

多功能手性膦催化活泼烯烃的不对称分子间Rauhut-Currier反应
收稿日期: 2016-07-23
网络出版日期: 2016-09-06
基金资助
项目受973计划(No.2015CB856600)、国家自然科学基金(Nos.21372084,21425205)和长江学者与创新团队发展计划(PCSIRT)资助.
Highly Enantioselective Intermolecular Rauhut–Currier Reaction of Activated Alkenes Catalyzed by Multifunctional Chiral Phosphine
Received date: 2016-07-23
Online published: 2016-09-06
Supported by
Project supported by the "973" Programs (No. 2015CB856600), the National Natural Science Foundation of China (Nos. 21372084, 21425205), and the Changjiang Scholars and Innovative Research Team in University (PCSIRT).
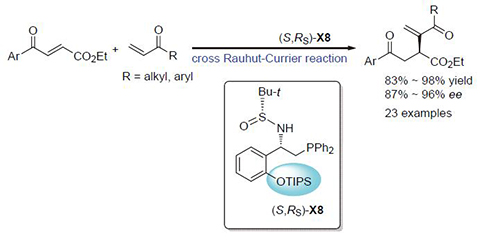
研究实现了多功能手性膦催化的3-芳酰基丙烯酸酯和烯酮的不对称分子间Rauhut-Currier反应,为多羰基手性化合物的构建提供了一种新方法.在使用(S,RS)-X8作为催化剂和甲苯作为溶剂的条件下,一系列含有不同取代基团的3-芳酰基丙烯酸酯和烯酮均可顺利地发生不对称Rauhut-Currier反应,从而高产率且高对映选择性地生成相应的产物.对照实验表明多功能手性膦催化剂中的N-H键对反应的对映选择性的控制起到了至关重要的作用;膦谱监测实验结果表明手性膦催化剂对烯酮的Michael加成是该反应的启动步骤.
关键词: 手性膦催化; Rauhut-Currier反应; 3-芳酰基丙烯酸酯; 烯酮; 亲核催化
周伟 , 高利华 , 陶梦娜 , 宿晓 , 赵庆杰 , 张俊良 . 多功能手性膦催化活泼烯烃的不对称分子间Rauhut-Currier反应[J]. 化学学报, 2016 , 74(10) : 800 -804 . DOI: 10.6023/A16070358
The Rauhut-Currier (R-C) reaction, first disclosed by Rauhut and Currier in 1963, is an atom economy strategy for the construction of carbon-carbon bond and a wide range of synthetic valuable building blocks. During the past few decades, significant progress on the enantioselective intramolecular R-C reactions has been achieved by utilizing diverse chiral nucleophilic catalysts such as cysteine derivatives, L-prolinol, hydrogen-bonding catalyst and β-aminephosphine. However, compared to the well-developed enantioselective intramolecular R-C reactions, the enantioselective cross intermolecular R-C reaction concerning two different alkenes has been rarely explored so far. The enantioselective cross R-C reaction still suffers from a series of drawbacks such as low reactivity, enantioselectivity, limited substrate scope and higher catalyst loading. Thus, the development of novel chiral nucleophilic catalyst, for highly enantioselective cross R-C reaction is highly desirable. In this article, we wish to report the application of Xiao-Phos in the highly enantioselective cross intermolecular R-C reaction of vinyl ketone and activated alkenes. Accordingly, a stirred solution of 3-aroyl acrylates 1 (0.2 mmol) and (S,RS)-X8 (0.02 mmol) in toluene (2 mL) was cooled to -20℃. Subsequently, vinyl ketone 2 (0.4 mmol for aryl vinyl ketone and 0.6 mmol for alkyl vinyl ketone) was added in one portion through a syringe. The mixture was stirred at this temperature until completion of 3-aroyl acrylate as indicated by TLC. After completion of the reaction, the reaction mixture was directly purified by silica gel chromatography to afford the desired RC product. What’s more, control experiments demonstrated that the presence of free sulfinamide N-H in Xiao-Phos is crucial for the enantioselective cross R-C reaction. Finally, for more details about (S,RS)-X8 in the enantioselective cross R-C reaction, control experiments monitored by 31P NMR spectroscopy were conducted and the 31P NMR experiments indicated that this cross intermolecular R-C reaction was initiated by the Michael addition of (S,RS)-X8 to vinyl ketone 2.

[1] Rauhut, M. M.; Currier, H. US 3074999, 1963[Chem. Abstr. 1963, 58, 11224a].
[2] For selected reviews on the R-C reaction see: (a) Methot, J. L.; Roush, W. R. Adv. Synth. Catal. 2004, 346, 1035; (b) Aroyan, C. E.; Dermenci, A.; Miller, S. J. Tetrahedron 2009, 65, 4069; (c) Xie, P.; Huang, Y. Eur. J. Org. Chem. 2013, 6213; (d) Bharadwaj, K. C. RSC Adv. 2015, 5, 75923.
[3] For references on the application of the R-C reaction in total synthesis see: (a) Ergüden, J.-K.; Moore, H. W. Org. Lett. 1999, 1, 375; (b) Mergott, D. J.; Frank, S. A.; Roush, W. R. Org. Lett. 2002, 4, 3157; (c) Agapiou, K.; Krische, M. J. Org. Lett. 2003, 5, 1737; (d) Mergott, D. J.; Frank, S. A.; Roush, W. R. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 11955; (e) Stark, L. M.; Pekari, K.; Sorensen, E. J. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 12064; (f) Winbush, S. M.; Mergott, D. J.; Roush, W. R. J. Org. Chem. 2008, 73, 1818; (g) Dermenci, A.; Selig, P. S.; Domaoal, R. A.; Spasov, K. A.; Anderson, K. S.; Miller, S. J. Chem. Sci. 2011, 2, 1568.
[4] For selected progress in the enantioselective intramolecular R-C reaction, see: (a) Aroyan, C. E.; Miller, S. J. J. Am. Chem. Soc. 2007, 129, 256; (b) Aroyan, C. E.; Dermenci, A.; Miller, S. J. J. Org. Chem. 2010, 75, 5784; (c) Osuna, S.; Dermenci, A.; Miller, S. J.; Houk, K. N. Chem. Eur. J. 2013, 19, 14245; (d) Marqués-López, E.; Herrera, R. P.; Marks, T.; Jacobs, W. C.; Könning, D.; de Figueiredo, R. M.; Christmann, M. Org. Lett. 2009, 11, 4116; (e) Wang, X.-F.; Peng, L.; An, J.; Li, C.; Yang, Q.-Q.; Lu, L.-Q.; Gu, F.-L.; Xiao, W.-J. Chem. Eur. J. 2011, 17, 6484; (f) Gong, J.-J.; Li, T.-Z.; Pan, K.; Wu, X.-Y. Chem. Commun. 2011, 47, 1491; (g) Zhang, X.-N.; Shi, M. Eur. J. Org. Chem. 2012, 6271; (h) Takizawa, S.; Nguyen, T. M.-N.; Grossmann, A.; Enders, D.; Sasai, H. Angew. Chem., Int. Ed. 2012, 51, 5423; (i) Takizawa, S.; Nguyen, T. M.-N.; Grossmann, A.; Suzuki, M.; Enders, D.; Sasai, H. Tetrahedron 2013, 69, 1202; (j) Jin, Z.; Yang, R.; Du, Y.; Tiwari, B.; Ganguly, R.; Chi, Y. R. Org. Lett. 2012, 14, 3226; (k) Scanes, R. J. H.; Grossmann, O.; Grossmann, A.; Spring, D. R. Org. Lett. 2015, 17, 2462.
[5] For selected reports on cross RC reactions see: (a) Jih, R. H.; Hakimelahi, G. H.; Chou, C.-T. Tetrahedron Lett. 1992, 33, 6469; (b) Reynolds, T. E.; Binkley, M. S.; Scheidt, K. A. Org. Lett. 2008, 10, 2449; (c) Kumar, R.; Kumar, T.; Mobin, S. M.; Nambothiri, I. N. N. J. Org. Chem. 2013, 78, 5073; (d) Shanbhag, P.; Nareddy, P. R.; Dadwal, M.; Mobin, S. M.; Namboothiri, I. N. N. Org. Biomol. Chem. 2010, 8, 4867; (e) Zhou, R.; Wang, J.; Yu, J.; He, Z. J. Org. Chem. 2013, 78, 10596.
[6] For selected reports on domino cyclization initiated by cross R-C reactions see: (a) Sun, X.; Sengupta, S.; Peterson, J. L.; Wang, H.; Lewis, J. P.; Shi, X. Org. Lett. 2007, 9, 4495; (b) Zhong, C.; Chen, Y.; Petersen, J. L.; Akhmedov, N. G.; Shi, X. Angew. Chem., Int. Ed. 2009, 48, 1279; (c) Yao, W.; Wu, Y.; Wang, G.; Zhang, Y.; Ma, C. Angew. Chem., Int. Ed. 2009, 48, 9713; (d) Ma, J.; Xie, P.; Hu, C.; Huang, Y.; Chen, R. Chem. Eur. J. 2011, 17, 7418; (e) Liu, W.; Zhou, J.; Zheng, C.; Chen, X.; Xiao, H.; Yang, Y.; Guo, Y.; Zhao, G. Tetrahedron 2011, 67, 1768; (f) Xie, P.; Huang, Y.; Lai, W.; Meng, X.; Chen, R. Org. Biomol. Chem. 2011, 9, 6707; (g) Shi, Z.; Tong, Q.; Leong, W. W. Y.; Zhong, G. Chem. Eur. J. 2012, 18, 9802; (h) Shi, Z.; Yu, P.; Loh, T.-P.; Zhong, G. Angew. Chem. Int. Ed. 2012, 51, 7825; (i) Hu, C.; Geng, Z.; Ma, J.; Huang, Y.; Chen, R. Chem. Asian J. 2012, 7, 2032; (j) Hu, C.; Zhang, Q.; Huang, Y. Chem. Asian J. 2013, 8, 1981; (k) Shi, Z.; Loh, T.-P. Angew. Chem., Int. Ed. 2013, 52, 8554; (l) Peng, J.; Huang, X.; Zheng, P.-F.; Chen, Y.-C. Org. Lett. 2013, 15, 5534; (m) Zhang, Y.-Y.; Gurubrahamam, R.; Chen, K. Adv. Synth. Catal. 2015, 357, 2457.
[7] Zhao, Q.-Y.; Pei, C.-K.; Guan, X.-Y.; Shi, M. Adv. Synth. Catal. 2011, 353, 1973.
[8] Dong, X.; Liang, L.; Li, E.; Huang, Y. Angew. Chem., Int. Ed. 2015, 54, 1621.
[9] (a) Zhang, Z.-M.; Chen, P.; Li, W.; Niu, Y.; Zhao, X.-L.; Zhang, J. Angew. Chem., Int. Ed. 2014, 53, 4350; (b) Su, X.; Zhou, W.; Li, Y.; Zhang, J. Angew. Chem., Int. Ed. 2015, 54, 6874; (c) Zhou, W.; Su. X.; Tao, M.; Zhu, C.; Zhao, Q.; Zhang, J. Angew. Chem., Int. Ed. 2015, 54, 14853; (d) Chen, P.; Su, X.; W.; Xiao, Y.; Zhang, J. Tetrahedron 2016, 72, 2700; (e) Zhou, W.; Chen, P.; Tao, M.; Su, X.; Zhao, Q.; Zhang, J. Chem. Commun. 2016, 52, 7612; (f) Zhang, Z.-M.; Xu, B.; Xu, S.; Wu, H.-H.; Zhang, J. Angew. Chem., Int. Ed. 2016, 55, 6324; (g) Hu, H.; Wang, Y.; Qian, D.; Zhang, Z.-M.; Liu, L.; Zhang, J. Org. Chem. Front. 2016, 3, 759.
[10] For reviews related to chiral phosphines catalysis, see: (a) Ye, L.-W.; Zhou, J.; Tang, Y. Chem. Soc. Rev. 2008, 37, 1140; (b) Wei, Y.; Shi, M. Acc. Chem. Res. 2010, 43, 1005; (c) Marinetti, A.; Voituriez, A. Synlett 2010, 174; (d) Wang, S.-X.; Han, X.; Zhong, F.; Wang, Y.; Lu, Y. Synlett 2011, 2766; (e) Zhao, Q.-Y.; Lian, Z.; Wei, Y.; Shi, M. Chem. Commun. 2012, 48, 1724; (f) Xu, L.-W. ChemCatChem 2013, 5, 2775; (g) Wei, Y.; Shi, M. Chem. Rev. 2013, 113, 6659; (h) Fan, Y. C.; Kwon, O. Chem. Commun. 2013, 49, 11588; (i) Wang, Z.; Xu, X.; Kwon, O. Chem. Soc. Rev. 2014, 43, 2927; (j) Wei, Y.; Shi, M. Chem. Asian J. 2014, 9, 2720; (k) Li, W.; Zhang, J. Chem. Soc. Rev. 2016, 45, 1657; (l) Yang, L.; Ma, J. Acta Chim. Sinica 2016, 74, 130(杨丽军, 马军安, 化学学报, 2016, 74, 130.); (m) Zhao, W.-X.; Yang, D.-Y.; Zhang, Y.-H. Chin. J. Org. Chem. 2016, 36, DOI: 10. 6023/ cjoc201603006. (赵文献, 杨代月, 张玉华, 有机化学, 2016, 36, DOI:10.6023/cjoc201603006.)
[11] For selected asymmetric β-aminephosphine catalysis, see: (a) Fang, Y.-Q.; Jacobsen, E. N. J. Am. Chem. Soc. 2008, 130, 5660; (b) Xiao, H.; Chai, Z.; Zheng, C.-W.; Yang, Y.-Q.; Liu, W.; Zhang, J.-K.; Zhao, G. Angew. Chem., Int. Ed. 2010, 49, 4467; (c) Han, X.; Wang, Y.; Zhong, F.; Lu, Y. J. Am. Chem. Soc. 2011, 133, 1726; (d) Zhong, F.; Luo, J.; Chen, G.-Y.; Dou, X.; Lu, Y. J. Am. Chem. Soc. 2012, 134, 10222; (e) Zhong, F.; Han, X.; Wang, Y.; Lu, Y. Chem. Sci. 2012, 3, 1231; (f) Zhong, F.; Dou, X.; Han, X.; Yao, W.; Zhu, Q.; Meng, Y.; Lu, Y. Angew. Chem., Int. Ed. 2013, 52, 943; (g) Fang, Q.; Tadross, P. M.; Jacobsen, E. N. J. Am. Chem. Soc. 2014, 136, 17966; (h) Han, X.; Yao, W.; Wang, T.; Tan, Y. R.; Yan, Z.; Kwiatkowski, J.; Lu, Y. Angew. Chem., Int. Ed. 2014, 53, 5643. (i) Fang, Y.-Q.; Tadross, P. M.; Jacobsen, E. N. J. Am. Chem. Soc. 2014, 136, 17966; (j) Henry, C. E.; Xu, Q.; Fan, Y. C.; Martin, T. J.; Belding, L.; Dudding, T.; Kwon, O. J. Am. Chem. Soc. 2014, 136, 11890; (k) Yao, W.; Dou, X.; Lu, Y. J. Am. Chem. Soc. 2015, 137, 54; (l) Wang, H.-Y.; Zhang, K.; Zhang, C.-W.; Chai, Z.; Cao, D.-D.; Zhang, J.-X.; Zhao, G. Angew. Chem., Int. Ed. 2015, 54, 1775; (m) Li, Y.; Xiao, S.; Zhou, W.; Li, W.; Zhang, J. Chem. Eur. J. 2015, 21, 4224; (n) Lou, Y.-P.; Zheng, C.-W.; Pang, R.-M.; Jin, Q.-W.; Zhao, G.; Li, Z. Org. Lett. 2015, 17, 688; (o) Wang, T.; Yu, Z.; Hoon, D. L.; Phee, C. Y.; Lan, Y.; Lu, Y. J. Am. Chem. Soc. 2016, 138, 265; (p) Yu, Y.-N.; Xu, M.-H. Acta Chim. Sinica 2014, 72, 815. (余月娜, 徐明华, 化学学报, 2014, 72, 815.); (q) Zheng, S.; Jia, L.; Liu, Z.; Jiang, D.; Huang, Y.; Nong, N.; Zhang, Q.; Shi, J. Chin. J. Org. Chem. 2014, 34, 1840. (郑珊, 贾莉, 刘志森, 蒋达洪, 黄艳仙, 农兰平, 张庆, 施继成, 有机化学, 2014, 34, 1840.).
[12] (a) Wang, T.; Yao, W.; Zhong, F.; Pang, G. H.; Lu, Y. Angew. Chem., Int. Ed. 2014, 53, 2964; (b) Zhong, F.; Han, X.; Wang, Y.; Lu, Y. Angew. Chem., Int. Ed. 2011, 50, 7837; (c) Han, X.; Zhong, F.; Wang, Y.; Lu, Y. Angew. Chem., Int. Ed. 2012, 51, 767.
[13] (a) Xia, Y.; Liang, Y.; Chen, Y.; Wang, M.; Jiao, L.; Huang, F.; Liu, S.; Li, Y.; Yu, Z.-X. J. Am. Chem. Soc. 2007, 129, 3470; (b) Huang, G.-T.; Lankau, T.; Yu, C.-H. J. Org. Chem. 2014, 79, 1700.
/
| 〈 |
|
〉 |