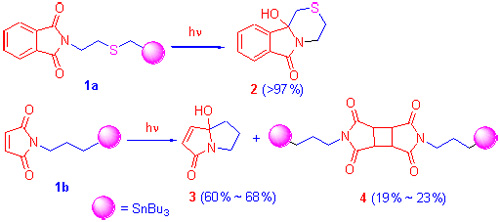
合成了两种新的分子内给受电子体系N-[2-(2-三丁基锡甲硫基)乙基]邻苯二甲酰亚胺(1a)和N-(3-三丁基锡丙基)马来酰亚胺(1b), 并在甲醇、乙腈-30%水、乙腈中进行了光诱导单电子转移反应. 化合物1a在光诱导下发生分子内单电子转移反应, 以很高的产率和区域选择性生成环胺醇2. 化合物1b在光诱导下发生分子内单电子转移反应生成环胺醇3, 同时有[2+2]环加成副反应产物4生成. 以上所有新化合物的结构经质谱和核磁共振谱验证.
金英学
,
王欣
,
曲凤玉
,
谭广慧
,
岳群峰
. N-(末端三丁基锡取代基)酰亚胺的光诱导单电子转移成环反应[J]. 有机化学, 2012
, 32(12)
: 2363
-2367
.
DOI: 10.6023/cjoc201205027
Two novel intramolecularly electronical donor-acceptor systemes N-[2-(2-tributylstannylmethyl-sulfenyl)-ethyl]phthalimide (1a) and N-(3-tributylstanylpropyl)maleimide (1b), were synthesized, and their photoinduced single electron transfer (SET) reactions in MeOH, CH3CN-30% H2O and pure CH3CN were carried out, respectively. Compound 1a occurred intramolecular SET to provide cyclic amidol 2 with highly yield and high regioselectivity. Compound 1b occurred intramolecular single electron transfer (SET) to provide cyclic amidol 3 with small amount of photoinduced [2+2] dimerization product 4. Structures of all new compounds were characterized by MS (FAB) and NMR data.
[1] Kanaoka, Y.; Migita, Y.; Koyama, K. Tetrahedron Lett. 1973, 1193.
[2] Kanaoka, Y.; Koyama, K.; Flippen, J. J. Am. Chem. Soc. 1974, 96, 4719.
[3] Machida, M.; Takechi, H.; Kanaoka, Y. Synthesis 1982, 1078.
[4] Yoon, U. C.; Mariano, P. S. Acc. Chem. Res. 2001, 34, 523.
[5] Cho, D. W.; Yoon, U. C.; Mariano, P. S. Acc. Chem. Res. 2011, 44, 204.
[6] Tan G.-H.; Wei, S.-Q.; Yue, Q.-F.; Zhao, J.-M.; Jin, Y.-X. Chin. J. Org. Chem. 2010, 30, 1021 (in Chinese).
(谭广慧, 魏树权, 岳群峰, 赵俊明, 金英学, 有机化学, 2010, 30, 1021.)
[7] Jin, Y.-X.; Yuan, W.; Qu, F.-Y.; Yoon, U. C.; Wei, S.-Q.; Yue, Q.-F.; Tan, G.-H. Acta Chim. Sinica 2011, 69, 2407 (in Chinese).
(金英学, 苑望, 曲凤玉, 尹雄灿, 魏树权, 岳群峰, 谭广慧, 化学学报, 2011, 69, 2407.)
[8] Thompson, M. C.; Busch, D. H. J. Am. Chem. Soc. 1962, 84, 1762.
[9] Pedersen, C. J. J. Am. Chem. Soc. 1967, 89, 7017.
[10] Zhang, X. M.; Yeh, S. P.; Hong, S.; Freccero, M.; Albin, A.; Falvery, D. F.; Mariano, P. S. J. Am. Chem. Soc. 1994, 116, 4211.
[11] Su, Z. Y.; Falvery, D. E.; Yoon, U. C.; Oh, S. W.; Mariano, P. S. J. Am. Chem. Soc. 1998, 120, 10676.