

马来海松酸三甲酯区域选择性格氏加成反应研究
收稿日期: 2012-08-08
修回日期: 2012-09-25
网络出版日期: 2012-09-26
基金资助
973计划前期研究专项(No. 2011CB512005);国家自然科学基金(Nos. 81260472, 21101035);广西自然科学基金重点(No. 2011GXNSFD018010);广西新世纪十百千人才工程人选(No. 2007228)资助项目.
Study on the Regioselectivie of Grignard Reagent Addition Reaction of Maleopimaric Acid Trimethyl Ester
Received date: 2012-08-08
Revised date: 2012-09-25
Online published: 2012-09-26
Supported by
Project supported by the Project 973 (No. 2011CB512005), the National Natural Science Foundation of China (Nos. 81260472, 21101035), the Natural Science Foundation of Guangxi Province (No. 2011GXNSFD018010) and the Project of Ten, Hundred, Thousand Distinguished Talents in New Century of Guangxi Province (No. 2007228).
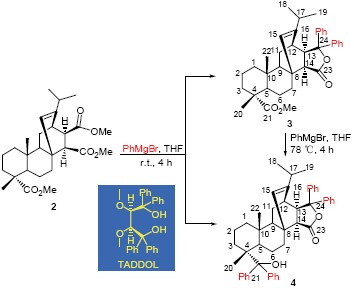
研究了格氏试剂与马来海松酸三甲酯(2)的各官能团的反应活性差异, 获得了2个区域选择性加成产物(化合物3和4), 各化合物的结构均经元素分析、NMR和MS表征, 用COSY, HMQC和HMBC进一步对化合物3进行了结构分析, 并由X 射线单晶衍射确认了其立体结构. 实验结果表明, 2上的3个甲酯基的位阻影响了加成反应的选择性, 内酯3的生成阻止了其进一步生成TADDOL (tetraaryl-1,3-dioxolane-4,5-dimethanols)类产物, 但反应不影响松香环式原有的立体结构. 产物3和4有望作为手性衍生试剂.
姚贵阳 , 韦京辰 , 戴伟龙 , 杨达 , 潘英明 , 王恒山 . 马来海松酸三甲酯区域选择性格氏加成反应研究[J]. 有机化学, 2013 , 33(01) : 138 -142 . DOI: 10.6023/cjoc201208006
Two regioselectivite adducts (compounds 3 and 4) were firstly synthesized from the Grignard reagent and maleopimaric acid trimethyl ester (2). The structures of all compounds were characterized by elemental analyses, NMR and MS, and stero-structures of 3 were further determined by COSY, HMQC and HMBC. The spatial structure of 3 was further confirmed by X-ray single crystal diffraction analysis method. Owing to the addition reaction selectivity of 2 was dependent on the steric hindrance of three methyl groups, the TADDOL (tetraaryl-1,3-dioxolane-4,5-dimethanols) product was prevented because of the formation of lactone 3, but the stereo-structure of the rosin-ring did not changed. Products of 3 and 4 are expected to apply as chiral derivatization reagents.

[1] Thomas, J. W.; Jolene, E. T. J. Org. Chem. 2000, 65, 5.
[2] Ann, E. L.; Thomas, J. W. Org. Lett. 2006, 8, 2823.
[3] Joseph G. Chirality 2008, 20, 5.
[4] Jiang X. X.; Zhang, Y. F.; Chan A. S. C.; Wang, R. Org. Lett. 2008, 11, 153.
[5] Jiang, X. X.; Zhang, Y. F.; Liu, X.; Zhang, G.; Lai, L. H.; Wu, L. P.; Zhang, J. N.; Wang, R. J. Org. Chem. 2009, 74, 5562.
[6] Jiang, X. X.; Zhang, Y. F.; Wu, L. P.; Zhang, G.; Liu, X.; Zhang, H. L.; Fu, D.; Wang, R. Adv. Synth. Catal. 2009, 351, 2096.
[7] Jiang, X. X.; Zhang, G.; Fu, D.; Cao, Y. M.; Shen, F. F.; Wang, R. Org. Lett. 2010, 12, 1544.
[8] Ye, F. G.; Wang, H. S.; Huang, B. J.; Zhao, S. L. Electrophoresis 2010, 31, 1488.
[9] Wang, H. S.; Zhao, S. L.; He, M.; Zhao, Z. C.; Pan, Y. M.; Liang, Q. J. Sep. Sci. 2007, 30, 2748.
[10] Zhao, S. L.; Wang, H. S.; Pan, Y. M.; He, M.; Zhao, Z. C. J. Chromatogr. A 2007, 1145, 246.
[11] Zhao, S. L.; Wang, H. S.; Zhang, R. C.; Tang, L. D.; Liu, Y. M. Electrophoresis 2006, 27, 17.
[12] Wang, H. S.; Zhang, R. C.; Zhao, S. L.; Tang, L. D.; Pan, Y. M. Anal. Chim. Acta 2006, 560, 64.
[13] Wang, H. S.; Tian, X. Y.; Yang, D.; Pan, Y. M.; Wu, Q.; He, C. H. Tetrahedron: Asymmetry 2011, 22, 381.
[14] Nishi, H.; Kuwahara, Y. J. Biochem. Biophys. Methods 2001, 48, 89.
[15] Nishi, H.; Kuwahara, Y. J. Pharm. Biomed. 2002, 27, 577.
[16] Chen, J.; Du, Y.; Zhu, F.; Chen, B. J. Chromatogr. A 2010, 1217, 7158.
[17] Zaher, M.; Baussanne, I.; Ravelet, C.; Halder, S.; Haroun, M.; Fize, J.; Décout, J.-L.; Peyrin, E. J. Chromatogr. A 2008, 1185, 291.
[18] Wang, H. S.; He, C. H.; Pan, Y. M.; Yao, G. Y.; Wu, Q.; Deng, H. G. J. Inclusion Phenom. Macrocyclic Chem. 2011, 73, 177.
[19] Sheldrick, G. M. SHELXTL, Version 6.10, Bruker AXS Inc., Madison, Wisconsin, USA, 2000.
[20] Pan, Y. M.; Yang, L.; Wang, H. S.; Zhang, R. C.; Zhang, Y. Acta Crystallogr., Sect. E 2006, 62, o5701.
/
| 〈 |
|
〉 |