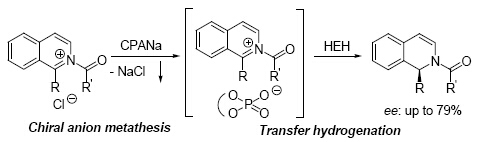
| [1] Akiyama, T.; Itoh, J.; Yokota, K.; Fuchibe, K. Angew. Chem. Int. Ed. 2004, 43, 1566.[2] Uraguchi, D.; Terada, M. J. Am. Chem. Soc. 2004, 126, 5356.[3] Rueping, M.; Sugiono, E.; Azap, C. Angew. Chem. Int. Ed. 2006, [4] Itoh, J.; Fuchibe, K.; Akiyama, T. Angew. Chem. Int. Ed. 2006, 45, 4796.[5] Rueping, M.; Azap, C. Angew. Chem. Int. Ed. 2006, 45, 7832.[6] Liu, H.; Cun, L.-F.; Mi, A.-Q.; Jiang, Y.-Z.; Gong, L.-Z. Org. Lett. 2006, 8, 6023. [7] Lv, J.; Zhong, X.-R.; Cheng, J.-P.; Luo, S.-Z. Acta Chim. Sinica 2012, 70, 1518. (吕健, 钟兴仁, 程津培, 罗三中, 化学学报, 2012, 70, 1518.)[8] Huang, J.-Z.; Luo, S.-W.; Gong, L.-Z. Acta Chim. Sinica 2013, 71, 879. (黄建洲, 罗时玮, 龚流柱, 化学学报, 2013, 71, 879.)[9] Rueping, M.; Sugiono, E.; Azap, C.; Theissmann, T.; Bolte, M. Org. Lett. 2005, 7, 3781.[10] Hoffmann, S.; Seayad, A. M.; List, B. Angew. Chem. Int. Ed. 2005, 44, 7424.[11] Rueping, M.; Antonchick, A. P.; Theissmann, T. Angew. Chem. Int. Ed. 2006, 45, 6751.[12] Li, G.-L.; Liang, Y.-X.; Antilla, J. C. J. Am. Chem. Soc. 2007, 129, 5830. [13] Rueping, M.; Sugiono, E.; Schoepke, F.-R. Synlett 2010, 852.[14] Kang, Q.; Zhao, Z.-A.; You, S.-L. Org. Lett. 2008, 10, 2031. [15] Wang, D.; Hou, C.-J.; Chen, L.-F.; Liu, X.-N.; An, Q.-D.; Hu, X.-P. Chin. J. Org. Chem. 2013, 33, 1355. (王东, 侯传金, 陈丽凤, 刘小宁, 安庆大, 胡向平, 有机化学, 2013, 33, 1355.)[16] Jia, Y.-X.; Zhong, J.; Zhu, S.-F.; Zhang, C.-M.; Zhou, Q.-L. Angew. Chem. Int. Ed. 2007, 46, 5565.[17] Kang, Q.; Zhao, Z.-A.; You, S.-L. J. Am. Chem. Soc. 2007, 129, 1484.[18] Sheng, Y.-F.; Zhang, A.-J.; Zheng, X.-J.; You, S.-L. Chin. J. Org. Chem. 2008, 28, 605. (盛益飞, 张安将, 郑晓建, 游书力, 有机化学, 2008, 28, 605.)[19] Terada, M.; Sorimachi, K. J. Am. Chem. Soc. 2007, 129, 292. [20] He, Z.-R.; Huang, Y.-Y.; Verpoort, F. Acta Chim. Sinica 2013, 71, 700. (何展荣, 黄毅勇, Verpoort Francis, 化学学报, 2013, 71, 700.)[21] Xu, S.; Wang, Z.; Zhang, X.; Zhang, X.; Ding, K. Angew. Chem. Int. Ed. 2008, 47, 2840. [22] Su, Y.-J.; Shi, F.-Q. Chin. J. Org. Chem. 2010, 30, 486. (苏亚军, 史福强, 有机化学, 2010, 30, 486.)[23] Wu, X.; Li, M.-L.; Gong, L.-Z. Acta Chim. Sinica 2013, 71, 1091. (吴祥, 李明丽, 龚流柱, 化学学报, 2013, 71, 1091.)[24] Lv, J.; Luo, S.-Z. Chem. Commun. 2013, 49, 847.[25] Xu, B.; Zhu, S.-F.; Zhang, Z.-C.; Yu, Z.-X.; Ma, Y.; Zhou, Q.-L. Chem. Sci. 2014, 5, 1442.[26] Qiu, H.; Li, M.; Jiang, L.-Q.; Lv, F.-P.; Zan, L.; Zhai, C.-W.; Doyle, M. P.; Hu, W.-H. Nature Chemistry 2012, 4, 733. [27] Qiu, H.; Zhang, D.; Liu, S.-Y.; Qiu, L.; Zhou, J.; Qian, Y.; Zhai, C.-W.; Hu, W.-H. Acta Chim. Sinica 2012, 70, 2484. (邱晃, 张丹, 刘顺英, 邱林, 周俊, 钱宇, 翟昌伟, 胡文浩, 化学学报, 2012, 70, 2484.)[28] Mahlau, M.; List, B. Angew. Chem. Int. Ed. 2013, 52, 518.[29] Rauniyar, V.; Lackner, A. D.; Hamilton, G. L.; Toste, F. D. Science 2011, 334, 1681.[30] Wang, Y.-M.; Wu, J.; Hoong, C.; Rauniyar, V.; Toste, F. D. J. Am. Chem. Soc. 2012, 134, 12928.[31] Phipps, R. J.; Toste, F. D. J. Am. Chem. Soc. 2013, 135, 1268.[32] Lackner, A. D.; Samant, A. V.; Toste, F. D. J. Am. Chem. Soc. 2013, 135, 14090.[33] Zheng, C.; You, S.-L. Chem. Soc. Rev. 2012, 41, 2498.[34] Rueping, M.; Antonchick, A. P.; Theissmann, T. Angew. Chem. Int. Ed. 2006, 45, 3683.[35] Glorius, F. Org. Biomol. Chem. 2005, 3, 4171.[36] Lu, S.-M.; Han, X.-W.; Zhou, Y.-G. Chin. J. Org. Chem. 2005, 25, 634. (卢胜梅, 韩秀文, 周永贵, 有机化学, 2005, 25, 634.)[37] Zhou, Y.-G. Acc. Chem. Res. 2007, 40, 1357.[38] Kuwano, R. Heterocycles 2008, 76, 909.[39] Wang, D.-S.; Chen, Q.-A.; Lu, S.-M.; Zhou, Y.-G. Chem. Rev. 2012, 112, 2557.[40] Xie, J.-H.; Zhou, Q.-L. Acta Chim. Sinica 2012, 70, 1427. (谢建华, 周其林, 化学学报, 2012, 70, 1427.)[41] He, Y.-M.; Song, F.-T.; Fan, Q.-H. Top. Curr. Chem. 2014, 343, 145.[42] Wang, W.-B.; Lu, S.-M.; Yang, P.-Y.; Han, X.-W.; Zhou, Y.-G. J. Am. Chem. Soc. 2003, 125, 10536.[43] Zhou, H.-F.; Li, Z.-W.; Wang, Z.-J.; Wang, T.-L.; Xu, L.-J.; He, Y.-M.; Fan, Q.-H.; Pan, J.; Gu, L.-Q.; Chan, A. S. C. Angew. Chem. Int. Ed. 2008, 47, 8464.[44] Wang, T.-L.; Zhuo, L.-G.; Li, Z.-W.; Chen, F.; Ding, Z.-Y.; He, Y.-M.; Fan, Q.-H.; Xiang, J.-F.; Yu, Z.-X.; Chan, A. S. C. J. Am. Chem. Soc. 2011, 133, 9878.[45] Lei, A.; Chen, M.; He, M.; Zhang, X. Eur. J. Org. Chem. 2006, 4343.[46] Kuwano, R.; Sato, K.; Kurokawa, T.; Karube, D.; Ito, Y. J. Am. Chem. Soc. 2000, 122, 7614.[47] Guo, Q.-S.; Du, D. M.; Xu, J.-X. Angew. Chem. Int. Ed. 2008, 47, 759.[48] Rueping, M.; Tato, F.; Schoepke, F. R. Chem. Eur. J. 2010, 16, 2688. [49] Rueping, M.; Antonchick, A. P. Angew. Chem. Int. Ed. 2007, 46, 4562.[50] Lu, S.-M.; Wang, Y.-Q.; Han, X.-W.; Zhou, Y.-G. Angew. Chem. Int. Ed. 2006, 45, 2260.[51] Shi, L.; Ye, Z.-S.; Cao, L.-L.; Guo, R.-N.; Hu, Y.; Zhou, Y.-G. Angew. Chem. Int. Ed. 2012, 51, 8286.[52] Ye, Z.-S.; Guo, R.-N.; Cai, X.-F.; Chen, M.-W.; Shi, L.; Zhou, Y.-G. Angew. Chem. Int. Ed. 2013, 52, 3685.[53] Iimuro, A.; Yamaji, K.; Kandula, S.; Nagano, T.; Kita, Y.; Mashima, K. Angew. Chem. Int. Ed. 2013, 52, 2046.[54] Guo, R.-N.; Cai, X.-F.; Shi, L.; Ye, Z.-S.; Chen, M.-W.; Zhou, Y.-G. Chem. Commun. 2013, 49, 8537. |