.
|
|
| [5] |
Wei, J.; Zhang, W.; Xi, Z. Chem. Sci. 2018, 9, 560.
|
| [6] |
Zhang, Y.; Wei, J.; Chi, Y.; Zhang, X.; Zhang, W.-X.; Xi, Z. J. Am. Chem. Soc. 2017, 139, 5309.
|
|
Liu, L.; Zhu, M.; Yu, H.-T.; Zhang, W.-X.; Xi, Z. J. Am. Chem. Soc. 2017, 139, 13688.
|
|
|
Zhang, Y.; Wei, J.; Zhu, M.; Chi, Y.; Zhang, W.-X.; Ye, S.; Xi, Z. Angew. Chem., Int. Ed. 2019, 58, 9625.
|
|
| [7] |
Wei, J.; Zhang, Y.; Chi, Y.; Liu, L.; Zhang, W.-X.; Xi, Z. J. Am. Chem. Soc. 2016, 138, 60.
|
|
An, K.; Shen, T.; Zhu, J. Organometallics 2017, 36, 3199.
|
|
|
Huang, Z.; Zhang, Y.; Zhang, W.-X.; Xi, Z. Organometallics 2019, 38, 2807.
|
|
| [8] |
Lv, Z.-J.; Huang, Z.; Shen, J.; Zhang, W.-X.; Xi, Z. J. Am. Chem. Soc. 2019, 141, 20547.
|
|
Wang, K.; Zhou, X. Chin. J. Org. Chem. 2020, 40, 1084 . (in Chinese)
|
|
|
王凯, 周锡庚, 有机化学, 2020, 40, 1084.
|
|
| [9] |
Frogley, B.J.; Wright, L. Angew. Chem., Int. Ed. 2017, 56, 143.
|
|
Ruan, W.; Leung, T.-F.; Shi, C.; Lee, K.H.; Sung, H.H.Y.; Williams, I.D.; Lin, Z.; Jia, G. Chem. Sci. 2018, 9, 5944.
|
|
|
Talavera, M. Peña-Gallego, A. Alonso-Gómez, J.L. Bolaño, S.; Chem. Commun., 2018, 54, 10974.
|
|
|
Zhang, M.-X.; Xu, Z.; Lu, T.; Yin, J.; Liu, S.H. Chem. Eur. J. 2018, 24, 14891.
|
|
|
Zhang, M.-X.; Zhang, J.; Jin, X.; Sun, X.; Yin, J.; Hartl, F.; Liu, S.H. Chem. Eur. J. 2018, 24, 18998.
|
|
|
Hu, Y.X.; Zhang, J.; Wang, X.; Lu, Z.; Zhang, F.; Yang, X.; Ma, Z.; Yin, J.; Xia, H.; Liu, S.H. Chem. Sci. 2019, 10, 10894.
|
|
|
Chu, Z.; He, G.; Cheng, X.; Deng, Z.; Chen, J. Angew. Chem., Int. Ed. 2019, 58, 9174.
|
|
| [10] |
Zhu, C.; Xia, H. Acc. Chem. Res. 2018, 51, 1691.
|
|
Luo, M.; Hua, Y.; Zhuo, K.; Long, L.; Lin, X.; Deng, Z.; Lin, Z.; Zhang, H.; Chen, D.; Xia, H. CCS Chem. 2020, 2, 758.
|
|
|
Lin, J.; Xu, Q.; Lin, X.; Hua, Y.; Chen, D.; Ruan, Y.; Zhang, H.; Xia, H. Chin. J. Chem. 2020, 38, 1273.
|
|
| [11] |
Zhu, C.; Li, S.; Luo, M.; Zhou, X.; Niu, Y.; Lin, M.; Zhu, J.; Cao, Z.; Lu, X.; Wen, T.; Xie, Z.; Schleyer, P.v.R.; Xia, H. Nat. Chem. 2013, 5, 698.
|
| [12] |
Zhu, C.; Yang, Y.; Luo, M.; Yang, C.; Wu, J.; Chen, L.; Liu, G.; Wen, T.; Zhu, J.; Xia, H. Angew. Chem., Int. Ed. 2015, 54, 6181.
|
|
Yang, C.; Lin, G.; Zhu, C.; Pang, X.; Wang, X.; Li, X.; Wang, B.; Xia, H.; Liu, G. J. Mater. Chem. B, 2018, 6, 2528.
|
|
|
Zhou, X.; Pang, X.; Nie, L.; Zhu, C.; Zhuo, K.; Zhuo, Q.; Chen, Z.; Liu, G.; Zhang, H.; Lin, Z.; Xia, H. Nat. Commun. 2019, 10, 1488.
|
|
| [13] |
Zhu, C.; Yang, C.; Wang, Y.; Lin, G.; Yang, Y.; Wang, X.; Zhu, J.; Chen, X.; Lu, X.; Liu, G.; Xia, H. Sci. Adv. 2016, 6, e1601031/1.
|
| [14] |
Li, R.; Lu, Z.; Cai, Y.; Jiang, F.; Tang, C.; Chen, Z.; Zheng, J.; Pi, J.; Zhang, R.; Liu, J.; Chen, Z.-B.; Yang, Y.; Shi, J.; Hong, W.; Xia, H. J. Am. Chem. Soc. 2017, 139, 14344.
|
| [15] |
Zhang, H.; Zhao, H.; Zhuo, K.; Hua, Y.; Chen, J.; He, X.; Weng, W.; Xia, H. Polym. Chem. 2019, 10, 386.
|
|
Chen, Y.; Yang, L.; Zheng, W.; Ouyang, P.; Zhang, H.; Ruan, Y.; Weng, W.; He, X.; Xia, H. ACS Macro Lett. 2020, 9, 344.
|
|
| [16] |
Zhuo, Q.; Lin, J.; Hua, Y.; Zhou, X.; Shao, Y.; Chen, S.; Chen, Z.; Zhu, J.; Zhang, H.; Xia, H. Nat. Commun. 2017, 8, 1912.
|
| [17] |
Zhuo, Q.; Zhang, H.; Hua, Y.; Kang, H.; Zhou, X.; Lin, X.; Chen, Z.; Lin, J.; Zhuo, K.; Xia, H. Sci. Adv. 2018, 4, eaat0336.
|
|
Li, J.; Kang, H.; Zhuo, K.; Zhuo, Q.; Zhang, H.; Lin, Y.-M.; Xia, H. Chin. J. Chem. 2018, 36, 1156.
|
|
| [18] |
Zhuo, Q.; Zhang, H.; Ding, L.; Lin, J.; Zhou, X.; Hua, Y.; Zhu, J.; Xia, H. iScience 2019, 19, 1214.
|
| [19] |
Schröder, F.G.; Sundermeyer, J. Organometallics 2015, 34, 1017.
|
|
Hariharan, P.S.; Mariyatra, M.B.; Mothi, E.M.; Neels, A. Rosair, G.; Anthony, S.P. New. J. Chem. 2017, 41, 4592.
|
|
| [20] |
Bleeke, J.R.; Behm, R. J. Am. Chem. Soc. 1997, 119, 8503.
|
|
Clark, G.R.; Johns, P.M.; Roper, W.R.; Wright, L.J. Organometallics 2008, 27, 451.
|
|
| [21] |
Wu, H.-P.; Ess, D.H.; Lanza, S.; Weakley, T.J.R.; Houk, K.N.; Baldridge, K.K.; Haley, M.M. Organometallics 2007, 26, 3957.
|
| [22] |
O'Connor, J.M.; Pu, L. J. Am. Chem. Soc. 1990, 112, 9663.
|
|
O'Connor, J.M.; Pu, L.; Chadha, R. Angew. Chem., Int. Ed. Engl. 1990, 29, 543.
|
|
| [23] |
Zhu, C.; Luo, M.; Zhu, Q.; Zhu, J.; Schleyer, P. v R.; Wu, J.I.-C.; Lu, X.; Xia, H. Nat. Commun. 2014, 5, 3265.
|
| [24] |
Ilg, K.; Werner, H. Organometallics 2001, 20, 3782.
|
|
Chin, C.S.; Kim, M.; Lee, H.; Noh, S.; Ok, K.M. Organometallics 2002, 21, 4785.
|
|
|
Torres, O.; Martín, M.; Sola, E. Organometallics 2010, 29, 3201.
|
|
| [25] |
Li, J.; Lin, Y.-M.; Zhang, H.; Chen, Y.; Lin, Z.; Xia, H. Chem. -Eur. J. 2019, 25, 5077.
|
|
Wu, F.; Huang, W.; Zhuo, K.; Hua, Y.; Lin, J.; He, G.; Chen, J.; Nie, L.; Xia, H. Chin. J. Org. Chem. 2019, 39, 1743 . (in Chinese)
|
|
|
吴凡, 黄文超, 卓凯玥, 华煜晖, 林剑锋, 何国梅, 陈江溪, 聂立铭, 夏海平, 有机化学, 2019, 39, 1743.
|
|
|
Lu, Z.; Zhu, Q.; Cai, Y.; Chen, Z.; Zhuo, K.; Zhu, J.; Zhang, H.; Xia, H. Sci. Adv. 2020, 6, eaay2535.
|
|
| [26] |
Bedard, T.C.; Moore, J.S. J. Am. Chem. Soc. 1995, 117, 10662.
|
| [27] |
Dubé, P.; Toste, F.D. J. Am. Chem. Soc. 2006, 128, 12062.
|
| [28] |
Kaiser, R.P.; Hessler, F.; Mosinger, J.; Císařová, I.; Kotora, M. Chem. Eur. J. 2015, 21, 13577.
|
| [1] | 陶鹏, 郑小康, 王国良, 盛星浩, 姜贺, 李文桃, 靳继彪, 王瑞鸿, 苗艳勤, 王华, 黄维扬. 新型双极传输特性橙光铱(III)配合物的设计、合成及其电致发光★[J]. 化学学报, 2023, 81(8): 891-897. |
| [2] | 王瑞祥, 赵庆如, 顾庆, 游书力. 金/铱接力催化炔基酰胺环化/不对称烯丙基苄基化串联反应★[J]. 化学学报, 2023, 81(5): 431-434. |
| [3] | 何倩, 李杰, 喻思佳, 吴东坪, 叶剑良, 黄培强. 铱催化叔酰胺与呋喃硅醚间的类插烯Aldol缩合反应: γ-亚苄基-丁烯酸内酯的合成★[J]. 化学学报, 2023, 81(10): 1265-1270. |
| [4] | 赵庆如, 蒋茹, 游书力. 铱催化串联不对称烯丙基取代/双键异构化构建轴手性化合物[J]. 化学学报, 2021, 79(9): 1107-1112. |
| [5] | 占林俊, 胡玮, 王梅, 黄斌, 龙亚秋. 亚胺氯化物介导一锅法合成3-吸电子基团取代吲哚衍生物[J]. 化学学报, 2021, 79(7): 903-907. |
| [6] | 杨普苏, 刘晨旭, 张文文, 游书力. 铱催化中氮茚衍生物的Friedel-Crafts类型不对称烯丙基取代反应[J]. 化学学报, 2021, 79(6): 742-746. |
| [7] | 杨妲, 张龙力, 刘欢, 杨朝合. 双功能配体修饰的Ir催化剂在“氢甲酰化-缩醛化”串联反应中的共催化作用[J]. 化学学报, 2021, 79(5): 658-662. |
| [8] | 任保轶, 依建成, 钟道昆, 赵玉志, 郭闰达, 盛永刚, 孙亚光, 解令海, 黄维. 含螺环位阻铱(III)配合物的共轭结构调控及其电致发光性能研究[J]. 化学学报, 2020, 78(1): 56-62. |
| [9] | 张琪, 刘奥, 于海珠, 傅尧. Ir(III)螯合物催化醚的硅氢化反应中负氢来源的机理研究[J]. 化学学报, 2018, 76(2): 113-120. |
| [10] | 陈仕琦, 代军, 周凯峰, 罗艳菊, 苏仕健, 蒲雪梅, 黄艳, 卢志云. 基于苯基修饰策略的新型可溶液加工红光铱配合物的设计、合成及电致发光性能研究[J]. 化学学报, 2017, 75(4): 367-374. |
| [11] | 刘旭, 韩召斌, 王正, 丁奎岭. SpinPHOX/Ir(I)催化的2-羟甲基-3-芳基丙烯酸的不对称氢化[J]. 化学学报, 2014, 72(7): 849-855. |
| [12] | 史清华, 彭谦, 孙少瑞, 帅志刚. 蓝光材料Ir(III)配合物的磷光效率与光谱的振动关联函数研究[J]. 化学学报, 2013, 71(06): 884-891. |
| [13] | 童碧海, 梅群波, 李志文, 董永平, 张千峰. 系列2-苯基喹啉类铱配合物的合成及电化学发光性能研究[J]. 化学学报, 2012, 70(23): 2451-2456. |
| [14] | 贾娟, 骆开均, 陈艳芳, 赵白静, 李茂伟. 以3-乙酰基樟脑作为辅助配体的环金属铱配合物的合成及光电性能研究[J]. 化学学报, 2012, 70(08): 1028-1034. |
| [15] | 曹韵波. D-A型环金属铱(III)配合物磷光材料的合成及其光电性能的研究[J]. 化学学报, 2011, 69(03): 325-334. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||