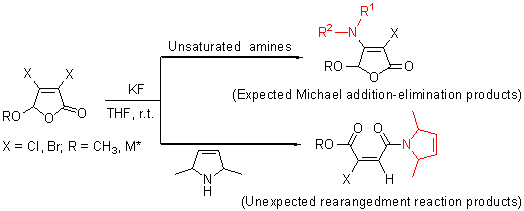
| [1] Juan, H. V. E.; Saad, J. R.; Giordano, O. S.; Garcia, C.; Martin, T.; Martin, V. S.; Sosa, M. E.; Tonn, C. E. J. Nat. Prod. 2008, 71, 190.[2] Wei, M.-X.; Feng, L.; Li, X.-Q.; Zhou, X.-Z.; Shao, Z.-H. Eur. J. Med. Chem. 2009, 44, 3340.[3] Pimentel-Elardo, S. M.; Kozytska, S.; Bugni, T. S.; Ireland, C. M.; Moll, H.; Hentschel, U. Mar. Drugs 2010, 8, 373.[4] Prasad, K. R.; Gandi, V. R. Tetrahedron: Asymmetry 2010, 21, 275.[5] Lattmann, E.; Dunn, S.; Niamsanit, S.; Sattayasai, N. Bioorg. Med. Chem. Lett. 2005, 15, 919.[6] Arayarat, P.; Singh, H.; Lattmann, E. Sci. Asia 2001, 27, 121.[7] Lattmann, E.; Billington, D. C.; Langley, C. A. Drug Des. Discovery 1999, 16, 243.[8] Lattmann, E.; Sattayasai, N.; Schwalbe, C. S.; Niamsanit, S.; Billington, D. C.; Lattmann, P.; Langley, C. A.; Singh. H.; Dunn, S. Curr. Drug Discovery Technol. 2006, 3, 125.[9] Tanoury, G. J.; Chen, M.; Dong, Y.; Forslund, R. E.; Magdziak, D. Org. Lett. 2008, 10, 185.[10] Yu, Z.-L.; Hu, S.-Q.; Li, S.-L.; Fu, Y.-Q. Chin. J. Org. Chem. 2008, 28, 1119 (in Chinese). (郁兆莲, 胡少强, 李森兰, 傅玉琴, 有机化学, 2008, 28, 1119.)[11] Song, X.-M.; Wang, Z.-Y.; Li, J.-X.; Fu, J.-H. Chin. J. Org. Chem. 2009, 29, 1804 (in Chinese). (宋秀美, 汪朝阳, 李建晓, 傅建花, 有机化学, 2009, 29, 1804.)[12] Mo, Y.-Q.; Wang, Z.-Y.; Li, J.-X.; Hong, W.-K. Chin. J. Org. Chem. 2010, 30, 1051 (in Chinese). (莫阳青, 汪朝阳, 李建晓, 洪文坤, 有机化学, 2010, 30, 1051.)[13] Song, X.-M.; Tan, Y.-H.; Li, J.-X.; Wang, Z.-Y. Chin. J. Org. Chem. 2010, 30, 1890 (in Chinese). (宋秀美, 谭越河, 李建晓, 汪朝阳, 有机化学, 2010, 30, 1890.)[14] Lattmann, E.; Lattmann, P.; Singh, H. WO 2004106315, 2004[Chem. Abstr. 2004, 142, 38133].[15] Kurbangalieva, A. R.; Devyatova, N. F.; Bogdanov, A. V.; Berdnikov, E. A.; Mannafov, T. G.; Krivolapov, D. B.; Litvinov, I. A.; Chmutova, G. A. Phosphorus, Sulfur Silicon Relat. Elem. 2007, 182, 607.[16] Devyatova, N. F.; Kosolapova, L. S.; Kurbangalieva, A. R.; Berdnikov, E. A.; Lodochnikova, O. A.; Litvinov, I. A.; Chmutova,G. A. Russ. J. Org. Chem. 2008, 44, 1225.[17] Kurbangalieva, A. R.; Devyatova, N. F.; Kosolapova, L. S.; Lodochnikova, O. A.; Berdnikov, E. A.; Litvinov, I. A.; Chmutova, G. A. Russ. Chem. Bull. 2009, 58, 126.[18] Polezhaeva, N. A.; Volodina, Y. M.; Kalinina, I. V.; Sakhibullina, V. G.; Chmutova, G. A.; Galkin, V. I.; Cherkasov, R. A. Russ. J. Gen. Chem. 2002, 72, 1283.[19] Kurbangalieva, A. R.; Bogdanov, A. V.; Movchan, A. I.; Chmutova, G. A. Russ. J. Org. Chem. 2004, 40, 1216.[20] Fu, J.-H.; Wang, Z.-Y.; Huo, J.-P.; Tan, Y.-H.; Zeng, R.-H. Chin. J. Org. Chem. 2012, 32, 104 (in Chinese). (傅建花, 汪朝阳, 霍景沛, 谭越河, 曾荣华, 有机化学, 2012, 32, 104.)[21] Teitei, T. Aust. J. Chem. 1986, 39, 503.[22] Mellor, J. M.; Wagland, A. M. J. Chem. Soc., Perkin Trans. 1 1989, 997.[23] Dawson, J. R.; Mellor, J. M. Tetrahedron Lett. 1995, 36, 9043.[24] Beccalli, E. M.; Marchesini, A. Tetrahedron 1995, 51, 2353.[25] Hashmi, A. S. K.; Grundl, M. A.; Nass, A. R.; Naumann, F.; Bats, J. W.; Bolte, M. Eur. J. Org. Chem. 2001, 4705.[26] Zhang, E.-J.; Zhang, Y. China Pharm. 1999, 8, 11 (in Chinese). (张恩娟, 张煜, 中国药业, 1999, 8, 11.)[27] Song, C.-S.; Wu, B.-P.; Wu, Q.-L.; Jiao, G.-J.; Li, S.-A. Appl. Chem. Ind. 2005, 34, 484 (in Chinese). (宋长生, 武宝萍, 吴庆利, 焦广俊, 李树安, 应用化工, 2005, 34, 484.)[28] Fu, J.-H.; Wang, Z.-Y.; Hao, Z.-F.; Huo, J.-P. Chin. J. Org. Chem. 2011, 31(9), 1460 (in Chinese). (傅建花, 汪朝阳, 郝志峰, 霍景沛, 有机化学, 2011, 31(9), 1460.)[29] Li, S.-L.; Guo, J.-B.; Yu, Z.-L.; Chen, Q.-H. Chin. J. Chem. 2004, 22, 384.[30] Wang, J.-P.; Cheng, X.-X.; Chen, Q.-H. Acta Chim. Sinica 2006, 64, 686 (in Chinese). (王建平, 程习星, 陈庆华, 化学学报, 2006, 64, 686.)[31] Somnitz, H.; Zellner, R. Phys. Chem. Chem. Phys. 2000, 2, 1899.[32] Pl?tzer, C.; Kleinermanns, K. Phys. Chem. Chem. Phys. 2002, 4, 4877.[33] Finnerty, J. J.; Wentrup, C. J. Org. Chem. 2004, 69, 1909. |