有机化学 ›› 2026, Vol. 46 ›› Issue (4): 1722-1729.DOI: 10.6023/cjoc202512043 上一篇 下一篇
研究论文
收稿日期:2025-12-28
修回日期:2026-02-10
发布日期:2026-03-13
通讯作者:
邓红平
作者简介:†共同第一作者.
基金资助:
Dongsheng Li, Haowen Zheng, Hongping Deng*( )
)
Received:2025-12-28
Revised:2026-02-10
Published:2026-03-13
Contact:
Hongping Deng
About author:†These authors contributed equally to this work.
Supported by:文章分享
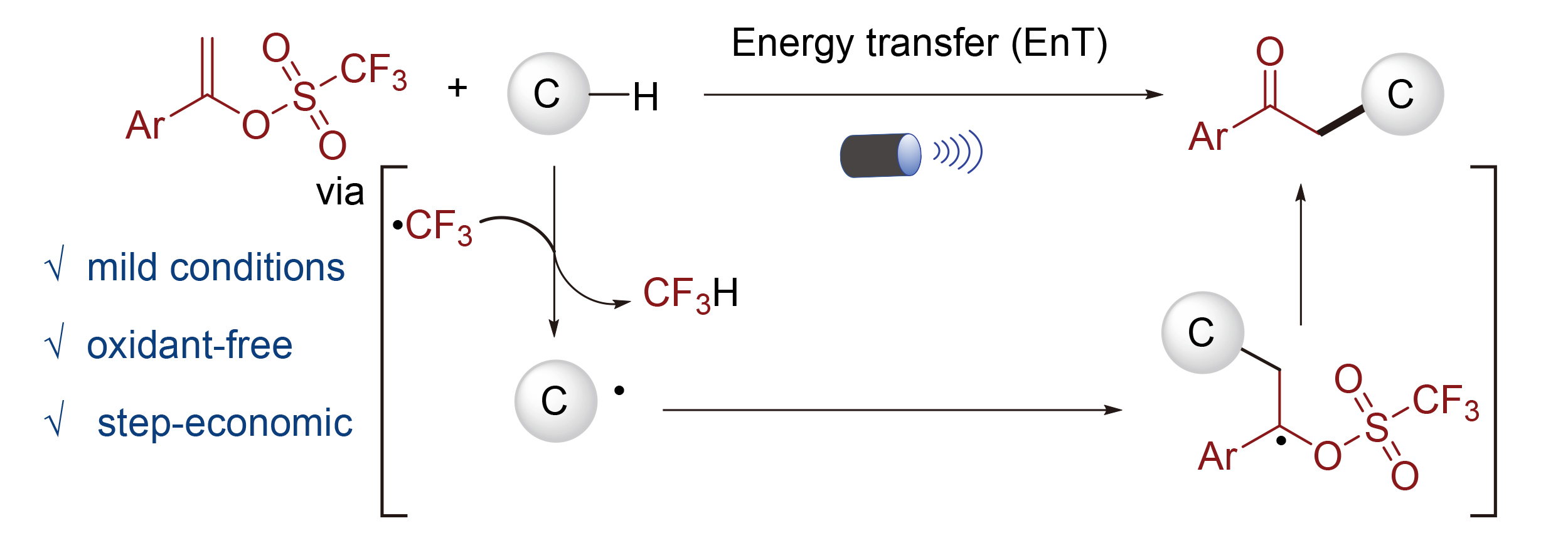
三氟甲基自由基作为一类三氟甲基化反应的重要中间体广为人知. 此外, 其还可作为氢原子转移试剂使用, 但相关研究目前仍较为有限. 发展了一种诱导噻吨酮催化三氟甲基自由基介导的C(sp3)—H键烷基化的方法. 该方法步骤经济性高, 条件温和, 无需外加氧化剂, 以廉价易得含C(sp3)—H键的化合物为原料, 高效合成了一系列苯乙酮衍生物. 机理研究表明, 该反应经过能量转移过程进行, 且是一个链式自由基反应.
李东生, 郑淏文, 邓红平. 光诱导噻吨酮催化三氟甲基自由基介导的C(sp3)—H键的烷基化[J]. 有机化学, 2026, 46(4): 1722-1729.
Dongsheng Li, Haowen Zheng, Hongping Deng. Trifluoromethyl Radical-Mediated C(sp3)—H Alkylation Enabled by Photoinduced Thioxanthone Catalysis[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1722-1729.
| Entry | Photocatalyst | Base (x/equiv.) | Solvent | Yielda/% |
|---|---|---|---|---|
| 1 | PC1 | KOH (2.5) | THF | 74 |
| 2 | PC2 | KOH (2.5) | THF | 0 |
| 3 | PC3 | KOH (2.5) | THF | 82 |
| 4 | PC3 | NaOH (2.5) | THF | 0 |
| 5 | PC3 | K2CO3 (2.5) | THF | 10 |
| 6 | PC3 | KOAc 2.5) | THF | 10 |
| 7 | PC3 | KOH (2.0) | THF | 78 |
| 8 | PC3 | KOH (3.0) | THF | 82 |
| 9b | PC3 | KOH (2.5) | THF | 87 |
| 10b,c | PC3 | KOH (2.5) | EA | 80 |
| 11b,c | PC3 | KOH (2.5) | MeCN | 0 |
| 12b,c | PC3 | KOH (2.5) | DCM | 0 |
| 13b,d | PC3 | KOH (2.5) | THF | 70 |
| 14b | PC3 | — | THF | 0 |
| 15b,e | PC3 | KOH (2.5) | THF | 0 |
| 16b | — | KOH (2.5) | THF | 0 |
| 17b,f | — | KOH (2.5) | THF | 50 |
| Entry | Photocatalyst | Base (x/equiv.) | Solvent | Yielda/% |
|---|---|---|---|---|
| 1 | PC1 | KOH (2.5) | THF | 74 |
| 2 | PC2 | KOH (2.5) | THF | 0 |
| 3 | PC3 | KOH (2.5) | THF | 82 |
| 4 | PC3 | NaOH (2.5) | THF | 0 |
| 5 | PC3 | K2CO3 (2.5) | THF | 10 |
| 6 | PC3 | KOAc 2.5) | THF | 10 |
| 7 | PC3 | KOH (2.0) | THF | 78 |
| 8 | PC3 | KOH (3.0) | THF | 82 |
| 9b | PC3 | KOH (2.5) | THF | 87 |
| 10b,c | PC3 | KOH (2.5) | EA | 80 |
| 11b,c | PC3 | KOH (2.5) | MeCN | 0 |
| 12b,c | PC3 | KOH (2.5) | DCM | 0 |
| 13b,d | PC3 | KOH (2.5) | THF | 70 |
| 14b | PC3 | — | THF | 0 |
| 15b,e | PC3 | KOH (2.5) | THF | 0 |
| 16b | — | KOH (2.5) | THF | 0 |
| 17b,f | — | KOH (2.5) | THF | 50 |
| [1] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [2] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(王曼曼, 习文慧, 吴昊, 白大昌, 有机化学, 2025, 45, 516.)
|
|
|
(g)
|
|
|
(黄广龙, 薛小松, 化学学报, 2024, 82, 132.)
|
|
|
(h)
|
|
|
(李珊, 路俊欣, 刘杰, 蒋绿齐, 易文斌, 化学学报, 2024, 82, 110.)
|
|
| [3] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
| [4] |
(a)
|
|
(b)
|
|
| [5] |
(a)
|
|
(b)
|
|
| [6] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [7] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
| [8] |
|
| [9] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
|
(洪洋, 邓红平, 有机化学, 2025, 45, 1569.)
|
|
|
(i)
|
|
| [10] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
| [11] |
|
| [12] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [13] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [14] |
(a)
|
|
(b)
|
|
| [15] |
|
| [16] |
(a)
|
|
(b)
|
|
|
(c)
|
| [1] | 郭仕勋, 王卫, 张永强. 三线态能量转移促进的自由基链式烯烃氢芳基化反应研究[J]. 有机化学, 2025, 45(5): 1716-1728. |
| [2] | 高玉兰, 龙江琴, 王秋生. 9-芴基甲氧基羰基-酪氨酸掺杂聚乙烯醇室温磷光薄膜性质研究[J]. 有机化学, 2025, 45(4): 1261-1267. |
| [3] | 蒋译锋, 尹艳丽, 江智勇. 卤素原子转移(XAT)策略及应用新进展[J]. 有机化学, 2024, 44(6): 1733-1759. |
| [4] | 孙静, 樊志杰, 杜纪宽, 李硕, 苗艳勤, 赵波, 董海亮, 王华. 一种Y型噻吨酮-咔唑分子的设计、合成及其蓝光和黄光有机发光二极管应用[J]. 有机化学, 2024, 44(4): 1210-1217. |
| [5] | 宋戈洋, 薛东. 光促进过渡金属催化的C-杂原子键偶联反应进展[J]. 有机化学, 2022, 42(8): 2275-2299. |
| [6] | 赵成军, 白治琴, 何建, 刘强. 吡唑化提高喹喔啉酮敏化效率: 氮杂环丁烷的合成及进一步开环反应[J]. 有机化学, 2022, 42(12): 4300-4314. |
| [7] | 张其奇, 林鹏鹏, 杨羚, 谭东航, 冯嗣欣, 王洪根, 李清江. 可见光促进三价铱催化单氟均二苯乙烯Z→E异构化反应[J]. 有机化学, 2020, 40(10): 3314-3326. |
| [8] | 杨滋琦, 刘兴坤, 姜鲁南, 王美. 基于荧光共振能量转移效应的比例计量型水合肼荧光探针的设计、合成及其应用研究[J]. 有机化学, 2019, 39(5): 1483-1488. |
| [9] | 张昊, 俞寿云. 可见光促进的烯烃异构化反应[J]. 有机化学, 2019, 39(1): 95-108. |
| [10] | 杨贺玮, 张宇哲, 李艳杰, 王京翔, 李小萌, 宋健, 张宝, 冯亚青. 发光材料新成员——白光凝胶的现状与未来[J]. 有机化学, 2017, 37(8): 1991-2001. |
| [11] | 阮利衡, 董振诚, 陈春欣, 吴爽, 孙京. 过渡金属镍与可见光双催化体系的研究进展[J]. 有机化学, 2017, 37(10): 2544-2554. |
| [12] | 沈宝星, 钱鹰. 新型萘酰亚胺-氟硼二吡咯荧光分子的合成、荧光共振能量转移及细胞成像[J]. 有机化学, 2016, 36(4): 774-781. |
| [13] | 陈忠林, 李红玲, 韦驾, 肖义, 于海波. 基于激发态能量转移机理比率型荧光探针的研究进展[J]. 有机化学, 2015, 35(4): 789-801. |
| [14] | 张鹏, 张有明, 林奇, 姚虹, 魏太保. 金属离子响应型荧光传感分子的设计原理及研究进展[J]. 有机化学, 2014, 34(7): 1300-1321. |
| [15] | 孙伟, 胡德禹, 吴志兵, 宋宝安, 杨松. 基于罗丹明的重金属和过渡金属阳离子荧光分子探针研究进展[J]. 有机化学, 2011, 31(07): 997-1010. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||