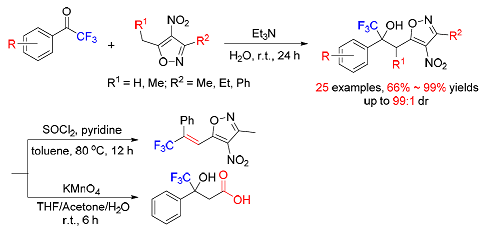
| [1] (a) Welch, J. T.; Eswarakrishman, S. Fluorine in Bioorganic Chemistry, Wiley, New York, 1991.
(b) Filler, R.; Kobayashi, Y.; Yagupolskii, L. M. Organofluorine Compounds in Medicinal Chemistry and Biomedical Applications, Elsevier, Amsterdam, 1993.
(c) Banks, R. E.; Smart, B. E.; Tatlow, C. J. Organofluorine Chemistry:Principles and Comercial Applications, Springer, New York, 1994.
(d) Hiyama, T.; Kanie, K.; Kusumoto, T.; Morizawa, Y.; Shimizu, M. Organofluorine Compounds:Chemistry and Applications, Springer-Verlag, Berlin, 2000.
(e) Kirsch, P. Modern Fluoroorganic Chemistry, Wiley-VCH, Weinheim, 2005.
(f) Uneyama, K. Organofluorine Chemistry, Blackwell, Oxford, 2007.
(g) Ojima, I. Fluorine in Medicinal Chemistry and Chemical Biology, Wiley-Blackwell, Chichester, 2009.
[2] (a) Kirk, K. L. J. Fluorine Chem. 2006, 127, 1013.
(b) Mîller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881.
(c) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320.
(d) Hagmann, W. K. J. Med. Chem. 2008, 51, 4359.
(e) Berger, R.; Resnati, G.; Metrangolo, P.; Weber, E.; Hulliger, J. Chem. Soc. Rev. 2011, 40, 3496.
(f) Ojima, I. J. Org. Chem. 2013, 78, 6358.
(g) Fujiwara, T.; O_Hagan, D. J. Fluorine Chem. 2014, 167, 16.
(h) Wang, J.; Sánchez-Roselló, M.; Aceña, J. L.; Pozo, C.; Sorochinsky, A. E.; Fustero, S.; Soloshonok, V. A.; Liu, H. Chem. Rev. 2014, 114, 2432.
(i) Gillis, E. P.; Eastman, K. J.; Hill, M. D.; Donnelly, D. J.; Meanwell, N. A. J. Med. Chem. 2015, 58, 8315.
[3] (a) Pierce, M. E.; Rodney, L. P. J.; Radesca, L. A.; Lo, Y. S.; Silverman, S.; Moore, J. R.; Islam, Q.; Choudhury, A.; Fortunak, J. M. D.; Nguyen, D.; Luo, C.; Morgan, S. J.; Davis, W. P.; Confalone, P. N.; Chen, C.-Y.; Tillyer, R. D.; Frey, L.; Tan, L.; Xu, F.; Zhao, D.; Thompson, A. S.; Corley, E. G.; Grabowski, E. J. J.; Reamer, R.; Reider, P. J. J. Org. Chem. 1998, 63, 8536.
(b) Corbett, J. W.; Ko, S. S.; Rodgers, J. D.; Gearhart, L. A.; Magnus, N. A.; Bacheler, L. T.; Diamond, S.; Jeffrey, S.; Klabe, R. M.; Cordova, B. C.; Garber, S.; Logue, K.; Trainor, G. L.; Anderson, P. S.; Erickson-Viitanen, S. K. J. Med. Chem. 2000, 43, 2019.
[4] Schenck, H. A.; Lenkowski, P. W.; Choudhury-Mukherjee, I.; Ko, S.-H.; Stables, J. P.; Patel, M. K.; Brown, M. L. Bioorg. Med. Chem. 2004, 12, 979.
[5] Fandrick, D. R.; Reeves, J. T.; Bakonyi, J. M.; Nyalapatla, P. R.; Tan, Z.; Niemeier, O.; Akalay, D.; Fandrick, K. R.; Wohlleben, W.; Ollenberger, S.; Song, J. J.; Sun, X.; Qu, B.; Haddad, N.; Sanyal, S.; Shen, S.; Ma, S.; Byrne, D.; Chitroda, A.; Fuchs, V.; Narayanan, B. A.; Grinberg, N.; Lee, H.; Yee, N.; Brenner, M.; Senanayake, C. H. J. Org. Chem. 2013, 78, 3592.
[6] (a) Betageri, R.; Zhang, Y.; Zindell, R. M.; Kuzmich, D.; Kirrane, T. M.; Bentzien, J.; Cardozo, M.; Capolino, A. J.; Fadra, T. N.; Nelson, R. M.; Paw, Z.; Shih, D.-T.; Shih, C.-K.; Zuvela-Jelaska, L.; Nebozny, G.; Thomson, D. Bioorg. Med. Chem. Lett. 2005, 15, 4761.
(b) Barker, M.; Clackers, M.; Copley, R.; Demaine, D. A.; Humphreys, D.; Inglis, G. G. A.; Johnston, M. J.; Jones, H. T.; Haase, M. V.; House, D.; Loiseau, R.; Nisbet, L.; Pacquet, F.; Skone, P. A.; Shanahan, S. E.; Tape, D.; Vinader, V. M.; Washington, M.; Uings, I.; Upton, R.; McLay, I. M.; Macdonald, S. J. F. J. Med. Chem. 2006, 49, 4216.
[7] (a) Sani, M.; Viani, F.; Binda, M.; Zaffaroni, N.; Zanda, M. Lett. Org. Chem. 2005, 2, 447.
(b) Betageri, R.; Gilmore, T.; Kuzmich, D.; Kirrane, T. M.; Bentzien, J.; Wiedenmayer, D.; Bekkali, Y.; Regan, J.; Berry, A.; Latli, B.; Kukulka, A. J.; Fedra, T. N.; Nelson, R. N.; Goldrick, S.; Zuvela-Jelaska, L.; Souza, D.; Pelletier, J.; Dinallo, R.; Panzenbeck, M.; Torcellini, C.; Lee, H.; Pack, E.; Harcken, C.; Nabozny, G.; Thomson, D. S. Bioorg. Med. Chem. Lett. 2011, 21, 6842.
[8] Carceller, E.; Merlos, M.; Giral, M.; Balsa, D.; Garciía-Rafanell, J.; Forn, J. J. Med. Chem. 1996, 39, 487.
[9] For reviews, see:(a) Nie, J.; Guo, H.-C.; Cahard, D.; Ma, J.-A. Chem. Rev. 2011, 111, 455.
(b) Kelly, C. B.; Mercadante, M. A.; Leadbeater, N. E. Chem. Commun. 2013, 49, 11133.
[10] For selected examples:(a) Hara, N.; Tamura, R.; Funahashi, Y.; Nakamura, S. Org. Lett. 2011, 13, 1662.
(b) Zhang, G.-W.; Meng, W.; Ma, H.; Nie, J.; Zhang, W.-Q.; Ma, J.-A. Angew. Chem., Int. Ed. 2011, 50, 3538.
(c) Cui, H.-F.; Wang, L.; Yang, L.-J.; Nie, J.; Zheng, Y.; Ma, J.-A. Tetrahedron 2011, 67, 8470.
(d) Li, X.-J.; Xiong, H.-Y.; Hua, M.-Q.; Nie, J.; Zheng, Y.; Ma, J.-A. Tetrahedron Lett. 2012, 53, 2117.
(e) Zheng, Y.; Xiong, H.-Y.; Nie, J.; Hua, M.-Q.; Ma, J.-A. Chem. Commun. 2012, 48, 4308.
(f) Luo, R.; Li, K.; Hu, Y.; Tang, W. Adv. Synth. Catal. 2013, 355, 1297.
(g) Zong, H.; Huang, H.; Bian, G.; Song, L. J. Org. Chem. 2014, 79, 11768.
(h) Wang, L.; Liu, N.; Dai, B.; Ma, X.; Shi, L. RSC Adv. 2015, 5, 10089.
(i) Jamal, Z.; Teo, Y.-C. RSC Adv. 2015, 5, 26949.
(j) Zhang, D.; Tanaka, F. Adv. Synth. Catal. 2015, 357, 3458.
(k) Tao, R.; Yin, X.-J.; Wang, K.-H.; Niu, Y.-Z.; Wang, Y.-L.; Huang, D.-F.; Su, Y.-P.; Wang, J.-X.; Hu, Y.-L.; Fu, Y.; Du, Z.-Y. Chin. Chem. Lett. 2015, 26, 1046.
(l) Jing, Z.; Bai, X.; Chen, W.; Zhang, G.; Zhu, B.; Jiang, Z. Org. Lett. 2016, 18, 260.
(m) Lutete, L. M.; Miyamoto, T.; Ikemoto, T. Tetrahedron Lett. 2016, 57, 1220.
(n) Cook, A. M.; Wolf, C. Angew. Chem., Int. Ed. 2016, 55, 2929.
(o) Lee, K.; Silverio, D. L.; Torker, S.; Robbins, D. W.; Haeffner, F.; Mei, F. W.; Hoveyda, A. H. Nat. Chem. 2016, 8, 768.
(p) Matador, E.; Monge, D.; Fernández, R.; Lassaletta, J. M. Green Chem. 2016, 18, 4042.
(q) Mszar, N. W.; Mikus, M. S.; Torker, S.; Haeffner, F.; Hoveyda, A. H. Angew. Chem., Int. Ed. 2017, 56, 8736.
(r) Noda, H.; Amemiya, F.; Weidner, K.; Kumagai, N.; Shibasaki, M. Chem. Sci. 2017, 8, 3260.
(s) Bai, X.; Zeng, G.; Shao, T.; Jiang, Z. Angew. Chem., Int. Ed. 2017, 56, 3684.
(t) Zheng, Y.; Tan, Y.; Harms, K.; Marsch, M.; Riedel, R.; Zhang, L.; Meggers, E. J. Am. Chem. Soc. 2017, 139, 4322.
[11] Bandini, M.; Sinisi, R. Org. Lett. 2009, 11, 2093.
[12] Zhang, Y.; Wei, B.-W.; Zou, L.-N.; Kang, M.-L.; Luo, H.-Q.; Fan, X.-L. Tetrahedron 2016, 72, 2472.
[13] Czerwinski, P.; Molga, E.; Cavallo, L.; Poater, A.; Michalak, M. Chem. Eur. J. 2016, 22, 8089.
[14] (a) Grieco, P. A. Organic Synthesis in Water, Blackie, London, 1998.
(b) Lindstrom, U. M. Organic Reactions in Water:Principles, Strategies and Applications, Blackwell, Oxford, U. K., 2007.
(c) Li, C.-J.; Chan, T.-H. Comprehensive Organic Reactions in Aqueous Media, Wiley, New York, 2007.
[15] For selected reviews:(a) Lindstrom, U. M. Chem. Rev. 2002, 102, 2751.
(b) Head-Gordon, T.; Hura, G. Chem. Rev. 2002, 102, 2651.
(c) Hayashi, Y. Angew. Chem., Int. Ed. 2006, 45, 8103.
(d) Mase, N.; Barbas Ⅲ, C. F. Org. Biomol. Chem. 2010, 8, 4043.
[16] For selected reviews:(a) Li, C.-J. Chem. Rev. 2005, 105, 3095.
(b) Li, C.-J.; Chen, L. Chem. Soc. Rev. 2006, 35, 68.
(c) Chanda, A.; Fokin, V. V. Chem. Rev. 2009, 109, 725.
(d) Butler, R. N.; Coyne, A. G. Chem. Rev. 2010, 110, 6302.
(e) Simon, M.-O.; Li, C.-J. Chem. Soc. Rev. 2012, 41, 1415.
(f) Gawande, M. B.; Bonifacio, V. D. B.; Luque, R.; Branco, P. S.; Varma, R. S. Chem. Soc. Rev. 2013, 42, 5522.
(g) Levin, E.; Ivry, E.; Diesendruck, C. E.; Lemcoff, N. G. Chem. Rev. 2015, 115, 4607.
(h) Butler, R. N.; Coyne, A. G. Org. Biomol. Chem. 2016, 14, 9945.
[17] For selected examples:(a) Phippen, C. B. W.; Beattie, J. K.; McErlean, C. S. P. Chem. Commun. 2010, 46, 8234.
(b) Fu, X.-P.; Liu, L.; Wang, D.; Chen, Y.-J.; Li, C.-J. Green. Chem. 2011, 13, 549.
(c) Sakakura, A.; Koshikari, Y.; Akakura, M.; Ishihara, K. Org. Lett. 2011, 14, 30.
(d) Paladhi, S.; Chauhan, A.; Dhara, K.; Tiwari, A. K.; Dash, J. Green. Chem. 2012, 14, 2990.
(e) Islam, S.; Larrosa, I. Chem. Eur. J. 2013, 19, 15093.
(f) Sengoden, M.; Punniyamurthy, T. Angew. Chem. Int. Ed. 2013, 52, 572.
(g) Paladhi, S.; Bhati, M.; Panda, D.; Dash, J. J. Org. Chem. 2014, 79, 1473.
(h) Thakur, P. B.; Meshram, H. M. RSC Adv. 2014, 4, 5343.
(i) Thakur, P. B.; Meshram, H. M. RSC Adv. 2014, 4, 6019.
(j) Yu, J.-S.; Liu, Y.-L.; Tang, J.; Wang, X.; Zhou, J. Angew. Chem. Int. Ed. 2014, 53, 9512.
(k) Yang, F.; Klumphu, P.; Liang, Y.-M.; Lipshutz, B. H. Chem. Commun. 2014, 50, 936.
(l) Zhang, F.-Z.; Tian, Y.; Li, G.-X.; Qu, J. J. Org. Chem. 2015, 80, 1107.
(m) SaiPrathima, P.; Srinivas, K.; Mohan Rao, M. Green. Chem. 2015, 17, 2339.
(n) Bhattacharjya, A.; Klumphu, P.; Lipshutz, B. H. Nat. Commun. 2015, 6, 7401.
(o) Cho, B. S.; Chung, Y. K. Chem. Commun. 2015, 51, 14543.
(p) Xiao, J.; Wen, H.; Wang, L.; Xu, L.; Hao, Z.; Shao, C.-L.; Wang, C.-Y. Green Chem. 2016, 18, 1032.
(q) Zhang, Y.; Wei, B.-W.; Lin, H.; Zhang, L.; Liu, J.-X.; Luo, H.-Q.; Fan, X.-L. Green Chem. 2015, 17, 3266.
(r) Ren, N.; Nie, J.; Ma, J.-A. Green Chem. 2016, 18, 6609.
[18] (a) Wang, J.; Kong, W.-G.; Li, F.; Liu, J.; Shen, Q.; Liu, L.; Zhao, W.-X. Org. Biomol. Chem. 2015, 13, 5399.
(b) Li, F.; Wang, J.; Xu, M.; Zhao, X.; Zhou, X.; Zhao, W.-X.; Liu, L. Org. Biomol. Chem. 2016, 14, 3981.
(c) Wang, J.; Li, F.; Shen, Q.; Pei, W.; Zhao, W.-X.; Liu, L. Synthesis 2016, 48, 441.
(d) Wang, J.; Li, F.; Xie, H.; Xu, M.; Zhao, X.; Liu, L.; Zhao, W.-X. Appl. Organomet. Chem. 2017, 31, e3545.
[19] CCDC 1558716(3a) and CCDC 1559048(5) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Center via www.ccdc.cam.ac.uk/data_request/cif.
[20] (a) Creary, X. J. Org. Chem. 1987, 52, 5026.
(b) Chong, J. M.; Mar, E. K. J. Org. Chem. 1991, 56, 893.
(c) Prakash, G. K. S.; Panja, C.; Vaghoo, H.; Surampudi, V.; Kultyshev, R.; Mandal, M.; Rasul, G.; Mathew, T.; Olah, G. A. J. Org. Chem. 2006, 71, 6806.
(d) Cheng, H.; Pei, Y.; Leng, F.; Li, J.; Liang, A.; Zou, D.; Wu, Y.; Wu, Y. Tetrahedron Lett. 2013, 54, 4483.
[21] (a) Adamo, M. F. A.; Duffy, E. F. Org. Lett. 2006, 8, 5157.
(b) Adamo, M. F. A.; Suresh, S. Tetrahedron 2009, 65, 990.
(c) Zhang, J.; Liu, X.; Ma, X.; Wang, R. Chem. Commun. 2013, 49, 9329.
[22] Gao, J.-R.; Wu, H.; Xiang, B.; Yu, W.-B.; Han, L.; Jia, Y.-X. J. Am. Chem. Soc. 2013, 135, 2983.
[23] Fiandra, C. D.; Piras, L.; Fini, F.; Disetti, P.; Moccia, M.; Adamo, M. F. A. Chem. Commun. 2012, 48, 3863.
[24] McBee, E. T.; Kim, Y. S.; Braendlin, H. P. J. Am. Chem. Soc. 1962, 84, 3154. |