| [1] Richette, P.; Bardin, T. Lancet 2010, 375, 318.
[2] Punzi, L.; Scanu, A.; Ramonda, R.; Oliviero, F. Autoimmun. Rev. 2012, 12, 66.
[3] Dalbeth, N.; Merriman, T. R.; Stamp, L. K. Lacent 2016, 388, 2039.
[4] Kuo, C, F.; Grainge, M. J.; Zhang, W. Y.; Doherty, M. Nat. Rev. Rheumatol. 2015, 11, 649.
[5] Hyndman, D.; Liu, S.; Miner, J. N. Curr. Rheumatol. Rep. 2016, 18, 34.
[6] Enomoto, A.; Kimura, H.; Chairoungdua, A.; Shigeta, Y.; Jutabha, P.; Cha, S. H.; Hosoyamada, M.; Takeda, M.; Sekine, T.; Igarashi, T.; Matsuo, H.; Kikuchi, Y.; Oda, T.; Ichida, K.; Hosoya, T.; Shimokata, K.; Niwa, T.; Kanai, Y.; Endou, H. Nature 2002, 417, 447.
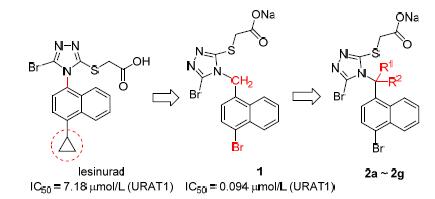
[7] Cai, W.; Liu, W.; Liu, C.; Wang, J.; Zhao, G. Chin. J. Struct. Chem. 2017, 36, 897.
[8] Tian, H.; Wu, J.; Liu, Y.; Xie, Y.; Wang, J.; Zhao, G. Chin. J. Org. Chem. 2017, 37, 1748(in Chinese). (田禾, 吴景卫, 刘钰强, 谢亚非, 王建武, 赵桂龙, 有机化学, 2017, 37, 1748.)
[9] Hoy, S. M. Drugs 2016, 76, 509.
[10] Shen, Z.; Yeh, L. T.; Wallach, K.; Zhu, N.; Kerr, B.; Gillen, M. Clin. Drug Invest. 2016, 36, 443.
[11] Zhang, X.; Wu, J.; Liu, W.; Liu, Y.; Xie, Y.; Shang, Q.; Zhou, Z.; Xu, W.; Tang, L.; Wang, J.; Zhao, G. Med. Chem. 2017, 13, 260.
[12] Miyake, H.; Nakao, Y.; Sasaki, M. Chem. Lett. 2006, 11, 1262.
[13] Miyake, H.; Nakao, Y.; Sasaki, M. Tetrahedron 2007, 63, 10433.
[14] Zhang, X.; Liu, Y.; Xie, Y.; Li, C.; Xin, X.; Xu, W.; Tang, L.; Zhao, G. Drugs Clin. 2015, 30, 1179(in Chinese). (张宪生, 刘钰强, 谢亚非, 李川, 辛晓, 徐为人, 汤立达, 赵桂龙, 现代药物与临床, 2015, 30, 1179.)
[15] Bertus, P.; Szymoniak, J. J. Org. Chem. 2003, 68, 7133.
[16] Michailidis, F. R.; Pupier, M.; Besnard, C.; Burgi, T.; Alexakis, A. Org. Lett. 2014, 16, 4988.
[17] Li, W. J.; Gao, J, J.; Lorenz, J. C.; Xu, J. H.; Johnson, J.; Ma, S. L.; Lee, H.; Grinberg, N.; Busacca, C. A.; Lu, B.; Senanayake, C. H. Org. Process Res. Dev. 2012, 16, 836.
[18] Haudrechy, A.; Chassaing, C.; Riche, C.; Langlois, Y. Tetrahedron 2000, 56, 3181.
[19] Murayama, K.; Yoshioka, T. Tetrahedron Lett. 1968, 11, 1363.
[20] Glaser, R.; Hillebrand, R.; Wycoff, W.; Camasta, C.; Gates, K, S. J. Org. Chem. 2015, 80, 4360.
[21] Cakmak, O.; Kahveci, I.; Demirta, I.; Hokelek, T.; Smith, K. Collect. Czech. Chem. Commun. 2000, 65, 791. |