Chinese Journal of Organic Chemistry ›› 2022, Vol. 42 ›› Issue (1): 147-159.DOI: 10.6023/cjoc202108006 Previous Articles Next Articles
Special Issue: 有机氟化学虚拟合辑
ARTICLES
收稿日期:2021-08-06
修回日期:2021-09-05
发布日期:2021-09-14
通讯作者:
陆熹, 傅尧
基金资助:
Zhe Chang, Jiaxin Wang, Xi Lu( ), Yao Fu(
), Yao Fu( )
)
Received:2021-08-06
Revised:2021-09-05
Published:2021-09-14
Contact:
Xi Lu, Yao Fu
Supported by:Share
Zhe Chang, Jiaxin Wang, Xi Lu, Yao Fu. Synthesis of gem-Difluoroalkenes through Nickel-Promoted Electrochemical Reductive Cross-Coupling[J]. Chinese Journal of Organic Chemistry, 2022, 42(1): 147-159.
| Entry | Variation from standard conditions | Conversionb/% of 1a | Yieldb/% of 4aa | |
|---|---|---|---|---|
| 1 | No | 81 | 77 (73c) | |
| 2 | NiCl2 in lieu of NiBr2(diglyme) | 86 | 76 | |
| 3 | Ni(acac)2 in lieu of NiBr2(diglyme) | 76 | 52 | |
| 4 | L1 in lieu of Pybox | 74 | 40 | |
| 5 | L2 in lieu of Pybox | 62 | 41 | |
| 6 | L3 in lieu of Pybox | 52 | 48 | |
| 7 | KBr in lieu of nBu4NBr | 78 | 72 | |
| 8 | LiBr in lieu of nBu4NBr | 77 | 62 | |
| 9 | 0.5 equiv. nBu4NBr | 76 | 75 | |
| 10 | 2.0 equiv. nBu4NBr | 74 | 70 | |
| 11 | DMAc in lieu of DMSO | 66 | 57 | |
| 12 | DMF in lieu of DMSO | 78 | 53 | |
| 13 | CH3CN in lieu of DMSO | 88 | <2 | |
| 14 | n(1a)∶n(2a)=1∶2 | 84 | 70 | |
| 15 | n(1a)∶n(2a)=2∶1 | 66 | 53 | |
| 16 | No NiBr2(diglyme) | 79 | 44 | |
| 17 | No electric current | 41 | 39 | |
| 18 | No NiBr2(diglyme), no electric current | 25 | 15 | |
| 19 | Fe(+) in lieu of Zn(+) | 70 | 55 | |
| 20 | Al(+) in lieu of Zn(+) | 95 | <2 | |
| 21 | Fe powder in lieu of electric current | <2 | <2 | |
| 22 | Al powder in lieu of electric current | <2 | <2 | |
| 23 | RVC(—) in lieu of Ni foam(—) | 87 | 73 | |
| 24 | RVC(—) in lieu of Ni foam(—), no NiBr2(diglyme) | 82 | 10 | |
| 25 | Graphite(—) in lieu of Ni foam(—) | 69 | 48 | |
| 26 | Graphite(—) in lieu of Ni foam(—), no NiBr2(diglyme) | 60 | 13 | |
| 27 | Pt(—) in lieu of Ni foam(—) | 81 | 35 | |
| 28 | 3a in lieu of 2a | 89 | 88 (84c) | |
| 29 | 3a in lieu of 2a, no NiBr2(diglyme) | 83 | 64 | |
| 30 | 3a in lieu of 2a, no electric current | 70 | 69 | |
| 31 | 3a in lieu of 2a, no NiBr2(diglyme), no electric current | 69 | 67 | |
| 32 | 3a in lieu of 2a, Fe(+) in lieu of Zn(+) | 60 | 42 | |
| 33 | 3a in lieu of 2a, Al(+) in lieu of Zn(+) | >99 | 31 | |
| 34 | 3a in lieu of 2a, Fe powder in lieu of electric current | <2 | <2 | |
| 35 | 3a in lieu of 2a, Al powder in lieu of electric current | <2 | <2 | |
| 36 | 3a in lieu of 2a, n(1a)∶n(3a)=1∶1.5 | 50 | 50 | |
| 37 | 3a in lieu of 2a, n(1a)∶n(3a)=1∶2 | 77 | 52 | |
| 38 | 3a in lieu of 2a, n(1a)∶n(3a)=1∶2.5 | 82 | 76 | |
| 39 | 3a in lieu of 2a, n(1a)∶n(3a)=2.5∶1 | 70 | 37 | |
| Entry | Variation from standard conditions | Conversionb/% of 1a | Yieldb/% of 4aa | |
|---|---|---|---|---|
| 1 | No | 81 | 77 (73c) | |
| 2 | NiCl2 in lieu of NiBr2(diglyme) | 86 | 76 | |
| 3 | Ni(acac)2 in lieu of NiBr2(diglyme) | 76 | 52 | |
| 4 | L1 in lieu of Pybox | 74 | 40 | |
| 5 | L2 in lieu of Pybox | 62 | 41 | |
| 6 | L3 in lieu of Pybox | 52 | 48 | |
| 7 | KBr in lieu of nBu4NBr | 78 | 72 | |
| 8 | LiBr in lieu of nBu4NBr | 77 | 62 | |
| 9 | 0.5 equiv. nBu4NBr | 76 | 75 | |
| 10 | 2.0 equiv. nBu4NBr | 74 | 70 | |
| 11 | DMAc in lieu of DMSO | 66 | 57 | |
| 12 | DMF in lieu of DMSO | 78 | 53 | |
| 13 | CH3CN in lieu of DMSO | 88 | <2 | |
| 14 | n(1a)∶n(2a)=1∶2 | 84 | 70 | |
| 15 | n(1a)∶n(2a)=2∶1 | 66 | 53 | |
| 16 | No NiBr2(diglyme) | 79 | 44 | |
| 17 | No electric current | 41 | 39 | |
| 18 | No NiBr2(diglyme), no electric current | 25 | 15 | |
| 19 | Fe(+) in lieu of Zn(+) | 70 | 55 | |
| 20 | Al(+) in lieu of Zn(+) | 95 | <2 | |
| 21 | Fe powder in lieu of electric current | <2 | <2 | |
| 22 | Al powder in lieu of electric current | <2 | <2 | |
| 23 | RVC(—) in lieu of Ni foam(—) | 87 | 73 | |
| 24 | RVC(—) in lieu of Ni foam(—), no NiBr2(diglyme) | 82 | 10 | |
| 25 | Graphite(—) in lieu of Ni foam(—) | 69 | 48 | |
| 26 | Graphite(—) in lieu of Ni foam(—), no NiBr2(diglyme) | 60 | 13 | |
| 27 | Pt(—) in lieu of Ni foam(—) | 81 | 35 | |
| 28 | 3a in lieu of 2a | 89 | 88 (84c) | |
| 29 | 3a in lieu of 2a, no NiBr2(diglyme) | 83 | 64 | |
| 30 | 3a in lieu of 2a, no electric current | 70 | 69 | |
| 31 | 3a in lieu of 2a, no NiBr2(diglyme), no electric current | 69 | 67 | |
| 32 | 3a in lieu of 2a, Fe(+) in lieu of Zn(+) | 60 | 42 | |
| 33 | 3a in lieu of 2a, Al(+) in lieu of Zn(+) | >99 | 31 | |
| 34 | 3a in lieu of 2a, Fe powder in lieu of electric current | <2 | <2 | |
| 35 | 3a in lieu of 2a, Al powder in lieu of electric current | <2 | <2 | |
| 36 | 3a in lieu of 2a, n(1a)∶n(3a)=1∶1.5 | 50 | 50 | |
| 37 | 3a in lieu of 2a, n(1a)∶n(3a)=1∶2 | 77 | 52 | |
| 38 | 3a in lieu of 2a, n(1a)∶n(3a)=1∶2.5 | 82 | 76 | |
| 39 | 3a in lieu of 2a, n(1a)∶n(3a)=2.5∶1 | 70 | 37 | |
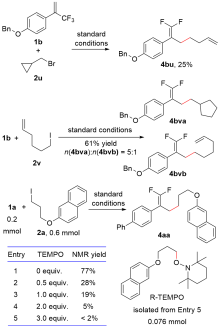
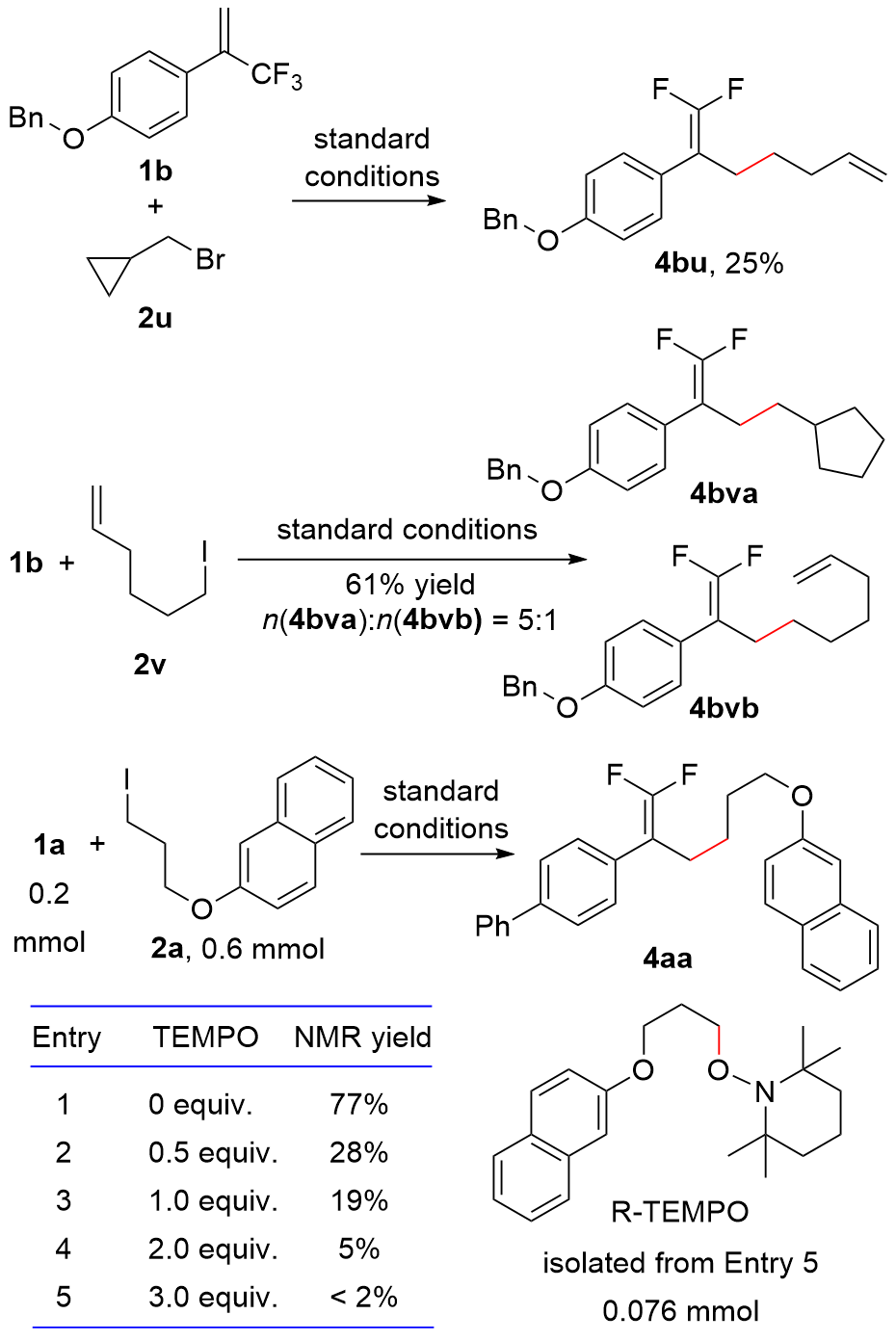
| [1] |
(a) Zhang, X.; Cao, S. Tetrahedron Lett. 2017, 58, 375.
doi: 10.1016/j.tetlet.2016.12.054 |
|
(b) Du, B.; Chan, C.-M.; Lee, P.-Y.; Cheung, L.-H.; Xu, X.; Lin, Z.; Yu, W.-Y. Nat. Commun. 2021, 12, 412.
doi: 10.1038/s41467-020-20725-9 |
|
|
(c) Zhang, X.-J.; Cheng, Y.-M.; Zhao, X.-W.; Cao, Z.-Y.; Xiao, X.; Xu, Y. Org. Chem. Front. 2021, 8, 2315.
doi: 10.1039/D0QO01630F |
|
|
(d) Zhuo, K.-F.; Xu, W.-Y.; Gong, T.-J.; Fu, Y. Chem. Commun. 2020, 56, 2340.
doi: 10.1039/C9CC08485A |
|
|
(e) Ma, X.; Song, Q. Chem. Soc. Rev. 2020, 49, 9197.
doi: 10.1039/D0CS00604A |
|
|
(f) Sheng, J.; Wu, N.; Liu, X.; Liu, F.; Liu, S.; Ding, W.; Liu, C.; Cheng, X. Chin. J. Org. Chem. 2020, 40, 3873. (in Chinese)
doi: 10.6023/cjoc202006071 |
|
|
(盛杰, 吴娜, 刘旭, 刘峰, 刘帅, 丁伟杰, 刘畅, 程旭, 有机化学, 2020, 40, 3873.)
doi: 10.6023/cjoc202006071 |
|
|
(g) Shen, X.; Liu, Q.; Ni, C.; Hu, J. Chin. J. Chem. 2014, 32, 703.
doi: 10.1002/cjoc.201400403 |
|
|
(h) Xiong, B.; Wang, T.; Sun, H.; Li, Y.; Kramer, S.; Cheng, G.-J.; Lian, Z. ACS Catal. 2020, 10, 13616.
doi: 10.1021/acscatal.0c03993 |
|
| [2] |
(a) Fujita, T.; Fuchibe, K.; Ichikawa, J. Angew. Chem., Int. Ed. 2018, 58, 390.
doi: 10.1002/anie.v58.2 |
|
(b) Cai, Y.; Tan, D.; Zhang, Q.; Lü, W.; Li, Q.; Wang, H. Chin. Chem. Lett. 2021, 32, 417.
doi: 10.1016/j.cclet.2020.03.031 |
|
| [3] |
(a) He, S.-J.; Pi, J.-J.; Li, Y.; Lu, X.; Fu, Y. Acta Chim. Sinica 2018, 76, 956. (in Chinese)
doi: 10.6023/A18080333 |
|
(何世江, 皮静静, 李炎, 陆熹, 傅尧, 化学学报, 2018, 76, 956.)
doi: 10.6023/A18080333 |
|
|
(b) Song, S.; Liu, H.; Wang, L.; Zhu, C.; Loh, T. P.; Feng, C. Chin. J. Chem. 2019, 37, 1036.
doi: 10.1002/cjoc.v37.10 |
|
|
(c) Gao, P.; Wang, G.; Xi, L.; Wang, M.; Li, S.; Shi, Z. Chin. J. Chem. 2019, 37, 1009.
doi: 10.1002/cjoc.v37.10 |
|
|
(d) Zhang, J.; Wang, B.; Liu, Y.; Cao, S. Chin. J. Org. Chem. 2019, 39, 249. (in Chinese)
doi: 10.6023/cjoc201807013 |
|
|
(张娟, 王碧云, 刘熠森, 曹松, 有机化学, 2019, 39, 249.)
doi: 10.6023/cjoc201807013 |
|
|
(e) He, S.-Y.; Zhang, X.-G. Org. Chem. Front. 2020, 7, 3174.
doi: 10.1039/D0QO00818D |
|
| [4] |
(a) Hu, X.-S.; Ding, P.-G.; Yu, J.-S.; Zhou, J. Org. Chem. Front. 2019, 6, 2500.
doi: 10.1039/C9QO00577C |
|
(b) Huang, X.; Zhao, W.; Liang, Y.; Wang, M.; Zhan, Y.; Zhang, Y.; Kong, L.; Wang, Z.-X.; Peng, B. Org. Chem. Front. 2021, 8, 1280.
doi: 10.1039/D0QO01513J |
|
| [5] |
(a) Lin, T.-Y.; Pan, Z.; Tu, Y.; Zhu, S.; Wu, H.-H.; Liu, Y.; Li, Z.; Zhang, J. Angew. Chem., Int. Ed. 2020, 59, 22957.
doi: 10.1002/anie.v59.51 |
|
(b) Hu, J.; Yang, Y.; Lou, Z.; Ni, C.; Hu, J. Chin. J. Chem. 2018, 36, 1202.
doi: 10.1002/cjoc.v36.12 |
|
| [6] |
Magueur, G.; Crousse, B.; Ourévitch, M.; Bonnet-Delpon, D.; Bégué, J.-P. J. Fluorine Chem. 2006, 127, 637.
doi: 10.1016/j.jfluchem.2005.12.013 |
| [7] |
(a) Qin, W.; Chen, J.; Xiong, W.; Liu, G. Chin. J. Org. Chem. 2020, 40, 3177. (in Chinese)
doi: 10.6023/cjoc202005016 |
|
(秦文兵, 陈嘉怡, 熊威, 刘国凯, 有机化学, 2020, 40, 3177.)
doi: 10.6023/cjoc202005016 |
|
|
(b) Jin, Y.; Wang, Y.; Bao, K.; Sheng, R.; Tao, X. Chin. J. Org. Chem. 2019, 39, 2726. (in Chinese)
doi: 10.6023/cjoc201903063 |
|
|
(陶雪芬, 盛荣, 鲍堃, 王玉新, 金银秀, 有机化学, 2019, 39, 2726.)
doi: 10.6023/cjoc201903063 |
|
|
(c) Xie, Q.; Hu, J. Chin. J. Chem. 2020, 38, 202.
doi: 10.1002/cjoc.v38.2 |
|
| [8] |
(a) Zhao, Y.; Huang, W.; Zhu, L.; Hu, J. Org. Lett. 2010, 12, 1444.
doi: 10.1021/ol100090r |
|
(b) Hu, M.; He, Z.; Gao, B.; Li, L.; Ni, C.; Hu, J. J. Am. Chem. Soc. 2013, 135, 17302.
doi: 10.1021/ja409941r |
|
|
(c) Hu, M.; Ni, C.; Li, L.; Han, Y.; Hu, J. J. Am. Chem. Soc. 2015, 137, 14496.
doi: 10.1021/jacs.5b09888 |
|
| [9] |
(a) Zheng, J.; Cai, J.; Lin, J.-H.; Guo, Y.; Xiao, J.-C. Chem. Commun. 2013, 49, 7513.
doi: 10.1039/c3cc44271c |
|
(b) Yu, J.; Lin, J.; Xiao, J. Chin. J. Org. Chem. 2019, 39, 265. (in Chinese)
|
|
|
(于蛟, 林锦鸿, 肖吉昌, 有机化学, 2019, 39, 265.)
doi: 10.6023/cjoc201806024 |
|
| [10] |
Zhang, Z.; Yu, W.; Wu, C.; Wang, C.; Zhang, Y.; Wang, J. Angew. Chem., Int. Ed. 2016, 55, 273.
doi: 10.1002/anie.201509711 |
| [11] |
(a) Zeng, H.; Cai, Y.; Jiang, H.; Zhu, C. Org. Lett. 2021, 23, 66.
doi: 10.1021/acs.orglett.0c03708 pmid: 30680998 |
|
(b) Liu, Y.; Zhou, Y.; Zhao, Y.; Qu, J. Org. Lett. 2017, 19, 946.
doi: 10.1021/acs.orglett.7b00168 pmid: 30680998 |
|
|
(c) Zeng, H.; Zhu, C.; Jiang, H. Org. Lett. 2019, 21, 1130.
doi: 10.1021/acs.orglett.9b00074 pmid: 30680998 |
|
|
(d) Cai, Y.; Zeng, H.; Zhu, C.; Liu, C.; Liu, G.; Jiang, H. Org. Chem. Front. 2020, 7, 1260.
doi: 10.1039/D0QO00121J pmid: 30680998 |
|
|
(e) Wu, X.; Xie, F.; Gridnev, I. D.; Zhang, W. Org. Lett. 2018, 20, 1638.
doi: 10.1021/acs.orglett.8b00379 pmid: 30680998 |
|
|
(f) Gao, P.; Yuan, C.; Zhao, Y.; Shi, Z. Chem 2018, 4, 2201.
doi: 10.1016/j.chempr.2018.07.003 pmid: 30680998 |
|
|
(g) Gao, P.; Gao, L.; Xi, L.; Zhang, Z.; Li, S.; Shi, Z. Org. Chem. Front. 2020, 7, 2618.
doi: 10.1039/D0QO00773K pmid: 30680998 |
|
|
(h) Chen, G.; Wang, L.; Liu, X.; Liu, P. Adv. Synth. Catal. 2020, 362, 2990.
doi: 10.1002/adsc.v362.14 pmid: 30680998 |
|
|
(i) Yao, C.; Wang, S.; Norton, J.; Hammond, M. J. Am. Chem. Soc. 2020, 142, 4793.
doi: 10.1021/jacs.9b13757 pmid: 30680998 |
|
|
(j) Chen, F.; Xu, X.; He, Y.; Huang, G.; Zhu, S. Angew. Chem., Int. Ed. 2020, 59, 5398.
doi: 10.1002/anie.v59.13 pmid: 30680998 |
|
|
(k) Fuchibe, K.; Hatta, H.; Oh, K.; Oki, R.; Ichikawa, J. Angew. Chem., Int. Ed. 2017, 56, 5890.
doi: 10.1002/anie.201701985 pmid: 30680998 |
|
|
(l) Fuchibe, K.; Takahashi, M.; Ichikawa, J. Angew. Chem., Int. Ed. 2012, 51, 12059.
doi: 10.1002/anie.201206946 pmid: 30680998 |
|
|
(m) Ichitsuka, T.; Fujita, T.; Arita, T.; Ichikawa, J. Angew. Chem., Int. Ed. 2014, 53, 7564.
doi: 10.1002/anie.201402695 pmid: 30680998 |
|
|
(n) Ichitsuka, T.; Fujita, T.; Ichikawa, J. ACS Catal. 2015, 5, 5947.
doi: 10.1021/acscatal.5b01463 pmid: 30680998 |
|
|
(o) Jaroschik, F. Chem. Eur. J. 2018, 24, 14572.
doi: 10.1002/chem.v24.55 pmid: 30680998 |
|
|
(p) Kojima, R.; Akiyama, S.; Ito, H. Angew. Chem., Int. Ed. 2018, 57, 7196.
doi: 10.1002/anie.v57.24 pmid: 30680998 |
|
|
(q) Kojima, R.; Kubota, K.; Ito, H. Chem. Commun. 2017, 53, 10688.
doi: 10.1039/C7CC05225A pmid: 30680998 |
|
|
(r) Kojima, Y.; Takata, T.; Hirano, K.; Miura, M. Chem. Lett. 2020, 49, 637.
doi: 10.1246/cl.200163 pmid: 30680998 |
|
|
(s) Liu, Y.; Li, C.; Liu, C.; He, J.; Zhao, X.; Cao, S. Tetrahedron Lett. 2020, 61, 151940.
doi: 10.1016/j.tetlet.2020.151940 pmid: 30680998 |
|
|
(t) Liu, Z.; Tu, X.-S.; Guo, L.-T.; Wang, X.-C. Chem. Sci. 2020, 11, 11548.
doi: 10.1039/D0SC03883K pmid: 30680998 |
|
|
(u) Yan, S.-S.; Wu, D.-S.; Ye, J.-H.; Gong, L.; Zeng, X.; Ran, C.-K.; Gui, Y.-Y.; Li, J.; Yu, D.-G. ACS Catal. 2019, 9, 6987.
doi: 10.1021/acscatal.9b02351 pmid: 30680998 |
|
|
(v) Pan, Q.; Ping, Y.; Wang, Y.; Guo, Y.; Kong, W. J. Am. Chem. Soc. 2021, 143, 10282.
doi: 10.1021/jacs.1c03827 pmid: 30680998 |
|
|
(w) Tian, F.; Yan, G.; Yu, J. Chem. Commun. 2019, 55, 13486.
doi: 10.1039/C9CC06465F pmid: 30680998 |
|
|
(x) Xie, S.-L.; Cui, X.-Y.; Gao, X.-T.; Zhou, F.; Wu, H.-H.; Zhou, J. Org. Chem. Front. 2019, 6, 3678.
doi: 10.1039/C9QO00923J pmid: 30680998 |
|
|
(y) Li, Y.; Wang, Y.; Zhu, L.; Qu, L.; Lan, Y. Chin. J. Org. Chem. 2019, 39, 38. (in Chinese)
doi: 10.6023/cjoc201810020 pmid: 30680998 |
|
|
(李园园, 王元鉴, 朱磊, 屈凌波, 蓝宇, 有机化学, 2019, 39, 38.)
doi: 10.6023/cjoc201810020 pmid: 30680998 |
|
| [12] |
Dai, W.; Lin, Y.; Wan, Y.; Cao, S. Org. Chem. Front. 2018, 5, 55.
doi: 10.1039/C7QO00716G |
| [13] |
Wang, M.; Pu, X.; Zhao, Y.; Wang, P.; Li, Z.; Zhu, C.; Shi, Z. J. Am. Chem. Soc. 2018, 140, 9061.
doi: 10.1021/jacs.8b04902 |
| [14] |
Miura, T.; Ito, Y.; Murakami, M. Chem. Lett. 2008, 37, 1006.
doi: 10.1246/cl.2008.1006 |
| [15] |
Huang, Y.; Hayashi, T. J. Am. Chem. Soc. 2016, 138, 12340.
doi: 10.1021/jacs.6b07844 |
| [16] |
Cheng, R.; Xu, C.; Zhang, X. Chin. J. Org. Chem. 2020, 40, 3307. (in Chinese)
doi: 10.6023/cjoc202005082 |
|
(程然, 徐畅, 张新刚, 有机化学, 2020, 40, 3307.)
doi: 10.6023/cjoc202005082 |
|
| [17] |
(a) Phelan, J. P.; Wiles, R. J.; Lang, S. B.; Kelly, C. B.; Molander, G. A. Chem. Sci. 2018, 9, 3215.
doi: 10.1039/c7sc05420c pmid: 29732105 |
|
(b) Lang, S. B.; Wiles, R. J.; Kelly, C. B.; Molander, G. A. Angew. Chem., Int. Ed. 2017, 56, 15073.
doi: 10.1002/anie.201709487 pmid: 29732105 |
|
|
(c) Wiles, R. J.; Phelan, J. P.; Molander, G. A. Chem. Commun. 2019, 55, 7599.
doi: 10.1039/C9CC04265B pmid: 29732105 |
|
| [18] |
(a) Chen, H.; Anand, D.; Zhou, L. Asian J. Org. Chem. 2019, 8, 661.
doi: 10.1002/ajoc.v8.5 |
|
(b) Li, L.; Xiao, T.; Chen, H.; Zhou, L. Chem. Eur. J. 2017, 23, 2249.
doi: 10.1002/chem.201605919 |
|
|
(c) Anand, D.; Sun, Z.; Zhou, L. Org. Lett. 2020, 22, 2371.
doi: 10.1021/acs.orglett.0c00568 |
|
|
(d) Chen, H.; Xiao, T.; Li, L.; Anand, D.; He, Y.; Zhou, L. Adv. Synth. Catal. 2017, 359, 3642.
doi: 10.1002/adsc.v359.20 |
|
|
(e) He, Y.; Anand, D.; Sun, Z.; Zhou, L. Org. Lett. 2019, 21, 3769.
doi: 10.1021/acs.orglett.9b01210 |
|
|
(f) Xiao, T.; Li, L.; Zhou, L. J. Org. Chem. 2016, 81, 7908.
doi: 10.1021/acs.joc.6b01620 |
|
| [19] |
(a) Guo, Y.-Q.; Wang, R.; Song, H.; Liu, Y.; Wang, Q. Org. Lett. 2020, 22, 709.
doi: 10.1021/acs.orglett.9b04504 |
|
(b) Guo, Y.-Q.; Wu, Y.; Wang, R.; Song, H.; Liu, Y.; Wang, Q. Org. Lett. 2021, 23, 2353.
doi: 10.1021/acs.orglett.1c00546 |
|
|
(c) Xiang, P.; He, L.; Li, H.; Qi, Z.; Zhang, M.; Fu, Q.; Wei, J.; Du, X.; Yi, D.; Wei, S. Tetrahedron Lett. 2020, 61, 152369.
doi: 10.1016/j.tetlet.2020.152369 |
|
| [20] |
Lu, X.; Wang, Y.; Zhang, B.; Pi, J.-J.; Wang, X.-X.; Gong, T.-J.; Xiao, B.; Fu, Y. J. Am. Chem. Soc. 2017, 139, 12632.
doi: 10.1021/jacs.7b06469 pmid: 28849923 |
| [21] |
Lu, X.; Wang, X.-X.; Gong, T.-J.; Pi, J.-J.; He, S.-J.; Fu, Y. Chem. Sci. 2019, 10, 809.
doi: 10.1039/C8SC04335C |
| [22] |
Lan, Y.; Yang, F.; Wang, C. ACS Catal. 2018, 8, 9245.
doi: 10.1021/acscatal.8b02784 |
| [23] |
(a) Yu, L.; Tang, M.-L.; Si, C.-M.; Meng, Z.; Liang, Y.; Han, J.; Sun, X. Org. Lett. 2018, 20, 4579.
doi: 10.1021/acs.orglett.8b01866 |
|
(b) Du, H.-W.; Chen, Y.; Sun, J.; Gao, Q.-S.; Wang, H.; Zhou, M.-D. S. Org. Lett. 2020, 22, 9342.
doi: 10.1021/acs.orglett.0c03554 |
|
|
(c) Li, Z.; Wang, K.-F.; Zhao, X.; Ti, H.; Liu, X.-G.; Wang, H. Nat. Commun. 2020, 11, 5036.
doi: 10.1038/s41467-020-18834-6 |
|
|
(d) Lin, Z.; Lan, Y.; Wang, C. Org. Lett. 2019, 21, 8316.
doi: 10.1021/acs.orglett.9b03102 |
|
| [24] |
(a) Ding, D.; Lan, Y.; Lin, Z.; Wang, C. Org. Lett. 2019, 21, 2723.
doi: 10.1021/acs.orglett.9b00692 |
|
(b) Lin, Z.; Lan, Y.; Wang, C. Org. Lett. 2020, 22, 3509.
doi: 10.1021/acs.orglett.0c00960 |
|
|
(c) Lu, X.-Y.; Jiang, R.-C.; Li, J.-M.; Liu, C.-C.; Wang, Q.-Q.; Zhou, H.-P. Org. Biomol. Chem. 2020, 18, 3674.
doi: 10.1039/D0OB00535E |
|
|
(d) Fan, P.; Zhang, C.; Lan, Y.; Lin, Z.; Zhang, L.; Wang, C. Chem. Commun. 2019, 55, 12691.
doi: 10.1039/C9CC07285C |
|
|
(e) Jin, Y.; Wu, J.; Lin, Z.; Lan, Y.; Wang, C. Org. Lett. 2020, 22, 5347.
doi: 10.1021/acs.orglett.0c01592 |
|
|
(f) Zhu, C.; Liu, Z.-Y.; Tang, L.; Zhang, H.; Zhang, Y.-F.; Walsh, P. J.; Feng, C. Nat. Commun. 2020, 11, 4860.
doi: 10.1038/s41467-020-18658-4 |
|
| [25] |
(a) Kawamata, Y.; Vantourout, J. C.; Hickey, D. P.; Bai, P.; Chen, L.; Hou, Q.; Qiao, W.; Barman, K.; Edwards, M. A.; Garrido-Castro, A. F.; deGruyter, J. N.; Nakamura, H.; Knouse, K.; Qin, C.; Clay, K. J.; Bao, D.; Li, C.; Starr, J. T.; Garcia-Irizarry, C.; Sach, N.; White, H. S.; Neurock, M.; Minteer, S. D.; Baran, P. S. J. Am. Chem. Soc. 2019, 141, 6392.
doi: 10.1021/jacs.9b01886 pmid: 30905151 |
|
(b) Kim, H.; Kim, H.; Lambert, T. H.; Lin, S. J. Am. Chem. Soc. 2020, 142, 2087.
doi: 10.1021/jacs.9b10678 pmid: 30905151 |
|
|
(c) Li, C.; Kawamata, Y.; Nakamura, H.; Vantourout, J. C.; Liu, Z.; Hou, Q.; Bao, D.; Starr, J. T.; Chen, J.; Yan, M.; Baran, P. S. Angew. Chem., Int. Ed. 2017, 56, 13088.
doi: 10.1002/anie.201707906 pmid: 30905151 |
|
|
(d) Wang, P.; Yang, Z.; Wang, Z.; Xu, C.; Huang, L.; Wang, S.; Zhang, H.; Lei, A. Angew. Chem., Int. Ed. 2019, 58, 15747.
doi: 10.1002/anie.v58.44 pmid: 30905151 |
|
|
(e) Liu, Q.; Sun, B.; Liu, Z.; Kao, Y.; Dong, B.-W.; Jiang, S.-D.; Li, F.; Liu, G.; Yang, Y.; Mo, F. Chem. Sci. 2018, 9, 8731.
doi: 10.1039/C8SC03346C pmid: 30905151 |
|
|
(f) Liu, D.; Ma, H.-X.; Fang, P.; Mei, T.-S. Angew. Chem., Int. Ed. 2019, 58, 5033.
doi: 10.1002/anie.v58.15 pmid: 30905151 |
|
|
(g) Zou, Z.; Zhang, W.; Wang, Y.; Pan, Y. Org. Chem. Front. 2021, 8, 2786.
doi: 10.1039/D1QO00054C pmid: 30905151 |
|
|
(h) Lu, L.; Li, H.; Zheng, Y.; Bu, F.; Lei, A. CCS Chem. 2020, 2, 2669.
pmid: 30905151 |
|
|
(i) Dou, G.-Y.; Jiang, Y.-Y.; Xu, K.; Zeng, C.-C. Org. Chem. Front. 2019, 6, 2392.
doi: 10.1039/C9QO00552H pmid: 30905151 |
|
|
(j) Liang, K.; Wang, S.; Cong, H.; Lu, L.; Lei, A. CCS Chem. 2021, 3, 1727.
pmid: 30905151 |
|
|
(k) Huang, C.; Xu, H.-C. Sci. China Chem. 2019, 62, 1501.
doi: 10.1007/s11426-019-9554-1 pmid: 30905151 |
|
|
(l) Feng, E.; Hou, Z.; Xu, H. Chin. J. Org. Chem. 2019, 39, 1424. (in Chinese)
doi: 10.6023/cjoc201812007 pmid: 30905151 |
|
|
(冯恩祺, 侯中伟, 徐海超, 有机化学, 2019, 39, 1424.)
doi: 10.6023/cjoc201812007 pmid: 30905151 |
|
|
(m) Zhang, C.; Bu, F.; Zeng, C.; Wang, D.; Lu, L.; Zhang, H.; Lei, A. CCS Chem. 2021, 3, 1404.
pmid: 30905151 |
|
|
(n) Deng, Y.; Lu, F.; You, S.; Xia, T.; Zheng, Y.; Lu, C.; Yang, G.; Chen, Z.; Gao, M.; Lei, A. Chin. J. Chem. 2019, 37, 817.
doi: 10.1002/cjoc.v37.8 pmid: 30905151 |
|
|
(o) Hou, Z. W.; Xu, H. C. Chin. J. Chem. 2020, 38, 394.
doi: 10.1002/cjoc.v38.4 pmid: 30905151 |
|
|
(p) Liu, S.; Zhao, W.; Li, J.; Wu, N.; Liu, C.; Wang, X.; Li, S.; Zhu, Y.; Liang, Y.; Cheng, X. CCS Chem. 2021, 3, 872.
pmid: 30905151 |
|
|
(q) Qiu, Y.; Zhu, C.; Stangier, M.; Struwe, J.; Ackermann, L. CCS Chem. 2020, 2, 1529.
pmid: 30905151 |
|
|
(r) Xu, Y.; Lin, L.; Han, Y.; Liu, Y. Chin. J. Org. Chem. 2021, 41, 934. (in Chinese)
doi: 10.6023/cjoc202008017 pmid: 30905151 |
|
|
(许颖, 林立青, 韩莹徽, 刘颖杰, 有机化学, 2021, 41, 934.)
doi: 10.6023/cjoc202008017 pmid: 30905151 |
|
|
(s) Yang, Q.-L.; Wang, X.-Y.; Weng, X.-J.; Yang, X.; Xu, X.-T.; Tong, X.; Fang, P.; Wu, X.-Y.; Mei, T.-S. Acta Chim. Sinica 2019, 77, 866. (in Chinese)
doi: 10.6023/A19040135 pmid: 30905151 |
|
|
(杨启亮, 王向阳, 翁信军, 杨祥, 徐学涛, 童晓峰, 方萍, 伍新燕, 梅天胜, 化学学报, 2019, 77, 866.)
doi: 10.6023/A19040135 pmid: 30905151 |
|
|
(t) Zhang, Q.; Liang, K.; Guo, C. CCS Chem. 2021, 3, 338.
doi: 10.31635/ccschem.021.202000720 pmid: 30905151 |
|
| [26] |
(a) Li, H.; Breen, C. P.; Seo, H.; Jamison, T. F.; Fang, Y.-Q.; Bio, M. M. Org. Lett. 2018, 20, 1338.
doi: 10.1021/acs.orglett.8b00070 |
|
(b) Jiao, K.-J.; Li, Z.-M.; Xu, X.-T.; Zhang, L.-P.; Li, Y.-Q.; Zhang, K.; Mei, T.-S. Org. Chem. Front. 2018, 5, 2244.
doi: 10.1039/C8QO00507A |
|
|
(c) Lai, Y.-L.; Huang, J.-M. Org. Lett. 2017, 19, 2022.
doi: 10.1021/acs.orglett.7b00473 |
|
|
(d) Qiu, H.; Shuai, B.; Wang, Y.-Z.; Liu, D.; Chen, Y.-G.; Gao, P.-S.; Ma, H.-X.; Chen, S.; Mei, T.-S. J. Am. Chem. Soc. 2020, 142, 9872.
doi: 10.1021/jacs.9b13117 |
|
| [27] |
(a) Chen, X.; Luo, X.; Peng, X.; Guo, J.; Zai, J.; Wang, P. Chem. Eur. J. 2020, 26, 3226.
doi: 10.1002/chem.v26.15 pmid: 28704055 |
|
(b) Gomes, P.; Gosmini, C.; Périchon, J. J. Org. Chem. 2003, 68, 1142.
doi: 10.1021/jo026421b pmid: 28704055 |
|
|
(c) Gomes, P.; Gosmini, C.; Périchon, J. Tetrahedron 2003, 59, 2999.
doi: 10.1016/S0040-4020(03)00404-6 pmid: 28704055 |
|
|
(d) Jiao, K.-J.; Liu, D.; Ma, H.-X.; Qiu, H.; Fang, P.; Mei, T.-S. Angew. Chem., Int. Ed. 2020, 59, 6520.
doi: 10.1002/anie.v59.16 pmid: 28704055 |
|
|
(e) Kumar, G. S.; Peshkov, A.; Brzozowska, A.; Nikolaienko, P.; Zhu, C.; Rueping, M. Angew. Chem., Int. Ed. 2020, 59, 6513.
doi: 10.1002/anie.v59.16 pmid: 28704055 |
|
|
(f) Wang, X.-X.; Lu, X.; Li, Y.; Wang, J.-W.; Fu, Y. Sci. China Chem. 2020, 63, 1586.
doi: 10.1007/s11426-020-9838-x pmid: 28704055 |
|
|
(g) Truesdell, B. L.; Hamby, T. B.; Sevov, C. S. J. Am. Chem. Soc. 2020, 142, 5884.
doi: 10.1021/jacs.0c01475 pmid: 28704055 |
|
|
(h) Perkins, R. J.; Pedro, D. J.; Hansen, E. C. Org. Lett. 2017, 19, 3755.
doi: 10.1021/acs.orglett.7b01598 pmid: 28704055 |
|
|
(i) Perkins, R. J.; Hughes, A. J.; Weix, D. J.; Hansen, E. C. Org. Process Res. Dev. 2019, 23, 1746.
doi: 10.1021/acs.oprd.9b00232 pmid: 28704055 |
|
| [28] |
Jiang, W.-T.; Yang, S.; Xu, M.-Y.; Xie, X.-Y.; Xiao, B. Chem. Sci. 2020, 11, 488.
doi: 10.1039/C9SC04288A |
| [29] |
(a) Wang, X.-X.; Yu, L.; Lu, X.; Zhang, Z.-L.; Liu, D.-G.; Tian, C.; Fu, Y. CCS Chem. 2021, 3, 727.
pmid: 27033405 |
|
(b) Lu, X.; Xiao, B.; Zhang, Z.; Gong, T.; Su, W.; Yi, J.; Fu, Y.; Liu, L. Nat. Commun. 2016, 7, 11129.
doi: 10.1038/ncomms11129 pmid: 27033405 |
|
|
(c) Wang, J.-W.; Li, Y.; Nie, W.; Chang, Z.; Yu, Z.-A.; Zhao, Y.-F.; Lu, X.; Fu, Y. Nat. Commun. 2021, 12, 1313.
doi: 10.1038/s41467-021-21600-x pmid: 27033405 |
|
| [30] |
Supranovich, V. I.; Levin, V. V.; Kokorekin, V. A.; Dilman, A. D. Adv. Synth. Catal. 2021, 363, 2888.
doi: 10.1002/adsc.v363.11 |
| [1] | Zhou Zhang, Yu Guo, Jing Yang, Dan Wu, Jiaxin Wang, Xinyue Hong, Peijun Cai, Liangce Rong. Electrochemically Promoted Halogenation of Imidazoland-[1,2-a]pyridine with Dichloro(bromo)ethylene and Iodoform [J]. Chinese Journal of Organic Chemistry, 2023, 43(6): 2104-2109. |
| [2] | Linlin Du, Hua Zhang. Photochemical and Electrochemical Borylation Involving Aryl and Alkyl Compounds [J]. Chinese Journal of Organic Chemistry, 2023, 43(5): 1726-1741. |
| [3] | Shuyong Song, Senmiao Xu. Recent Progress in Selective C-F Bond Activation of Trifluoromethyl Alkenes [J]. Chinese Journal of Organic Chemistry, 2023, 43(2): 411-425. |
| [4] | Wanjie Wei, Lei Zhan, Lei Gao, Guobao Huang, Xianli Ma. Research Progress of Electrochemical Synthesis of C-Sulfonyl Compounds [J]. Chinese Journal of Organic Chemistry, 2023, 43(1): 17-35. |
| [5] | Yu Zheng, Shencheng Qian, Pengcheng Xu, Binnan Zheng, Shenlin Huang. Electrochemical Oxidative Thiocyanosulfonylation of Aryl Acetylenes [J]. Chinese Journal of Organic Chemistry, 2022, 42(12): 4275-4281. |
| [6] | Xiuying Li, Pingfang Tao, Yongyu Cheng, Qiong Hu, Weijuan Huang, Yun Li, Zhihui Luo, Guobao Huang. Recent Progress on the Electrochemical Difunctionalization of Alkenes/Alkynes [J]. Chinese Journal of Organic Chemistry, 2022, 42(12): 4169-4201. |
| [7] | Yuanyuan Ping, Haixia Song, Wangqing Kong. Recent Advances in Ni-Catalyzed Asymmetric Reductive Difunctionalization of Alkenes [J]. Chinese Journal of Organic Chemistry, 2022, 42(10): 3302-3321. |
| [8] | Yingjie Liu, Zhichuan Wang, Jianping Meng, Chen Li, Kai Sun. Research Progress of Photoelectric Co-catalysis [J]. Chinese Journal of Organic Chemistry, 2022, 42(1): 100-110. |
| [9] | Jintao Wu, Zhongquan Liu. Advances in Free-Radical Promoted C(sp3)—C(sp3) Bond Conversion [J]. Chinese Journal of Organic Chemistry, 2022, 42(1): 16-32. |
| [10] | Xiang Liu, Wen Li, Canzhan Zhuang, Hua Cao. Application of Photochemical/Electrochemical Synthesis in C—H Functionalization of Quinoxalin-2(1H)-one [J]. Chinese Journal of Organic Chemistry, 2021, 41(9): 3459-3481. |
| [11] | Junqing Gao, Xinjun Weng, Cong Ma, Xuetao Xu, Ping Fang, Tiansheng Mei. Electrochemical 2,2,6,6-tetramethylpiperidinyl-N-oxyl (TEMPO)-Mediated α-Cyanation and Phosphonylation of Cyclic Amines with Metal-Free Conditions [J]. Chinese Journal of Organic Chemistry, 2021, 41(8): 3223-3234. |
| [12] | Wei Meng, Kun Xu, Bingbing Guo, Chengchu Zeng. Recent Advances in Minisci Reactions under Electrochemical Conditions [J]. Chinese Journal of Organic Chemistry, 2021, 41(7): 2621-2635. |
| [13] | Zeyin Meng, Chengtao Feng, Kun Xu. Recent Advances in the Electrochemical Formation of Carbon-Nitrogen Bonds [J]. Chinese Journal of Organic Chemistry, 2021, 41(7): 2535-2570. |
| [14] | Mei Wu, Ling Yu, Huiqing Hou, Houzheng Chen, Qinglong Zhuang, Sunying Zhou, Xiaoyan Lin. Electrochemistry-Enabled Copper-Catalyzed Oxidation of Benzyl Alcohols for the Preparation of Quinazolinones in Water [J]. Chinese Journal of Organic Chemistry, 2021, 41(6): 2326-2334. |
| [15] | Buqing Cheng, Danhua Ge, Xin Wang, Xueqiang Chu. Perfluoroalkyl Halides as Fluorine-Containing Building Blocks for the Synthesis of Fluoroalkylated Heterocycles [J]. Chinese Journal of Organic Chemistry, 2021, 41(5): 1925-1938. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||