

Platencin(平板素)的全合成研究进展
收稿日期: 2016-04-17
修回日期: 2016-05-10
网络出版日期: 2016-05-17
基金资助
国家自然科学基金(Nos.21172246,21290180,21472210)和中国科学院青年创新促进会(No.2012202)资助项目.
Progresses in Total Synthesis of Platencin
Received date: 2016-04-17
Revised date: 2016-05-10
Online published: 2016-05-17
Supported by
Project supported by the National Natural Science Foundation of China (Nos.21172246, 21290180, 21472210), and the Youth Innovation Promotion Association CAS (No.2012202).
刘楝 , 周淑晶 , 孙炳峰 . Platencin(平板素)的全合成研究进展[J]. 有机化学, 2016 , 36(9) : 2091 -2104 . DOI: 10.6023/cjoc201604034
Platencin was isolated from a new strain of Streptomyces platensis MA7339 found in a soil sample collected in Spain. It has a novel molecular structure and exhibits a broad spectrum of antibacterial activity. In this review, the progresses since 2010 in the total synthesis of platencin are summarized with an emphasis on the synthetic strategies of the routes.
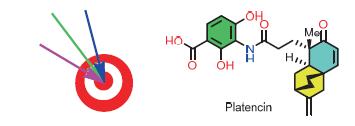
Key words: platencin; total synthesis; synthetic strategy
[1] Newman,D.J.;Cragg,G.M.J.Nat.Prod.2012,75,311.
[2] Wang,J.;Soisson,S.M.;Young,K.;Shoop,W.;Kodali,S.;Galgoci,A.;Painter,R.;Parthasarathy,G.;Tang,Y.S.;Cummings,R.;Ha,S.;Dorso,K.;Motyl,M.;Basilio,A.;Tormo,J.R.;Genilloud,O.;Vicente,F.;Pelaez,F.;Colwell,L.;Lee,S.H.;Michael,B.;Felcetto,T.;Gill,C.;Silver,L.L.;Hermes,J.D.;Bartizal,K.;Barrett,J.;Schmatz,D.;Becker,J.W.;Cully,D.;Singh,S.B.Nature 2006,441,358.
[3] Nicolaou,K.C.;Tria,G.S.;Edmonds,D.J.Angew.Chem.,Int.Ed.2008,47,1780.
[4] Palanichamy,K.;Kaliappan,K.P.Chem.Asian J.2010,5,668.
[5] Hirai,S.;Nakada M.Tetrahedron Lett.2010,51,5076.
[6] Li,P.;Yamamoto.H.Chem.Commun.2010,46,6294.
[7] Singh,V.;Sahu,B.C.;Bansal,V.;Mobin,S.M.Org.Biomol.Chem.2010,8,4472.
[8] Singh,V.;Das,B.;Mobin.S.M.Synlett 2013,24,1583.
[9] Leung,G.Y.C.;Li,H.;Toh,Q.-Y.;Ng,A.M.-Y.;Sum,R.J.;Bandow,J.E.;Chen,D.Y.-K.Eur.J.Org.Chem.2011,183.
[10] Palanichamy,K.;Subrahmanyam,A.V.;Kaliappan,K.P.Org Biomol.Chem.2011,9,7877.
[11] Yoshimistu,T.;Nojima,S.;Hashimoto,M.;Tanaka,T.Org.Lett.2011,13,3698.
[12] Moustafa,G,A;Saku,Y.;Aoyama,H;Yoshimitsu,T.Chem.Commun.2014,50,15706.
[13] Yadav,J.S.;Goreti,R.;Pabbaraja,S.;Sridhar,B.Org.Lett.2013,15,3782.
[14] Zhu,L.;Zhou,C.;Yang,W.;He,S.;Cheng,G.-J.;Zhang,X.;Lee,C.-S.J.Org.Chem.2013,78,7912.
[15] Chang,E.L.;Schwartz,B.D.;Draffan,A.G.;Banwell,M.G.Chem.Asian J.2015,10,427.
[16] Banwell,M.G.;Edwards,A.J.;Harfoot,G.J.;Jolliffe,K.A.;McLeod,M.D.;McRae,K.J.;Stewart,S.G.;Vçgtle,M.Pure Appl.Chem.2003,75,223.
[17] Muhammad,R.N.;Draffan,A.G.;Banwell,M.G.;Willis,A.C.Synlett 2016,27,61.
[18] Wang,J.;Sun,W.-B.;Li,Y.-Z.;Wang,X.;Sun,B.-F.;Lin,G.-Q.;Zou,J.-P.Org.Chem.Front.2015,2,674.
/
| 〈 |
|
〉 |