

亚甲氧基桥键弯曲型液晶合成与性能研究
收稿日期: 2013-01-16
修回日期: 2013-04-01
网络出版日期: 2013-05-08
基金资助
国家自然科学基金(No.11074054);湖北省教育厅科技基金(Z20091801);武汉工业学院研究生教育创新基金(No. 2011cx003)资助项目
Synthesis and Property of Oxymethylene-Bridged Bent-core Liquid Crystals
Received date: 2013-01-16
Revised date: 2013-04-01
Online published: 2013-05-08
Supported by
Project supported by the National Natural Science Foundation of China (No.11074054), the Scientific Research Found of Hubei Provincial Education Department (No. 20091801) and the Educational and Innovative Foundation for the Graduate Students of Wuhan Polytechnic University (No. 2011cx003)
王勇丽 , 杜琼 , 王国华 , 张智勇 , 戴志群 , 关金涛 , 项颖 . 亚甲氧基桥键弯曲型液晶合成与性能研究[J]. 有机化学, 2013 , 33(9) : 2010 -2015 . DOI: 10.6023/cjoc201301033
A novel series of bent-core molecules with a central bent core based on 2,5-bis(4-hydroxyphenyl)-1,3,4-oxadiazole, oxymethylene-bridged (CH2O), and alkyl tails with carbon numbers m=3, 5, 6, 7, 8, 9, 10 were prepared. Structures of the titled compounds were confirmed by 1H NMR, 13C NMR, IR, MS techniques, their transition temperatures and phase transition were investigated using differential scanning calorimetric (DSC) and polarizing optical microscopy (POM). Comparing with ester-bridged, target compounds 6a~6g have right chemical structure, possessed low melting points that below 100 ℃ (except 6c) and exhibited liquid crystal phases. Compounds 6d~6g exist wide phase temperature range and show odd-even effect.
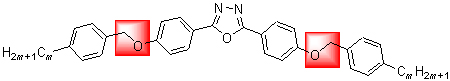
Key words: bent-core liquid crystal; oxadiazol; oxymethylene; synthesis; low melting points
/
| 〈 |
|
〉 |